Revolutionizing Spirotetronic Acid Derivative Production with Novel One-Pot Cyclization Technology
Revolutionizing Spirotetronic Acid Derivative Production with Novel One-Pot Cyclization Technology
The landscape of fine chemical manufacturing is constantly evolving, driven by the need for more efficient, sustainable, and cost-effective synthetic routes. A pivotal advancement in this domain is detailed in patent CN1247562C, which discloses a novel method for preparing spirotetronic acid derivatives. These compounds are critical scaffolds in the development of high-performance agrochemicals and pharmaceutical intermediates, known for their biological activity and structural complexity. The patented process introduces a streamlined one-pot synthesis strategy that fundamentally alters the production economics by eliminating intermediate isolation steps. For R&D directors and procurement specialists seeking a reliable agrochemical intermediate supplier, understanding the mechanistic nuances and commercial implications of this technology is essential for securing a competitive supply chain. This report provides a deep technical analysis of the process, highlighting its potential to redefine industry standards for purity and operational efficiency.
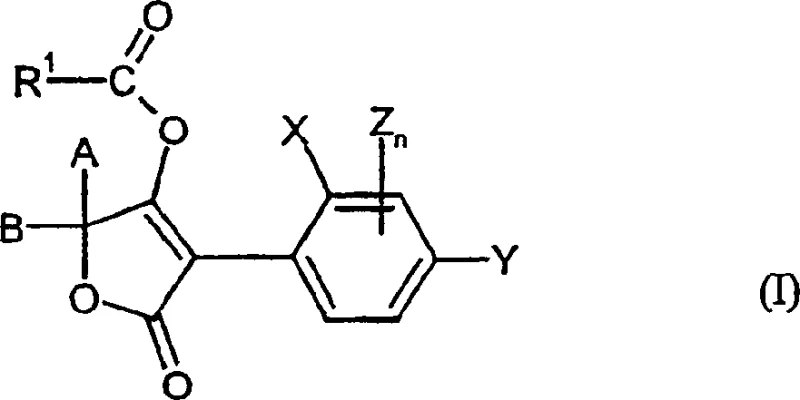
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for constructing the spirotetronic acid core often involve multi-step sequences that are inherently inefficient and resource-intensive. Conventional methods typically require the discrete isolation and purification of reactive intermediates, such as enol salts or cyclic precursors, before proceeding to the final acylation or functionalization steps. This fragmentation of the synthesis leads to significant material losses at each transfer stage, cumulatively depressing the overall yield. Furthermore, the handling of unstable intermediates necessitates stringent control over atmospheric conditions and temperature, increasing the operational complexity and safety risks associated with the manufacturing process. From a supply chain perspective, these additional unit operations translate to longer lead times and higher capital expenditure for equipment dedicated to filtration, drying, and storage. The accumulation of solvent waste from multiple workup procedures also poses substantial environmental compliance challenges, making conventional routes less attractive in an era of increasing regulatory scrutiny on chemical manufacturing emissions and waste disposal protocols.
The Novel Approach
In stark contrast, the methodology described in patent CN1247562C utilizes a telescoped one-pot procedure that seamlessly integrates the cyclization and acylation steps into a single reactor vessel. By reacting the diester precursor (Formula II) directly with a base to generate the enol salt in situ, followed immediately by the addition of the acylating agent (Formula III), the process bypasses the need for intermediate isolation entirely. This continuous flow of chemistry not only preserves the integrity of the reactive species but also maximizes atom economy by minimizing side reactions that often occur during workup and storage. The ability to conduct the reaction in a single pot significantly reduces the footprint of the manufacturing operation, allowing for higher throughput within existing infrastructure. For procurement managers, this translates to a more robust supply capability, as the simplified process is less prone to bottlenecks and equipment failures. The novel approach effectively decouples production capacity from the limitations of batch processing inherent in multi-step syntheses, offering a scalable solution that aligns with modern principles of green chemistry and sustainable industrial practice.
Mechanistic Insights into Base-Catalyzed Cyclization and Acylation
The core of this innovative synthesis lies in the precise control of the base-catalyzed cyclization mechanism, which converts the linear diester precursor into the rigid spirotetronic framework. The process initiates with the deprotonation of the active methylene group in the diester by a strong base, such as sodium hydroxide, in a polar aprotic solvent like N,N-dimethylacetamide. This deprotonation generates a nucleophilic enolate species that undergoes intramolecular attack on the adjacent ester carbonyl, closing the ring to form the tetronic acid core. Crucially, the reaction conditions, including temperature ranges from 0°C to 150°C and reduced pressure (50 to 500 mbar), are optimized to drive the equilibrium towards the formation of the stable enol sodium salt while continuously removing volatile by-products like alcohols. This dynamic removal of distillate prevents the reverse reaction, ensuring high conversion rates without the need for excessive reagent excess. The stability of the resulting enol salt in the reaction medium is key, as it allows the subsequent acylation to proceed without the degradation often seen with isolated intermediates.
