Advanced Isothermal Crystallization Technology for High-Purity Esomeprazole Sodium Manufacturing
The pharmaceutical industry continuously demands higher purity standards for Proton Pump Inhibitors (PPIs), particularly for the S-isomer of omeprazole, known as Esomeprazole Sodium. Recent advancements documented in patent CN112661744B introduce a groundbreaking purification methodology that addresses the critical stability and purity challenges inherent in previous manufacturing routes. This technology leverages a sophisticated mixed-solvent crystallization strategy that eliminates the need for thermal cycling, thereby preventing the degradation often associated with heat-sensitive benzimidazole derivatives. By operating strictly within an isothermal range of 0-30°C, the process ensures that the delicate sulfinyl bridge remains intact, significantly reducing the formation of sulfone impurities and R-enantiomers. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this innovation represents a pivotal shift towards more robust and scalable production capabilities. The structural integrity of the final molecule is paramount for clinical efficacy, and this method guarantees a purity profile exceeding 99.5% with R-enantiomer content suppressed to less than 0.01%.
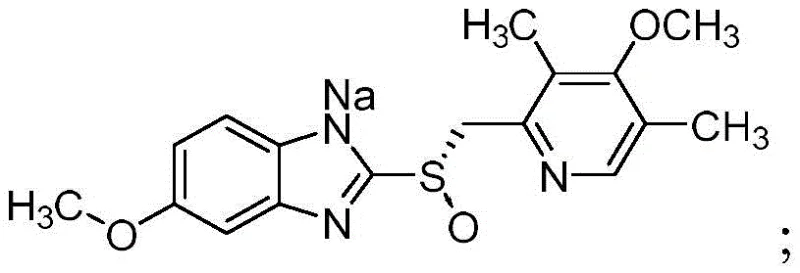
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of Esomeprazole Sodium has been plagued by significant technical hurdles that compromise both yield and optical purity. Prior art methods, such as those disclosed in WO 94/027988, relied heavily on toluene, a solvent with high toxicity profiles that pose severe environmental and occupational health risks in large-scale manufacturing. Other approaches utilizing acetone or acetone-water mixtures often failed to adequately remove single impurities, frequently leaving levels above 0.1%, which is unacceptable for modern regulatory standards. Furthermore, traditional recrystallization techniques typically necessitate a heating-dissolution followed by a cooling-crystallization cycle. This thermal shock is detrimental to Esomeprazole Sodium, as the compound is prone to decomposition at elevated temperatures, leading to increased sulfone impurity levels and a reduction in overall recovery rates. Additionally, methods involving chiral chromatography, while effective for separation, are prohibitively expensive and difficult to scale for industrial tonnage production, creating a bottleneck for cost reduction in pharmaceutical intermediate manufacturing.
The Novel Approach
The innovative process described in the patent data fundamentally reengineers the solvation dynamics to achieve purification without thermal stress. Instead of relying on temperature gradients to drive supersaturation, this method exploits the differential solubility of Esomeprazole Sodium in mixed solvent systems comprising both protic and aprotic components. The protocol initiates by dissolving the crude material in a specific ratio of a protic solvent, such as isopropanol, and an aprotic solvent, like butyl acetate or tetrahydrofuran. Once the crude is fully solubilized at ambient conditions, a secondary addition of aprotic solvent is introduced to the filtrate. This addition drastically reduces the solubility of the target compound, inducing rapid and controlled crystallization without the need for external cooling. This isothermal approach not only preserves the chemical stability of the sulfinyl group but also simplifies the equipment requirements, removing the need for complex heating and chilling jackets. Consequently, the commercial scale-up of complex pharmaceutical intermediates becomes significantly more feasible and economically viable.
Mechanistic Insights into Mixed-Solvent Isothermal Crystallization
The core mechanism driving this purification success lies in the precise manipulation of solvent polarity and hydrogen bonding interactions. Esomeprazole Sodium exhibits distinct solubility characteristics where it is highly soluble in protic solvents due to favorable hydrogen bonding with the benzimidazole nitrogen and the sulfinyl oxygen. Conversely, its solubility drops precipitously in aprotic organic solvents. By initially creating a homogeneous phase with a balanced mixture, the process ensures that all impurities, including the challenging R-enantiomer and sulfone byproducts, remain in solution or are filtered out prior to the crystallization trigger. When the excess aprotic solvent is added, the dielectric constant of the medium shifts, lowering the solvation energy of the S-enantiomer salt. This forces the target molecule out of the solution in a highly ordered crystal lattice. Because this phase transition occurs without temperature fluctuation, the kinetic energy of the molecules remains stable, minimizing the risk of racemization or oxidative degradation that typically occurs during heating phases. This mechanistic control is essential for maintaining the stringent stereochemical purity required for high-purity OLED material or pharmaceutical applications.
Furthermore, the selection of specific aprotic solvents such as butyl acetate plays a dual role in both crystallization efficiency and impurity rejection. Butyl acetate acts as an anti-solvent that is miscible with the protic component but poor at solvating the ionic sodium salt form of the drug. This selective precipitation ensures that non-polar organic impurities remain dissolved in the mother liquor while the polar drug substance crystallizes. The absence of a cooling step also prevents the occlusion of mother liquor within the crystal lattice, a common phenomenon in rapid cooling scenarios that leads to high solvent retention. By allowing the crystals to grow under steady-state isothermal conditions, the lattice formation is more perfect, effectively excluding impurity molecules from the crystal structure. This results in a final product with exceptionally low residual solvent levels and superior flow properties, which are critical downstream attributes for tablet compression and formulation processes in the pharmaceutical industry.
