Advanced Synthesis of Optically Active Octanoic Acid Derivatives for Commercial Pharmaceutical Applications
Advanced Synthesis of Optically Active Octanoic Acid Derivatives for Commercial Pharmaceutical Applications
The pharmaceutical industry continuously demands more efficient and stereoselective pathways for producing complex chiral intermediates, a challenge directly addressed by the technological breakthroughs detailed in patent CN1144796C. This seminal intellectual property outlines a sophisticated methodology for the preparation of optically active octanoic acid derivatives, specifically focusing on the high-value target molecule 2R-propyl octanoic acid, which serves as a critical building block for neurodegenerative disease therapeutics. The core innovation lies in the strategic application of (1S)-(-)-2,10-camphorsultam as a chiral auxiliary, enabling a drastic reduction in synthetic steps while simultaneously elevating optical purity to levels previously unattainable through standard resolution techniques. By shifting away from inefficient racemic resolutions that suffer from inherent 50% yield losses, this process offers a linear, high-yielding trajectory that is exceptionally well-suited for industrial scale-up. For R&D directors and procurement specialists alike, understanding the mechanistic advantages of this route is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering consistent quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically active 2R-propyl octanoic acid has been plagued by significant inefficiencies that render traditional methods economically and technically unviable for large-scale commercial operations. Prior art, such as the methods disclosed in Japanese Patent publications, typically relies on optical resolution of racemates using chiral amines or the utilization of chiral pool starting materials like prolinol, both of which present severe bottlenecks. For instance, resolution strategies starting from methyl hexylmalonate require a cumbersome six-step sequence that culminates in a dismal total synthesis yield of merely 5.9%, alongside a suboptimal optical purity of 90.0% e.e. Similarly, approaches utilizing optically active prolinol necessitate five reaction steps and achieve a total yield of only 20.1%, which is insufficient for cost-effective manufacturing. These legacy processes not only consume excessive raw materials and solvent volumes but also generate substantial waste, creating a heavy burden on supply chain logistics and environmental compliance teams who must manage the disposal of low-value byproducts.
The Novel Approach
In stark contrast to these outdated methodologies, the novel approach defined in CN1144796C introduces a streamlined four-step synthetic route that fundamentally transforms the production economics of this valuable intermediate. By leveraging the stereocontrolling power of camphorsultam derivatives, the process achieves a remarkable total synthesis yield ranging from 42.5% to 72.1% starting from the inexpensive and readily available octanoyl chloride. This represents a multiplicative increase in efficiency compared to the single-digit yields of the past, directly translating to significant cost reduction in pharmaceutical intermediate manufacturing. Furthermore, the intermediates generated in this pathway, such as N-(2S-(2-propenyl)octanoyl)-(1S)-(-)-2,10-camphorsultam, exhibit excellent crystallinity, allowing for purification through simple recrystallization rather than complex chromatography. This capability ensures that the final product consistently meets stringent purity specifications, with optical purity values reaching between 95% and 99% e.e., thereby eliminating the need for costly downstream polishing steps.
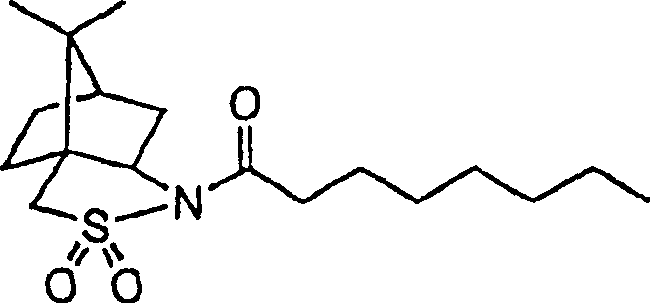
Mechanistic Insights into Camphorsultam-Mediated Asymmetric Alkylation
The success of this synthetic strategy is rooted in the precise mechanistic behavior of the (1S)-(-)-2,10-camphorsultam auxiliary, which acts as a robust template for inducing chirality during the carbon-carbon bond-forming events. The process begins with the acylation of the sultam nitrogen with octanoyl chloride, creating an activated substrate that is primed for diastereoselective alkylation. When treated with strong bases such as lithium diisopropylamide at cryogenic temperatures, the alpha-proton is removed to form a rigid enolate species, the geometry of which is strictly controlled by the bulky camphor framework. Subsequent addition of allyl or propargyl halides occurs with high facial selectivity, ensuring that the new stereocenter is established with the desired configuration. This level of control is critical for preventing the formation of unwanted diastereomers, which would otherwise complicate purification and reduce overall yield. The ability to tune the reaction conditions, including temperature and solvent choice, allows for further optimization of the diastereomeric excess, often exceeding 99% after a single recrystallization step.
Another pivotal mechanistic advancement in this patent is the development of a novel hydrolysis protocol using tetraalkylammonium hydroxide, a reagent system that had not been previously disclosed for cleaving camphorsultam derivatives without racemization. Traditional hydrolysis methods using alkali metal hydroxides can sometimes lead to epimerization at the alpha-position under harsh conditions, compromising the hard-won optical purity. However, the use of tetraalkylammonium hydroxide in mixed solvent systems containing peroxides allows for mild cleavage of the amide bond at temperatures as low as -20°C to 40°C. This gentle yet effective method preserves the stereochemical integrity of the molecule throughout the final liberation of the free acid. Additionally, for derivatives containing unsaturated bonds, the protocol includes safeguards such as the addition of excess olefins to prevent oxidative side reactions, demonstrating a deep understanding of potential degradation pathways. This attention to mechanistic detail ensures that the commercial scale-up of complex pharmaceutical intermediates remains robust and reproducible.
