Advanced Manufacturing of DTPA Derivatives for Next-Generation MRI Contrast Agents
Advanced Manufacturing of DTPA Derivatives for Next-Generation MRI Contrast Agents
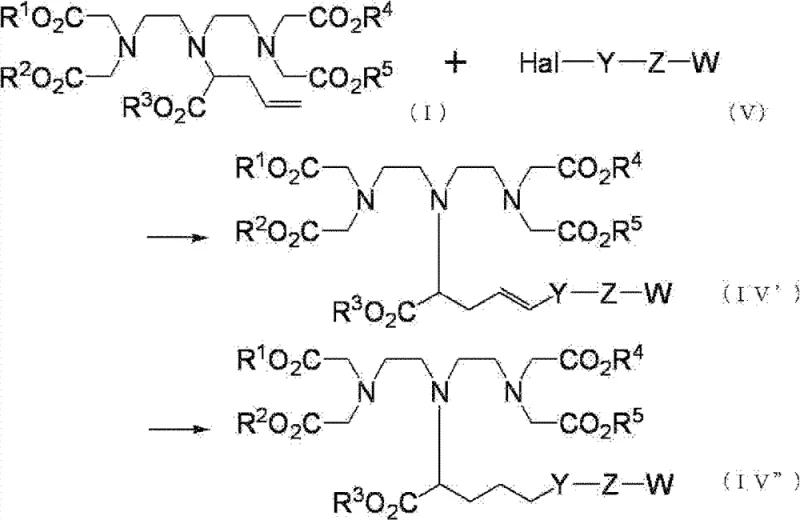
The landscape of diagnostic imaging is continuously evolving, driven by the demand for safer and more effective Magnetic Resonance Imaging (MRI) contrast agents. Central to this advancement is the synthesis of specialized ligands capable of chelating gadolinium ions while exhibiting improved pharmacokinetic properties, such as extended blood retention or organ specificity. Patent CN102548956B introduces a groundbreaking methodology for the preparation of diethylenetriaminepentaacetic acid (DTPA) derivatives, addressing critical bottlenecks in the current manufacturing of these high-value pharmaceutical intermediates. This technology offers a streamlined, three-step process that enhances regioselectivity and operational efficiency, positioning it as a vital asset for reliable pharmaceutical intermediate suppliers aiming to optimize their production pipelines for next-generation diagnostic agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of DTPA derivatives modified with substituents for improved blood retention has been fraught with chemical challenges. Traditional approaches often rely on multi-step condensation reactions between low-molecular-weight compounds containing the desired substituent and amine or carboxylic acid precursors. These legacy methods suffer from significant drawbacks, including poor atom economy and the generation of substantial quantities of waste, such as halide ion salts. Furthermore, achieving site-specific modification on the DTPA backbone is notoriously difficult; conventional alkylation strategies frequently result in a mixture of products where substituents are attached to multiple nitrogen atoms or incorrect carbon positions. This lack of selectivity necessitates complex and costly purification processes, drastically reducing the overall yield and making industrial scale-up economically unviable for many manufacturers seeking cost reduction in MRI contrast agent manufacturing.
The Novel Approach
In stark contrast to these inefficient legacy protocols, the process disclosed in CN102548956B employs a clever strategy centered on a modified Stevens rearrangement. By intentionally dividing the reaction into three distinct stages—quaternization, purification, and rearrangement—the inventors have achieved unprecedented control over the molecular architecture. The core innovation lies in the initial quaternization step, which is conducted under base-free conditions. This subtle yet powerful modification allows the reaction to proceed reversibly, naturally converging on the chemically most stable quaternary ammonium salt at the central nitrogen atom. Following the removal of excess reagents, a subsequent base treatment triggers the rearrangement, cleanly transferring the allyl group to the alpha-carbon position. This approach eliminates the need for harsh conditions that typically degrade sensitive ester groups, thereby preserving the integrity of the chelating scaffold while introducing the necessary reactive handle for further functionalization.