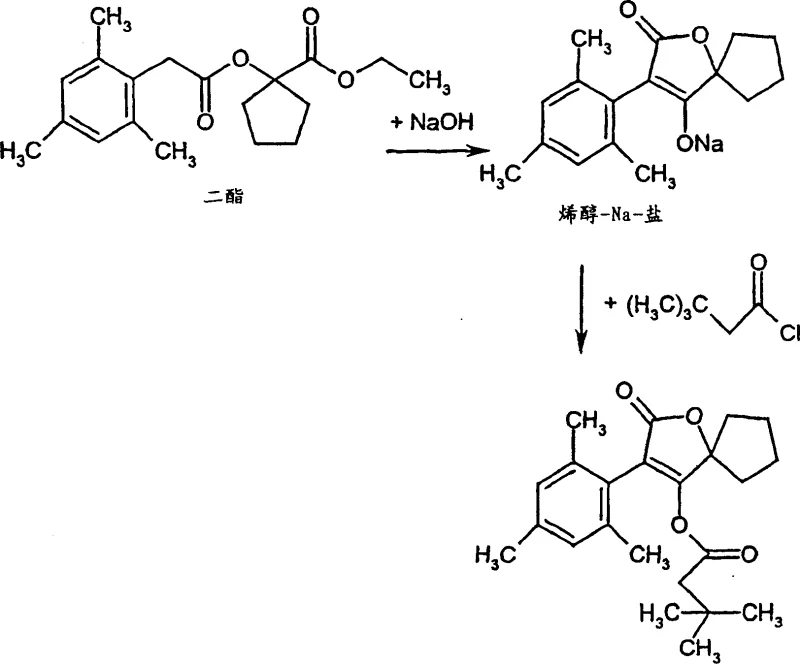
Following the cyclization, the introduction of the acid halide triggers the final acylation step, where the oxygen atom of the enol salt acts as a nucleophile to attack the carbonyl carbon of the acyl halide. This step is facilitated by the presence of an acid acceptor, such as triethylamine or inorganic carbonates, which scavenges the hydrogen halide by-product generated during the reaction. The choice of acid acceptor is critical for maintaining the pH balance of the reaction mixture and preventing acid-catalyzed decomposition of the sensitive spirotetronic ring system. The mechanistic elegance of this sequence ensures that the final product precipitates or can be easily extracted with high purity, as the side products are largely soluble in the aqueous wash phases. For quality control teams, this mechanism offers a distinct advantage in impurity management, as the absence of isolation steps minimizes the introduction of external contaminants and reduces the formation of oligomeric by-products. The result is a highly selective transformation that delivers the target molecule with a purity profile that meets the rigorous specifications required for downstream formulation in agrochemical or pharmaceutical applications.
How to Synthesize Spirotetronic Acid Derivatives Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize the benefits of the one-pot design. The process begins with the preparation of the base solution, followed by the controlled addition of the diester substrate under vacuum to facilitate the removal of volatiles. Maintaining the correct stoichiometry between the base, the diester, and the subsequent acylating agent is vital for achieving optimal yields. The reaction temperature must be ramped appropriately to ensure complete cyclization before the addition of the acid halide, preventing competing hydrolysis reactions. Detailed standard operating procedures regarding the rate of addition, stirring efficiency, and cooling profiles are essential for reproducibility on a commercial scale. The following guide outlines the standardized synthesis steps derived from the patent examples to ensure consistent high-quality output.
- React the diester precursor (Formula II) with a strong base such as sodium hydroxide in a polar aprotic solvent like N,N-dimethylacetamide under reduced pressure to form the enol sodium salt.
- Maintain the reaction mixture at elevated temperatures (0°C to 150°C) while removing distillate to drive the cyclization equilibrium forward without isolating the intermediate salt.
- Directly add an acid halide (Formula III) and an acid acceptor to the reaction solution to acylate the enol salt, yielding the final spirotetronic acid derivative after workup.
