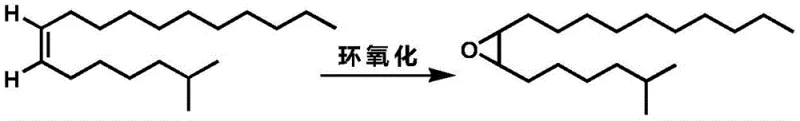
Advanced Synthesis of cis-7,8-epoxy-2-methyloctadecane for Commercial Agrochemical Applications
The agricultural sector continuously demands higher efficacy in pest management solutions, driving the need for precise synthetic routes for semiochemicals such as the gypsy moth pheromone. Patent CN115353437A, published in late 2022, introduces a groundbreaking methodology for synthesizing cis-2-methyl-7-octadecene and its subsequent epoxidation to cis-7,8-epoxy-2-methyloctadecane. This technical disclosure addresses long-standing challenges in stereochemical control that have plagued previous manufacturing attempts. By leveraging an N-sulfonyl hydrazone and phosphonium salt coupling strategy, the inventors have established a pathway that inherently suppresses the formation of unwanted trans-isomers. For R&D directors and procurement specialists, this represents a significant shift away from reliance on precious metal catalysts towards more robust organic synthesis techniques. The reported yields ranging from 68% to 96% coupled with purity exceeding 98.8% demonstrate a level of process reliability that is critical for consistent agrochemical supply chains. This report analyzes the technical merits of this invention and its implications for cost reduction in agrochemical intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of the key intermediate cis-2-methyl-7-octadecene relied heavily on the cis-hydrogenation of 2-methyl-7-octadecyne using Lindlar catalysts or bulky boron reagents. While theoretically sound, these conventional methods suffer from inherent selectivity issues that compromise the quality of the final pheromone product. The hydrogenation process frequently generates trans-2-methyl-7-octadecene as a persistent byproduct, which is difficult to separate due to similar physical properties. This contamination drastically lowers the purity of the subsequent epoxidation product, rendering it less effective for field applications in pest monitoring and trapping. Furthermore, the use of heterogeneous palladium catalysts introduces risks of heavy metal residue, necessitating expensive and time-consuming purification steps to meet regulatory standards. The variability in catalyst activity also leads to inconsistent batch-to-batch results, creating significant bottlenecks for supply chain heads managing inventory levels. These technical deficiencies translate directly into higher operational costs and reduced reliability for manufacturers of high-purity agrochemical intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN115353437A utilizes a one-step substitution reaction between an N-sulfonyl hydrazone and a phosphonium salt under the influence of an organic base. This approach fundamentally bypasses the hydrogenation step, thereby eliminating the mechanistic pathway that leads to trans-isomer formation. The reaction proceeds smoothly at low temperatures between -80°C and -30°C, ensuring high stereocontrol throughout the carbon-carbon bond formation. By avoiding transition metals entirely, the process simplifies the downstream workup, requiring only standard aqueous quenching and chromatographic purification. The resulting cis-2-methyl-7-octadecene exhibits exceptional purity, which directly correlates to the high yield and purity of the final cis-7,8-epoxy-2-methyloctadecane product. This strategic shift not only enhances product quality but also streamlines the overall manufacturing workflow, offering substantial cost savings potential through reduced material waste and simplified processing.
Mechanistic Insights into N-sulfonyl Hydrazone Substitution
The core of this innovation lies in the precise generation of a diazo intermediate from the N-sulfonyl hydrazone precursor under basic conditions. When treated with strong organic bases such as n-butyllithium or lithium diisopropylamide, the hydrazone undergoes deprotonation and subsequent elimination of the sulfonyl group to form a reactive diazo species. This species then interacts with the phosphonium salt, likely proceeding through a carbene or ylide-like transition state that favors the formation of the cis-alkene geometry. The steric environment provided by the long alkyl chains and the specific choice of the aromatic sulfonyl group play crucial roles in directing the stereochemical outcome. Maintaining the reaction temperature strictly below -30°C is vital to prevent thermal decomposition of the diazo intermediate and to minimize side reactions that could lead to isomerization. This low-temperature protocol ensures that the kinetic product, which is the desired cis-isomer, is formed exclusively over the thermodynamic trans-isomer. Understanding this mechanistic nuance is essential for R&D teams aiming to replicate or optimize this process for commercial scale-up of complex organic intermediates.
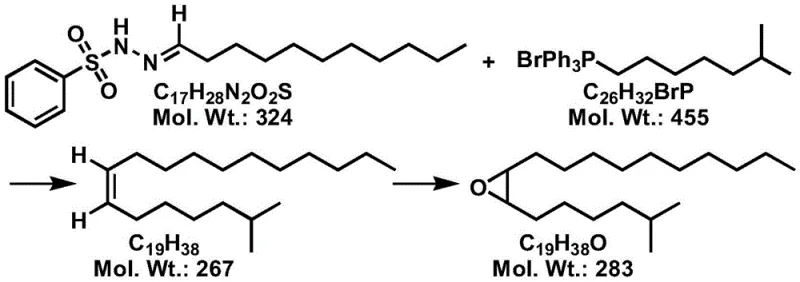
Impurity control is further enhanced by the specific purification techniques outlined in the patent, particularly the use of silver nitrate-loaded silica gel. Silver ions have a known affinity for pi-electrons in alkenes, forming reversible complexes that allow for the separation of cis and trans isomers based on their subtle differences in complexation stability. By employing this specialized stationary phase during column chromatography, manufacturers can effectively remove any trace amounts of trans-isomers that might have formed during the reaction or workup. This step is critical for achieving the reported gas phase purity of over 98.8% for the intermediate. Additionally, the subsequent epoxidation step using peroxy acids like m-chloroperoxybenzoic acid is highly selective for the electron-rich double bond. The reaction conditions are mild, typically ranging from -10°C to 30°C, which prevents ring-opening side reactions or rearrangement of the epoxy group. This rigorous control over both the alkene synthesis and the oxidation steps ensures that the final pheromone component meets the stringent specifications required for biological efficacy in the field.
