Advanced Synthesis of Fluorinated Agrochemical Intermediates for Commercial Scale Production
The global demand for high-performance agrochemicals continues to drive innovation in intermediate synthesis, particularly for fluorinated compounds that offer enhanced biological activity and environmental stability. Patent CN1210241C introduces a significant technological breakthrough in the preparation of 1,4-diaryl-2-fluoro-1,3-butadiene compounds, which serve as critical precursors for next-generation insecticides and acaricides. This intellectual property outlines a novel synthetic pathway that fundamentally alters the production landscape by eliminating the reliance on costly and problematic phosphonium halide reagents. For R&D directors and procurement strategists, understanding the implications of this patent is essential for optimizing supply chains and reducing manufacturing overheads. The technology described herein provides a robust framework for producing high-purity agrochemical intermediates with improved process efficiency. By leveraging this specific chemical architecture, manufacturers can achieve superior impurity profiles while maintaining strict control over reaction parameters. This report analyzes the technical merits and commercial viability of the methods disclosed in CN1210241C, offering a strategic perspective for stakeholders in the fine chemical industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,4-diaryl-2-fluoro-1,3-butadiene compounds has been hindered by the methodologies described in prior art such as EP 811593-A1, which necessitates the use of phosphonium halide compounds. These traditional reagents present substantial challenges in an industrial setting, primarily due to their high procurement costs and the complex waste streams they generate. The reaction by-products associated with phosphonium salts are notoriously difficult to separate from the desired final product, often requiring extensive purification steps that lower overall yield and increase processing time. Furthermore, the handling of phosphonium halides demands stringent safety protocols and specialized equipment, adding to the operational expenditure of any manufacturing facility. From a supply chain perspective, the dependency on these specific reagents introduces vulnerability, as their availability can be subject to market fluctuations and regulatory constraints. The accumulation of phosphorus-containing waste also poses environmental compliance issues, requiring additional treatment facilities to meet discharge standards. Consequently, the conventional approach creates a bottleneck that limits scalability and inflates the cost of goods sold for the final agrochemical active ingredients.
The Novel Approach
In stark contrast to the legacy methods, the technology protected under CN1210241C proposes a streamlined synthetic route that completely circumvents the use of phosphonium halide compounds. This novel approach utilizes 1,4-diaryl-2-fluoro-1-buten-3-ol compounds as key intermediates, which are subsequently converted into the target butadiene structures through a reaction with sulfonyl chlorides or sulfonic anhydrides in the presence of a base. This chemical transformation is not only more direct but also generates by-products that are significantly easier to manage and remove during the workup phase. The shift to sulfonyl-based reagents allows for the use of common organic solvents such as tetrahydrofuran and ethyl acetate, which are readily available and cost-effective on a global scale. By simplifying the reaction mechanism, the new process enhances the overall atom economy and reduces the environmental footprint of the manufacturing operation. This methodological improvement translates directly into a more resilient supply chain, as the raw materials required are commodity chemicals with stable pricing and availability. The ability to produce high-purity intermediates without the burden of phosphorus waste removal represents a paradigm shift in the manufacturing of fluorinated agrochemical building blocks.