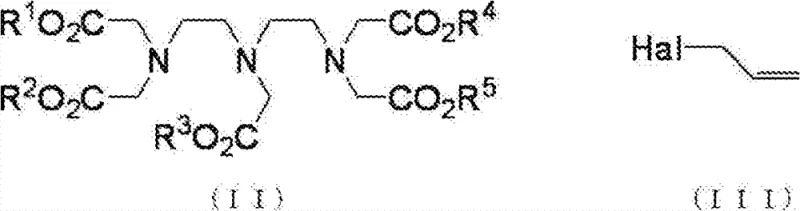
Mechanistic Insights into Base-Free Quaternization and Stevens Rearrangement
The mechanistic elegance of this process provides R&D directors with a robust framework for understanding impurity control and yield optimization. In the first stage, the reaction between the DTPA pentaester and the allyl halide occurs in an aprotic solvent without the addition of a base. Under standard conditions, strong bases would deprotonate the alpha-carbons, leading to uncontrolled nucleophilic attacks and a loss of position specificity. However, by omitting the base, the reaction relies on the nucleophilicity of the amine lone pairs. The central nitrogen atom exhibits slightly higher reactivity, and crucially, the quaternization reaction is reversible. This reversibility acts as a self-correcting mechanism; even if allylation occurs at the terminal nitrogens or if multiple allyl groups are added, the system equilibrates towards the thermodynamically favored mono-quaternized species at the central nitrogen. This dynamic equilibrium is the key to the high regioselectivity observed, effectively suppressing the formation of poly-alkylated byproducts that plague conventional syntheses.
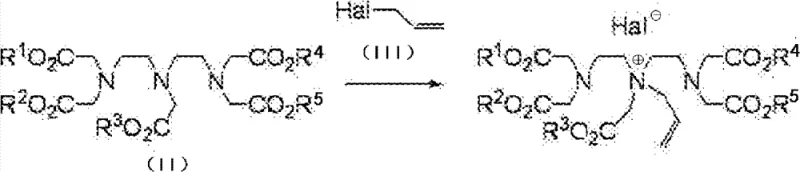
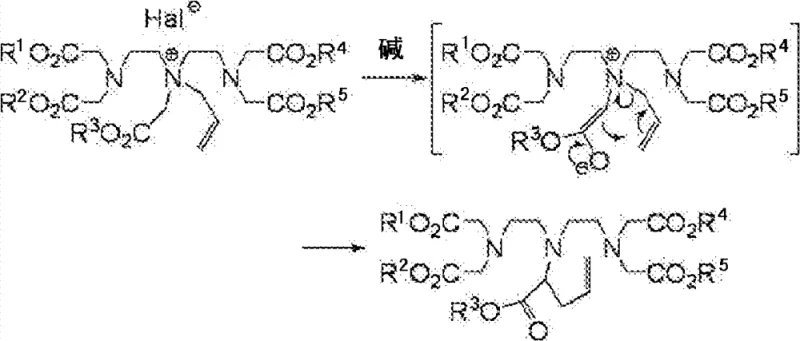
Following the isolation of the quaternary intermediate, the third stage involves the addition of a weak base, such as potassium carbonate, to induce the Stevens rearrangement. This step is critical for migrating the allyl group from the nitrogen to the adjacent carbon atom. The use of weak bases is particularly advantageous as it minimizes the risk of hydrolyzing the sensitive ester groups present on the DTPA backbone, a common side reaction when strong alkalis like sodium hydroxide are employed. The rearrangement proceeds through an ylide intermediate, ultimately yielding the desired alpha-allyl substituted DTPA derivative. This precise control over the reaction pathway ensures that the final product possesses the exact structural configuration required for subsequent coupling reactions, such as palladium-catalyzed cross-couplings, which are essential for attaching organ-specific targeting moieties. The ability to maintain high purity throughout these transformations significantly reduces the burden on downstream purification units.
How to Synthesize DTPA Derivatives Efficiently
The implementation of this synthesis route requires careful attention to solvent selection and stoichiometry to maximize the benefits of the reversible quaternization step. The process begins by dissolving the DTPA pentaester in an aprotic solvent like DMF or acetonitrile, followed by the addition of a large excess of allyl halide to drive the equilibrium forward. After the quaternization is complete, typically requiring temperatures between 20°C and 60°C, the excess allyl halide must be rigorously removed, often through repeated vacuum concentration cycles, to prevent interference in the rearrangement step. Finally, the residue is treated with a mild base in a suitable solvent to effect the rearrangement. For detailed operational parameters, safety guidelines, and specific workup procedures, please refer to the standardized synthesis guide below.
