Advanced Manufacturing of Pyrimidine Intermediates for Antiretroviral Drug Production
Advanced Manufacturing of Pyrimidine Intermediates for Antiretroviral Drug Production
The global pharmaceutical landscape is continuously evolving, driven by the urgent need for more efficient and cost-effective synthesis routes for critical antiretroviral therapies. At the heart of many next-generation Non-Nucleoside Reverse Transcriptase Inhibitors (NNRTIs) lies a specialized pyrimidine scaffold, specifically identified as 4-[(4,6-dihydroxy-2-pyrimidinyl)amino]benzonitrile. This compound, registered under CAS number 374067-80-8, serves as a pivotal building block in the construction of potent HIV-1 inhibitors. Recent intellectual property developments, specifically detailed in Chinese Patent CN101993420A, have unveiled a transformative manufacturing methodology that addresses long-standing bottlenecks in the production of this high-value pharmaceutical intermediate. This technical insight report analyzes the breakthrough process, highlighting its potential to redefine supply chain reliability and cost structures for multinational pharmaceutical corporations seeking a reliable pharmaceutical intermediate supplier.
![Chemical structure of 4-[(4,6-dihydroxy-2-pyrimidinyl)amino]benzonitrile (Compound I)](/insights/img/pyrimidine-benzonitrile-synthesis-pharma-supplier-20260313231814-01.png)
The significance of this chemical entity extends beyond its immediate application; it represents a class of heterocyclic compounds where precise substitution patterns are critical for biological activity. The molecular architecture features a central pyrimidine ring fused with a nitrile-substituted phenyl group, requiring meticulous control over regioselectivity during synthesis. Traditional methods often struggle to maintain the integrity of the nitrile group while effecting the cyclization of the pyrimidine ring, leading to complex impurity profiles that necessitate expensive purification steps. The innovation presented in the patent data offers a streamlined alternative that not only simplifies the synthetic trajectory but also enhances the environmental profile of the manufacturing process, aligning with the increasingly stringent regulatory standards governing API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the advent of the optimized route described in CN101993420A, the industry relied heavily on methodologies exemplified by international patent applications such as WO2008/080964. These legacy processes were fraught with inefficiencies that compounded costs and extended lead times. A primary drawback was the reliance on p-cyanoaniline hydrochloride as a starting material, a reagent that is notoriously difficult to source commercially in high purity and often requires an additional, cumbersome synthesis step from p-cyanoaniline using toxic thionyl chloride or similar chlorinating agents. Furthermore, the conventional cyclization step utilized ethylene glycol dimethyl ether (glyme) as the solvent, a substance associated with significant reproductive toxicity and high procurement costs, thereby complicating waste management and operator safety protocols.

Beyond the safety and sourcing issues, the chemical efficiency of the traditional route was suboptimal. The cyclization reaction, typically employing diethyl malonate and sodium ethoxide in ethanol, suffered from competing side reactions. Specifically, under the strongly alkaline conditions required for ring closure, the diethyl malonate was prone to undergoing Claisen condensation with itself, generating polymeric byproducts that contaminated the reaction mixture. This side reactivity resulted in a disappointing isolated yield of merely 57% for the final step, coupled with a crude product that was reddish in color and difficult to filter due to the formation of viscous tars. The necessity for multiple recrystallizations to achieve acceptable purity further eroded the overall mass balance, making the process economically unviable for large-scale commercial adoption.
The Novel Approach
In stark contrast to the convoluted legacy pathways, the novel approach delineated in the patent data introduces a robust, two-step sequence that begins with readily available commodity chemicals. The first stage involves the direct guanidinylation of p-cyanoaniline using cyanamide aqueous solution in the presence of hydrochloric acid. This reaction is conducted in benign alcoholic solvents such as n-butanol or even water, eliminating the need for hazardous chlorinated solvents or expensive glymes. The resulting p-guanidinobenzonitrile hydrochloride salt is obtained in high yield and purity, serving as a stable intermediate that can be easily isolated or telescoped into the subsequent step. This strategic shift in starting materials immediately removes the supply chain vulnerability associated with sourcing specialized hydrochloride salts.
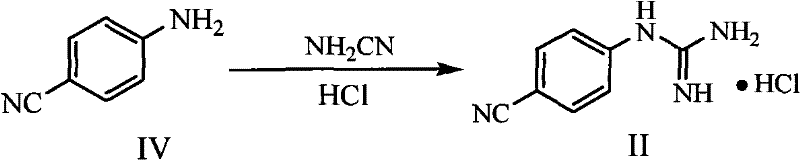
The second stage of the novel process refines the cyclization chemistry by substituting diethyl malonate with dimethyl malonate and utilizing sodium methoxide in methanol. This seemingly minor change in ester alkyl group and solvent system has profound effects on reaction kinetics and thermodynamics. The use of dimethyl malonate reduces steric hindrance during the nucleophilic attack, facilitating a smoother ring closure. Moreover, the solubility profile of the intermediates in methanol allows for a much cleaner reaction progression, minimizing the formation of the tarry byproducts observed in the ethanol-based systems. The result is a dramatic improvement in process performance, with yields consistently exceeding 80% and product purity surpassing 98% without the need for extensive chromatographic purification, marking a significant leap forward in cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Base-Catalyzed Pyrimidine Cyclization
To fully appreciate the technical superiority of this method, one must delve into the mechanistic nuances of the base-catalyzed condensation. The reaction proceeds through the deprotonation of the guanidine nitrogen by sodium methoxide, generating a highly nucleophilic amidine anion. This species then attacks the ester carbonyl of the dimethyl malonate. In conventional systems using sodium ethoxide and diethyl malonate, the equilibrium is less favorable, and the ethoxide ion is a stronger nucleophile that can compete with the guanidine for the ester, leading to transesterification and subsequent Claisen self-condensation of the malonate. By switching to the methoxide/methanol system, the nucleophilicity is better matched to the substrate, and the leaving group (methoxide) is identical to the base, preventing transesterification equilibria that complicate the reaction mixture.