How to Synthesize Esomeprazole Sodium Efficiently
Implementing this purification strategy requires precise control over solvent ratios and mixing dynamics to ensure reproducibility at scale. The process begins with the careful selection of a protic solvent, with isopropanol being the preferred choice due to its optimal balance of solvency and ease of removal. The crude Esomeprazole Sodium is introduced to a mixture of isopropanol and an aprotic solvent like butyl acetate in a mass ratio ranging from 0.5:1 to 3.0:1 relative to the crude. Once dissolution is complete and insoluble particulates are removed via filtration, the critical crystallization step involves the addition of a larger volume of aprotic solvent, typically 4 to 10 times the mass of the crude material. This addition must be managed to ensure uniform supersaturation without localized over-concentration that could lead to oiling out. The detailed standardized synthesis steps see the guide below.
- Dissolve crude Esomeprazole Sodium in a mixed solvent system comprising a protic solvent (e.g., isopropanol) and an aprotic solvent (e.g., butyl acetate) at ambient temperature (0-30°C).
- Filter the solution to remove insoluble impurities, then introduce a significant volume of additional aprotic solvent to induce crystallization without cooling.
- Separate the resulting crystals via filtration or centrifugation and dry under reduced pressure at moderate temperatures (30-50°C) to obtain the refined product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this isothermal purification technology offers profound strategic benefits beyond mere technical specifications. The elimination of thermal cycling translates directly into substantial cost savings by reducing energy consumption associated with heating and refrigeration systems. Moreover, the stability of the process means that batch-to-batch variability is drastically minimized, ensuring a consistent supply of high-quality material that reduces the risk of production delays or rejected batches. The use of lower-toxicity solvents like butyl acetate and isopropanol, as opposed to toluene or methanol-heavy systems, simplifies waste treatment protocols and lowers the environmental compliance burden. This operational efficiency allows manufacturers to offer more competitive pricing structures while maintaining rigorous quality standards. Additionally, the high yield reported in the patent data, exceeding 85%, implies a more efficient utilization of raw materials, further driving down the cost of goods sold (COGS) for the final active pharmaceutical ingredient.
- Cost Reduction in Manufacturing: The removal of heating and cooling requirements significantly lowers utility costs and reduces the wear and tear on processing equipment. By avoiding thermal degradation, the process maximizes the recovery of the valuable S-enantiomer, meaning less starting material is wasted to decomposition. The simplified solvent system also reduces the complexity of solvent recovery and recycling operations, leading to streamlined operational expenditures. Furthermore, the high purity achieved in a single crystallization step potentially eliminates the need for secondary purification stages, shortening the overall production cycle time and reducing labor costs associated with extended processing.
- Enhanced Supply Chain Reliability: The robustness of this isothermal method makes it highly suitable for large-scale industrial production, ensuring that supply commitments can be met consistently. Since the process does not rely on sensitive temperature ramps, it is less susceptible to fluctuations in ambient conditions or equipment performance, guaranteeing stable output. The use of commercially available and widely sourced solvents like isopropanol and butyl acetate mitigates the risk of raw material shortages that might occur with more specialized reagents. This reliability is crucial for maintaining continuous manufacturing lines and preventing stockouts of critical gastrointestinal medications in the global market.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial production is straightforward because it avoids the heat transfer limitations often encountered in large reactors during heating and cooling cycles. The isothermal nature ensures uniform conditions throughout the vessel, regardless of size, facilitating seamless technology transfer. From an environmental perspective, the reduction in hazardous solvent usage and the minimization of degradation byproducts result in a greener manufacturing footprint. This aligns with increasingly strict global environmental regulations and corporate sustainability goals, making the supply chain more resilient against regulatory changes and enhancing the brand reputation of the manufacturing partner.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this novel purification technology. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering clarity on how this method outperforms traditional techniques. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this process into their existing manufacturing workflows. The answers highlight the specific advantages in terms of impurity control, solvent management, and operational simplicity that define this innovation.
Q: How does this purification method control the R-enantiomer impurity?
A: The method utilizes a specific solubility differential in mixed protic-aprotic solvents that favors the crystallization of the S-enantiomer while keeping impurities and the R-enantiomer in the mother liquor, achieving levels below 0.01%.
Q: Why is the isothermal process superior to traditional heating-cooling recrystallization?
A: Esomeprazole sodium is thermally sensitive. Traditional methods requiring heating to dissolve and cooling to crystallize often cause degradation and sulfone impurity formation. This isothermal method operates at 0-30°C, preserving chemical stability.
Q: What are the residual solvent advantages of this technique?
A: By using low-toxicity aprotic solvents like butyl acetate and avoiding high-boiling toxic solvents like toluene, the process ensures minimal solvent retention, easily meeting stringent ICH Q3C guidelines for pharmaceutical ingredients.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Esomeprazole Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent CN112661744B can be reliably replicated at an industrial level. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced HPLC and chiral column technologies to verify that every batch meets the <0.01% R-enantiomer threshold. Our commitment to quality assurance means that we do not just supply chemicals; we deliver validated solutions that integrate seamlessly into your downstream formulation processes, minimizing your risk and accelerating your time to market.
We invite you to engage with our technical procurement team to discuss how this advanced purification route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your project needs. Whether you are looking to secure a long-term supply of high-purity intermediates or need assistance with process optimization, NINGBO INNO PHARMCHEM is positioned to be your strategic ally in the competitive pharmaceutical landscape.