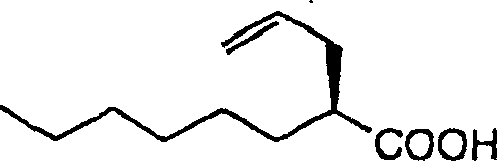
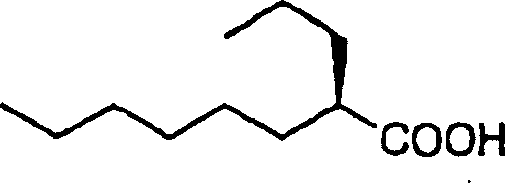
How to Synthesize 2R-Propyl Octanoic Acid Efficiently
Implementing this synthesis requires careful adherence to the specific reaction parameters outlined in the patent to maximize yield and optical purity. The process is designed to be modular, allowing manufacturers to produce either the unsaturated precursors or the fully saturated 2R-propyl octanoic acid depending on their specific downstream requirements. Detailed standard operating procedures for each transformation, including precise stoichiometry, temperature profiles, and workup protocols, are essential for maintaining consistency across batches. The following guide summarizes the critical operational phases required to execute this technology effectively in a pilot or production environment.
- React (1S)-(-)-2,10-camphorsultam with octanoyl chloride to form the N-acyl intermediate.
- Perform diastereoselective alkylation using allyl or propargyl halides to introduce the side chain.
- Execute hydrolysis using tetraalkylammonium hydroxide or catalytic reduction to obtain the final optically active acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers profound strategic advantages that extend far beyond simple chemical yield improvements. The primary benefit lies in the drastic simplification of the manufacturing workflow, which inherently reduces the exposure to supply chain disruptions associated with multi-step syntheses. By condensing the process from six steps down to four, the number of unit operations, solvent exchanges, and intermediate isolations is significantly decreased, leading to a shorter overall production cycle time. This acceleration enhances supply chain reliability by allowing for faster turnaround on orders and reducing the inventory holding costs associated with work-in-progress materials. Furthermore, the reliance on commodity chemicals like octanoyl chloride and allyl bromide ensures that raw material sourcing remains stable and cost-effective, mitigating the risks associated with specialty reagent shortages.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substantial increase in overall mass efficiency, which directly lowers the cost of goods sold per kilogram of final product. By achieving total yields of over 70% compared to the single-digit yields of legacy methods, the amount of starting material required to produce a fixed quantity of API intermediate is drastically reduced. Additionally, the elimination of expensive chiral resolving agents and the ability to recover and recycle the camphorsultam auxiliary further contribute to cost optimization. The high crystallinity of the intermediates also means that purification can be achieved through crystallization rather than expensive preparative chromatography, resulting in substantial cost savings in both consumables and labor. These factors combine to create a highly competitive cost structure that supports long-term pricing stability for customers.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route translates directly into improved supply security for downstream pharmaceutical manufacturers. Because the reaction conditions are well-defined and tolerant of minor variations, the risk of batch failure is minimized, ensuring a consistent flow of high-quality material. The use of standard laboratory equipment and common solvents means that the process can be easily transferred between different manufacturing sites without the need for specialized hardware, enhancing flexibility in production planning. Moreover, the high optical purity achieved without the need for complex chiral separations reduces the dependency on external tolling partners for resolution services, keeping the supply chain vertically integrated and under direct control. This autonomy is crucial for maintaining continuity of supply in the face of global market fluctuations.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this process aligns perfectly with modern green chemistry principles by minimizing waste generation and solvent usage. The higher atom economy and reduced step count mean that less chemical waste is produced per unit of product, simplifying waste treatment and lowering disposal costs. The ability to perform reactions at moderate temperatures and pressures also reduces energy consumption, contributing to a lower carbon footprint for the manufacturing facility. Furthermore, the avoidance of toxic heavy metal catalysts in the key stereoselective steps simplifies the regulatory filing process for the final drug substance, as residual metal limits are easier to meet. This environmental compatibility facilitates smoother regulatory approvals and strengthens the sustainability profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What optical purity can be achieved with this synthesis method?
A: The process described in patent CN1144796C consistently achieves high optical purity ranging from 95% to 99% e.e., significantly surpassing prior art methods which often struggle to exceed 90% e.e.
Q: How does this process improve upon conventional synthesis routes?
A: Unlike conventional methods requiring six steps with a total yield of only 5.9%, this novel approach reduces the synthesis to just four steps while increasing the total yield to between 42.5% and 72.1%.
Q: What are the key intermediates involved in this pathway?
A: The synthesis relies on novel camphorsultam derivatives, specifically N-(2S-(2-propenyl)octanoyl)-(1S)-(-)-2,10-camphorsultam, which possess excellent crystallinity allowing for purification via recrystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2R-Propyl Octanoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthetic technologies to maintain a competitive edge in the global pharmaceutical market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent literature can be successfully translated into industrial reality. We are equipped with rigorous QC labs and state-of-the-art analytical instrumentation to guarantee stringent purity specifications for every batch of optically active octanoic acid derivatives we produce. Our commitment to quality assurance means that we can deliver materials that meet the exacting standards required for clinical and commercial drug manufacturing, providing our partners with peace of mind regarding product consistency and regulatory compliance.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By partnering with us, you gain access to a Customized Cost-Saving Analysis that evaluates the potential economic benefits of switching to this high-efficiency process for your supply chain. We encourage you to request specific COA data and route feasibility assessments to verify the technical capabilities of our facility. Whether you require small quantities for R&D purposes or metric tons for commercial launch, NINGBO INNO PHARMCHEM stands ready to serve as your trusted partner in delivering high-purity pharmaceutical intermediates with unmatched reliability and speed.