- React diethylenetriaminepentaacetic acid pentaester with an allyl halide in an aprotic solvent without a base to achieve selective quaternization at the central nitrogen.
- Remove the excess allyl halide reagent from the reaction mixture, typically via reduced pressure concentration, to prevent poly-alkylation in subsequent steps.
- Treat the quaternized intermediate with a weak base in a solvent to induce a Stevens rearrangement, shifting the allyl group to the alpha-carbon position.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic advantages beyond mere technical superiority. The simplification of the synthetic route directly impacts the cost structure of producing high-purity pharmaceutical intermediates. By eliminating the need for multiple protection and deprotection steps, the process reduces the consumption of expensive reagents and solvents, leading to substantial cost savings in raw material procurement. Furthermore, the high regioselectivity minimizes the formation of difficult-to-separate isomers, which streamlines the purification process and increases the overall throughput of the manufacturing facility. This efficiency gain is crucial for maintaining competitive pricing in the global market for diagnostic imaging agents.
- Cost Reduction in Manufacturing: The elimination of complex multi-step condensation sequences significantly lowers the operational expenditure associated with labor, energy, and waste disposal. Since the process avoids the use of expensive transition metal catalysts in the initial functionalization step and relies on readily available commodity chemicals like allyl bromide and DTPA esters, the direct material costs are drastically optimized. Additionally, the high conversion rates and selectivity reduce the volume of organic waste generated per kilogram of product, aligning with modern environmental compliance standards and reducing the financial burden of waste treatment.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials ensures a stable and resilient supply chain. Unlike proprietary precursors that may be subject to single-source bottlenecks, the key inputs for this synthesis are produced by multiple global suppliers, mitigating the risk of disruption. The robustness of the reaction conditions, which tolerate moderate temperatures and do not require stringent anhydrous environments for the rearrangement step, further enhances manufacturing reliability, allowing for consistent production schedules and reduced lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex pharmaceutical intermediates. The use of common aprotic solvents and weak inorganic bases simplifies the engineering requirements for large-scale reactors, avoiding the need for specialized equipment required for cryogenic or high-pressure reactions. Moreover, the reduction in halide salt waste and the avoidance of heavy metal catalysts in the primary bond-forming steps contribute to a greener manufacturing profile, facilitating easier regulatory approval and supporting corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this DTPA derivative synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on the practical application of the Stevens rearrangement strategy in an industrial setting.
Q: How does this new process improve regioselectivity compared to conventional methods?
A: Conventional methods often struggle with controlling the position of substitution on the DTPA backbone, leading to mixtures. This patent utilizes a unique base-free quaternization step where the reaction reversibility allows the system to converge on the thermodynamically most stable central nitrogen quaternary salt, ensuring high regioselectivity before the rearrangement step.
Q: What are the primary advantages for large-scale manufacturing of MRI contrast agent intermediates?
A: The process significantly simplifies the synthetic route by reducing the number of steps required to introduce functional substituents. It avoids the need for complex protection-deprotection sequences typical in older methodologies, thereby reducing waste generation and improving overall throughput for industrial production.
Q: Can this method be adapted for different functional groups on the final contrast agent?
A: Yes, the resulting DTPA derivative contains a reactive allyl group. This handle allows for further diversification through palladium-catalyzed coupling reactions with various halogenated aryl or vinyl compounds, enabling the synthesis of a wide range of organ-specific or blood-pool MRI contrast agents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable DTPA Derivative Supplier
As the demand for advanced diagnostic agents grows, partnering with a manufacturer who understands the nuances of complex ligand synthesis is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging deep technical expertise to deliver high-quality intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous volume requirements of global pharmaceutical partners. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of DTPA derivative meets the exacting standards required for GMP-grade MRI contrast agent production.
We invite you to explore how our advanced manufacturing capabilities can support your development pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain efficiency and accelerate your time to market.