How to Synthesize cis-7,8-epoxy-2-methyloctadecane Efficiently
The synthesis protocol described in the patent offers a clear roadmap for producing this valuable agrochemical intermediate with high fidelity. The process begins with the careful preparation of the reaction vessel under an inert argon atmosphere to exclude moisture and oxygen, which could deactivate the sensitive organolithium reagents. Following the addition of the phosphonium salt and solvent, the system is cooled to cryogenic temperatures before the base is introduced to generate the active ylide species in situ.
- React N-sulfonyl hydrazone with phosphonium salt under organic base at -80°C to -30°C to form cis-2-methyl-7-octadecene.
- Purify the intermediate using silver nitrate-loaded silica gel column chromatography to ensure high cis-isomer purity.
- Perform epoxidation on the purified alkene using peroxy acid at -10°C to 30°C to yield the final epoxy product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers compelling economic and logistical benefits that extend beyond simple yield metrics. The elimination of palladium-based catalysts removes a major cost driver associated with precious metal procurement and recovery, leading to significantly reduced raw material expenses. Furthermore, the avoidance of heavy metals simplifies the regulatory compliance landscape, reducing the administrative burden and testing costs associated with residual metal analysis. The robustness of the reaction conditions allows for more predictable production scheduling, which is essential for maintaining continuity in the supply of critical pest control agents. By minimizing the formation of difficult-to-separate impurities, the process reduces the volume of solvents and silica gel required for purification, contributing to a more sustainable and cost-efficient manufacturing footprint. These factors collectively enhance the overall value proposition for companies seeking a reliable agrochemical intermediate supplier.
- Cost Reduction in Manufacturing: The substitution of expensive transition metal catalysts with readily available organic bases and phosphonium salts drastically lowers the direct material costs per kilogram of product. Additionally, the simplified purification process reduces the consumption of chromatography media and solvents, which are often significant overhead items in fine chemical production. The high selectivity of the reaction minimizes the loss of valuable starting materials to byproduct formation, thereby improving the overall atom economy of the process. These efficiencies compound over large production volumes, resulting in substantial cost savings that can be passed down the supply chain or reinvested in R&D initiatives.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, such as N-sulfonyl hydrazones and phosphonium salts, are derived from common chemical feedstocks that are widely available in the global market. This reduces the risk of supply disruptions caused by geopolitical issues or single-source dependencies often associated with specialized catalysts. The robustness of the synthetic route also means that production can be scaled up or adjusted more flexibly in response to fluctuating market demand for pheromone traps. Consequently, partners can expect more consistent lead times and greater assurance of product availability during peak pest seasons.
- Scalability and Environmental Compliance: The process operates under relatively mild conditions and avoids the generation of hazardous heavy metal waste streams, aligning with increasingly strict environmental regulations. The use of standard organic solvents like tetrahydrofuran and dichloromethane allows for established recovery and recycling protocols to be implemented easily. This environmental compatibility facilitates smoother permitting processes for manufacturing facilities and reduces the long-term liability associated with waste disposal. The scalability of the reaction from gram to multi-kilogram scales has been demonstrated in the patent examples, confirming its viability for industrial batch production without significant re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. They are derived from the specific experimental data and beneficial effects detailed in the patent documentation to provide clarity for potential partners.
Q: How does this new method improve purity compared to traditional hydrogenation?
A: Traditional Lindlar hydrogenation of alkynes often generates trans-isomer impurities. This novel N-sulfonyl hydrazone route specifically avoids trans-2-methyl-7-octadecene formation, achieving purity levels higher than 98.8% for the intermediate and over 99% for the final epoxy product.
Q: What are the critical reaction conditions for the substitution step?
A: The substitution reaction requires strict temperature control between -80°C and -30°C, typically using organic bases like n-butyllithium or lithium diisopropylamide in solvents such as THF or methyl tert-butyl ether under an argon atmosphere.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method utilizes easily available raw materials and avoids expensive transition metal catalysts. The simple operation and convenient product separation make it highly suitable for industrial batch production and commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable cis-7,8-epoxy-2-methyloctadecane Supplier
The technical advancements detailed in CN115353437A highlight the immense potential for optimizing the production of gypsy moth pheromone intermediates through modern organic synthesis. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are successfully translated into industrial reality. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, including the detection of trace trans-isomers that could compromise product performance. We understand the critical nature of agrochemical supply chains and are committed to delivering high-purity pheromone intermediates that meet the exacting standards of the global market.
We invite you to collaborate with us to leverage this advanced technology for your specific application needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your current manufacturing setup. Please contact us to request specific COA data and route feasibility assessments that demonstrate how we can support your production goals. By partnering with us, you gain access to a supply chain that prioritizes quality, consistency, and technical excellence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