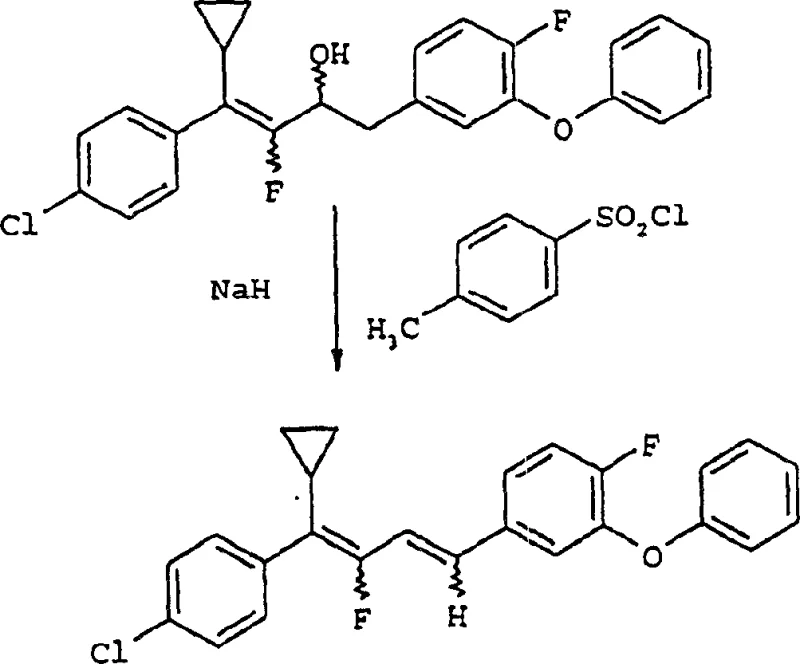
Mechanistic Insights into Sulfonyl-Mediated Elimination
The core of this technological advancement lies in the precise manipulation of the 1,4-diaryl-2-fluoro-1-buten-3-ol intermediate, structurally defined as Formula I in the patent documentation. This molecule features a specific arrangement of aryl groups and a fluorine atom that is critical for the subsequent elimination reaction. The stability and reactivity of Formula I are governed by the electronic properties of the substituents on the aromatic rings, which can be tuned to optimize the reaction kinetics. Understanding the stereochemistry and conformational flexibility of this intermediate is vital for R&D teams aiming to replicate the process at scale. The presence of the hydroxyl group at the 3-position allows for activation by sulfonyl chlorides, creating a good leaving group that facilitates the formation of the conjugated diene system. This mechanistic pathway ensures that the fluorine atom is retained in the final structure, preserving the biological potency required for insecticidal applications. The careful selection of the base, such as alkali metal hydrides, further drives the equilibrium towards the desired product while minimizing side reactions that could lead to impurity formation.
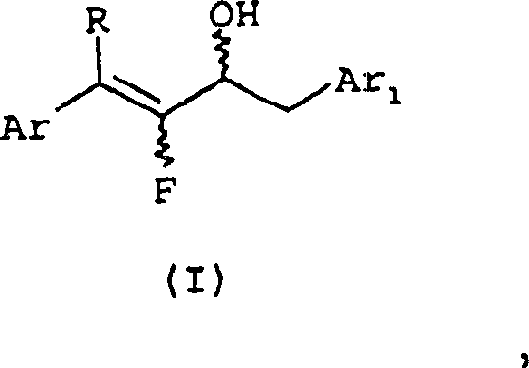
Impurity control is a paramount concern in the synthesis of agrochemical intermediates, and the mechanism described in CN1210241C offers inherent advantages in this regard. By avoiding the formation of phosphonium salts, the process eliminates a major source of persistent contaminants that are difficult to purge from the final API or intermediate. The reaction conditions, typically ranging from 20°C to 80°C, are mild enough to prevent thermal degradation of the sensitive fluorinated backbone yet sufficient to drive the elimination to completion. The use of standard extraction solvents allows for efficient partitioning of the product from inorganic salts and organic by-products, resulting in a crude material that requires minimal downstream processing. This high level of purity is essential for meeting the stringent specifications demanded by regulatory bodies and end-users in the crop protection sector. Furthermore, the robustness of the reaction against variations in temperature and reagent stoichiometry provides a wide operating window, making the process forgiving and reliable for commercial production teams. The mechanistic clarity provided by this patent empowers chemists to troubleshoot potential issues proactively and maintain consistent quality across different production batches.
How to Synthesize 1,4-diaryl-2-fluoro-1,3-butadiene Efficiently
Implementing this synthesis route requires a clear understanding of the operational parameters to ensure safety and efficiency. The process begins with the preparation of the Formula I intermediate, followed by its activation and elimination to form the target diene. Detailed standard operating procedures are critical for maintaining reproducibility, especially when scaling from laboratory to plant scale. The following guide outlines the fundamental steps derived from the patent examples to assist technical teams in process validation.
- React 1,4-diaryl-2-fluoro-1-buten-3-ol with sulfonyl chloride or sulfonic anhydride in the presence of a base.
- Maintain reaction temperature between 20°C to 80°C to ensure optimal conversion rates.