Furthermore, the control of impurities is intrinsically linked to the workup procedure. The patent specifies a precise pH adjustment to the acidic range (pH 3-6) using dilute hydrochloric acid after the reaction is complete. This step is critical for protonating the pyrimidine nitrogen and precipitating the product in its neutral, least soluble form. In older methods, the precipitation was often incomplete or co-precipitated with sodium salts, leading to the difficult filtration described previously. The new protocol employs a slurry washing technique with alcohols like 95% ethanol or isopropanol, which effectively dissolves residual malonate esters and inorganic salts while leaving the high-melting pyrimidine product intact. This differential solubility is the key to achieving the reported 98%+ purity, ensuring that the impurity profile is well within the stringent limits required for GMP production of active pharmaceutical ingredients.
How to Synthesize 4-[(4,6-Dihydroxy-2-Pyrimidinyl)Amino]Benzonitrile Efficiently
The implementation of this synthesis route requires careful attention to stoichiometry and temperature control to maximize the benefits outlined above. The process is designed to be operationally simple, avoiding the need for cryogenic conditions or high-pressure equipment, which facilitates its adoption in multi-purpose chemical plants. The following guide summarizes the critical operational parameters derived from the patent examples, providing a roadmap for process chemists to replicate the high yields and purity in a pilot or production setting. For the detailed standardized synthesis steps, please refer to the guide below.
- React p-cyanoaniline with cyanamide aqueous solution in an alcoholic solvent using hydrochloric acid as a catalyst to form p-guanidinobenzonitrile hydrochloride.
- Condense the resulting guanidine salt with dimethyl malonate in methanol using sodium methoxide as the base to form the pyrimidine ring.
- Purify the final product by adjusting pH to acidic conditions and performing a slurry wash with alcohol to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel manufacturing process represents a strategic opportunity to optimize the cost of goods sold (COGS) and mitigate supply risks. The elimination of toxic and expensive solvents like glyme directly translates to lower raw material expenditures and reduced costs associated with hazardous waste disposal. Additionally, the shortening of the synthetic route by removing the prerequisite synthesis of p-cyanoaniline hydrochloride reduces the overall cycle time, allowing for faster response to market demand fluctuations. The robustness of the reaction conditions ensures consistent batch-to-batch quality, reducing the risk of production delays caused by failed batches or out-of-specification results.
- Cost Reduction in Manufacturing: The substitution of diethyl malonate with the more economical dimethyl malonate, combined with the use of methanol as a solvent, significantly lowers the direct material costs. Methanol is a bulk commodity chemical with a stable price history, unlike specialized ethers. Furthermore, the high yield of over 80% means that less raw material is wasted per kilogram of final product, effectively amplifying the purchasing power of the procurement budget. The simplified workup also reduces energy consumption by minimizing the duration of distillation and drying steps, contributing to substantial cost savings in utility overheads.
- Enhanced Supply Chain Reliability: By relying on p-cyanoaniline, a widely produced industrial chemical, rather than its hydrochloride salt, the supply chain becomes more resilient. There are multiple global suppliers for the free amine, reducing the risk of single-source dependency. The stability of the intermediate guanidine salt also allows for potential stockpiling or decoupling of the two reaction steps, providing greater flexibility in production scheduling. This flexibility is crucial for maintaining continuity of supply in the face of logistical disruptions or sudden spikes in demand for the downstream antiretroviral drugs.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated in multi-hundred gram scales in the patent examples with consistent results. The absence of heavy metal catalysts and the use of aqueous workups simplify the environmental compliance burden. Wastewater treatment is more straightforward as the effluent primarily contains biodegradable alcohols and inorganic salts, avoiding the complex remediation required for chlorinated or glycol-based waste streams. This environmental friendliness future-proofs the manufacturing site against tightening environmental regulations, ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity for technical teams evaluating the feasibility of this route for their specific supply chain needs.
Q: How does this new method improve yield compared to prior art?
A: The novel process eliminates the need for difficult-to-source p-cyanoaniline hydrochloride and avoids toxic glyme solvents, increasing the overall yield from approximately 57% in legacy methods to over 80%.
Q: What are the primary impurities controlled in this synthesis?
A: By utilizing dimethyl malonate instead of diethyl malonate and optimizing the base addition, the process significantly suppresses Claisen condensation side reactions, resulting in a product purity exceeding 98%.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the use of common solvents like methanol and n-butanol, along with a simplified workup procedure involving slurry washing rather than complex recrystallization, makes it highly scalable and cost-effective.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-[(4,6-Dihydroxy-2-Pyrimidinyl)Amino]Benzonitrile Supplier
At NINGBO INNO PHARMCHEM, we understand that the transition from laboratory patent to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields reported in CN101993420A can be reliably replicated on an industrial scale. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of this critical HIV intermediate meets the exacting standards required by global regulatory bodies.
We invite procurement leaders and R&D directors to engage with our technical procurement team to discuss how this optimized synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic benefits tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest level of confidence and technical support.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