Commercial Advantages for Procurement and Supply Chain Teams
The adoption of this one-pot synthesis technology offers profound commercial advantages that extend far beyond the laboratory bench, directly impacting the bottom line for chemical manufacturers and their clients. By consolidating multiple reaction steps into a single operational unit, the process drastically simplifies the manufacturing workflow, reducing the requirement for intermediate storage tanks and transfer lines. This simplification leads to a significant reduction in capital expenditure for new facilities and allows for the debottlenecking of existing production lines, thereby enhancing overall plant utilization rates. For supply chain heads, the reduced complexity translates to greater reliability and continuity of supply, as there are fewer points of failure in the production sequence. The elimination of intermediate isolation steps also means that the total processing time per batch is substantially shortened, enabling faster turnaround times for customer orders and improved responsiveness to market demand fluctuations without compromising on product quality or specification compliance.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the reduction in solvent usage and waste disposal costs associated with multiple workup stages. Since the intermediate enol salt is not isolated, the volumes of solvents required for washing, crystallization, and drying are minimized, leading to substantial savings in raw material procurement. Furthermore, the higher overall yield achieved through the telescoped process means that less starting material is required to produce the same amount of final product, effectively lowering the cost of goods sold. The energy consumption is also optimized, as heating and cooling cycles are consolidated, reducing the utility load on the manufacturing facility. These cumulative efficiencies allow for a more competitive pricing structure, providing buyers with access to high-purity intermediates at a reduced total cost of ownership while maintaining healthy margins for the producer.
- Enhanced Supply Chain Reliability: From a logistics and planning perspective, the streamlined nature of this synthesis enhances the predictability of production schedules. The reduced number of unit operations decreases the likelihood of delays caused by equipment maintenance or intermediate quality failures, ensuring a steady flow of finished goods. The robustness of the reaction conditions, which tolerate a range of bases and solvents, provides flexibility in sourcing raw materials, mitigating the risk of supply disruptions due to vendor-specific issues. This resilience is crucial for long-term supply agreements, where consistency and on-time delivery are paramount. Additionally, the simplified process facilitates easier scale-up from pilot to commercial production, allowing suppliers to rapidly increase capacity in response to surging demand without the need for extensive process re-validation or new infrastructure investment.
- Scalability and Environmental Compliance: The environmental footprint of chemical manufacturing is a critical factor for modern enterprises, and this one-pot method aligns perfectly with sustainability goals. By minimizing waste generation and solvent emissions, the process helps manufacturers meet increasingly stringent environmental regulations and corporate social responsibility targets. The reduced volume of hazardous waste simplifies disposal logistics and lowers associated compliance costs. Moreover, the scalability of the reaction is excellent, as the heat and mass transfer requirements are manageable even in large-scale reactors, ensuring that the quality observed in the lab is maintained in tonnage production. This alignment of economic efficiency with environmental stewardship makes the technology highly attractive for companies aiming to build a sustainable and future-proof supply chain for complex organic intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of spirotetronic acid derivatives using this advanced methodology. These insights are derived directly from the patent specifications and practical implementation data, providing clarity on the process capabilities and limitations. Understanding these details helps stakeholders make informed decisions regarding sourcing and integration into their own value chains. The answers reflect the consensus on best practices for handling these versatile chemical building blocks.
Q: What are the primary advantages of the one-pot synthesis method for spirotetronic acid derivatives?
A: The one-pot method eliminates the need for isolating intermediate enol salts, significantly reducing solvent consumption, processing time, and waste generation while improving overall yield and purity.
Q: Which bases are suitable for the cyclization step in this process?
A: The process supports a wide range of bases including alkali metal hydroxides like sodium hydroxide, carbonates, and alkoxides, allowing flexibility based on cost and availability.
Q: How does this method impact the impurity profile of the final product?
A: By avoiding the isolation of unstable intermediates and minimizing handling steps, the method reduces the formation of degradation by-products, resulting in a cleaner impurity profile suitable for high-specification applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spirotetronic Acid Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthesis routes like the one described in patent CN1247562C for maintaining a competitive edge in the global chemical market. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical intermediates. Our state-of-the-art facilities are equipped to handle the specific requirements of base-catalyzed cyclizations and one-pot syntheses, adhering to stringent purity specifications and rigorous QC labs protocols. We are committed to delivering high-purity spirotetronic acid derivatives that meet the exacting standards of the agrochemical and pharmaceutical industries, supporting your R&D and commercialization efforts with unwavering quality and technical expertise.
We invite you to collaborate with us to leverage these technological advancements for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how our optimized processes can reduce your overall procurement costs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments for your target molecules. Let us be your partner in navigating the complexities of fine chemical synthesis, ensuring your supply chain remains robust, compliant, and cost-effective in an ever-evolving market landscape.