- Isolate the 1,4-diaryl-2-fluoro-1,3-butadiene product via aqueous workup and solvent extraction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology disclosed in CN1210241C offers tangible strategic benefits that extend beyond mere chemical novelty. The primary advantage lies in the significant reduction of raw material costs associated with eliminating expensive phosphonium halides from the bill of materials. This substitution with more common sulfonyl chlorides and anhydrides directly lowers the variable cost per kilogram of the produced intermediate. Additionally, the simplified purification process reduces the consumption of solvents and energy, contributing to overall operational efficiency. The reliability of the supply chain is enhanced because the new reagents are widely produced commodities, reducing the risk of shortages that can halt production lines. This stability allows for better long-term planning and contract negotiation with downstream customers who require guaranteed delivery schedules. Moreover, the environmental benefits of the new process align with increasingly strict global regulations on chemical manufacturing, reducing the liability and compliance costs associated with waste disposal.
- Cost Reduction in Manufacturing: The elimination of phosphonium halide compounds from the synthesis route removes a major cost driver that has historically inflated the price of fluorinated intermediates. Phosphonium salts are not only expensive to purchase but also require specialized handling and disposal protocols that add hidden costs to the manufacturing budget. By switching to sulfonyl chlorides and bases like sodium hydride, the process leverages commodity chemicals that benefit from economies of scale and competitive pricing. The reduction in purification steps further decreases the consumption of utilities such as steam and electricity, leading to substantial cost savings in the overall production cycle. These efficiencies allow manufacturers to offer more competitive pricing to their clients while maintaining healthy profit margins. The qualitative improvement in cost structure makes this technology a compelling choice for companies looking to optimize their manufacturing expenditures without compromising on product quality.
- Enhanced Supply Chain Reliability: Dependence on niche reagents like phosphonium halides introduces significant risk into the supply chain, as their production is often limited to a few specialized suppliers. The new method utilizes widely available reagents such as p-toluenesulfonyl chloride and sodium hydride, which are stocked by numerous chemical distributors globally. This diversification of the supply base ensures that production can continue uninterrupted even if one supplier faces logistical challenges. The robustness of the reaction also means that lead times can be shortened, as there is less need for extended purification or reprocessing of off-spec material. For supply chain heads, this translates to improved on-time delivery performance and the ability to respond quickly to fluctuations in market demand. The increased reliability fosters stronger relationships with downstream partners who depend on consistent availability of critical agrochemical intermediates for their own formulation processes.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden complexities, but the methodology in CN1210241C is designed with industrial practicality in mind. The reaction conditions are mild and do not require extreme pressures or temperatures, making them suitable for standard glass-lined or stainless steel reactors found in most multipurpose plants. The waste profile of the process is significantly cleaner, as it avoids the generation of phosphorus-containing waste streams that are difficult to treat in standard effluent plants. This environmental compatibility simplifies the permitting process for new production lines and reduces the ongoing costs of waste management. The ability to scale from kilogram to multi-ton quantities without fundamental changes to the chemistry provides a clear path for commercial expansion. Companies adopting this technology can confidently invest in capacity increases, knowing that the process is robust enough to handle large-scale operations while meeting rigorous environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the data provided in CN1210241C to ensure accuracy and relevance for decision-makers. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing production portfolios.
Q: Why is the new method superior to EP 811593-A1?
A: The conventional method described in EP 811593-A1 relies on expensive phosphonium halide compounds which generate difficult-to-remove by-products. The novel approach in CN1210241C avoids these reagents entirely, simplifying purification and reducing raw material costs.
Q: What are the key reaction conditions for this synthesis?
A: The process utilizes sulfonyl chlorides or sulfonic anhydrides with a base such as sodium hydride. The reaction proceeds efficiently in ether solvents like tetrahydrofuran at temperatures ranging from 20°C to 80°C.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the avoidance of phosphonium salts and the use of standard extraction solvents like ethyl acetate and toluene make the workflow highly scalable and compatible with existing industrial infrastructure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4-diaryl-2-fluoro-1-buten-3-ol Supplier
As the chemical industry evolves, having a partner with the technical expertise to navigate complex synthetic pathways is invaluable. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, offering deep expertise in the scale-up of fluorinated intermediates and agrochemical building blocks. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab to plant is seamless. We understand the critical importance of stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the highest industry standards. Our commitment to quality and reliability makes us the ideal partner for companies seeking to leverage the advancements described in CN1210241C.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through innovative chemistry. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs. We encourage you to contact us to request specific COA data and route feasibility assessments for your target molecules. By partnering with NINGBO INNO PHARMCHEM, you gain access to a wealth of knowledge and capacity that can drive your business forward in a competitive market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
