Advanced Synthesis of 2-Chloro-3-Fluorotoluene for High-Performance Herbicide Manufacturing
The global demand for high-performance agrochemicals continues to drive innovation in intermediate synthesis, particularly for herbicides like Penoxsulam which rely on fluorinated aromatic building blocks. Patent CN116120145A introduces a groundbreaking preparation method for 2-chloro-3-fluorotoluene, a critical precursor in this value chain, by fundamentally rethinking raw material sourcing. Instead of relying on expensive, dedicated starting materials, this technology leverages the comprehensive utilization of 2-chloro-3-nitrotoluene, a ubiquitous by-product from the nitration of o-chlorotoluene. This strategic shift not only addresses waste management challenges inherent in large-scale chemical manufacturing but also establishes a robust economic foundation for production. By integrating catalytic hydrogenation and diazotization-fluorination technologies, the process achieves exceptional efficiency and purity profiles that are essential for downstream pharmaceutical and agrochemical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-chloro-3-fluorotoluene has been plagued by inefficient multi-step pathways that burden supply chains with excessive operational costs and complex purification requirements. Traditional methods, such as those reported in prior art like CN107488098, often commence with m-toluidine and necessitate a cumbersome sequence involving diazotization, sulfonation, chlorination, and finally, desulfonation to achieve the desired substitution pattern. This elongated synthetic trajectory inherently suffers from cumulative yield losses at each stage, requiring significant volumes of solvents and reagents while generating substantial chemical waste. Furthermore, the chlorination of the benzene ring in these conventional routes frequently produces unwanted isomers, mandating additional energy-intensive rectification processes to isolate the target compound. These structural inefficiencies translate directly into higher manufacturing overheads and extended lead times, creating vulnerabilities in the supply of critical agrochemical intermediates.
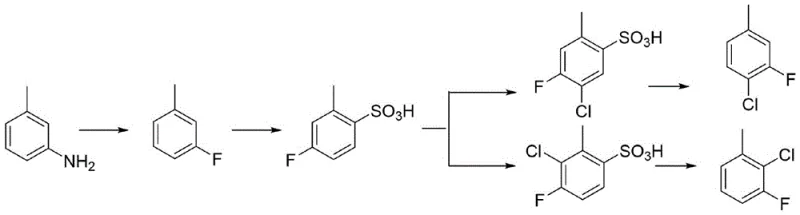
The Novel Approach
In stark contrast to these legacy processes, the methodology disclosed in CN116120145A streamlines production into a concise, two-stage operation that maximizes atom economy and minimizes environmental footprint. The innovation lies in the direct conversion of 2-chloro-3-nitrotoluene, transforming a low-value by-product into a high-value fluorinated intermediate through catalytic hydrogenation followed by a Schiemann-type fluorination. This approach eliminates the need for blocking groups like sulfonic acid, thereby removing entire unit operations from the manufacturing schedule. The reaction pathway is highly selective, avoiding the formation of difficult-to-separate isomers that plague electrophilic substitution methods. By shortening the synthetic route and utilizing readily available waste streams as feedstock, this novel approach offers a compelling solution for cost reduction in agrochemical intermediate manufacturing while maintaining rigorous quality standards required by global regulatory bodies.
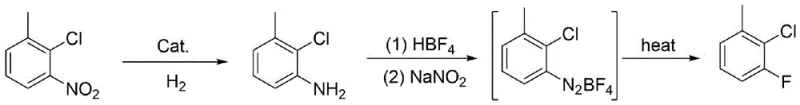
Mechanistic Insights into Catalytic Hydrogenation and Diazotization
The first stage of this sophisticated synthesis involves the catalytic hydrogenation reduction of the nitro group to an amino group, a transformation that requires precise control over reaction parameters to ensure safety and selectivity. Utilizing catalysts such as Raney Nickel, Pd/C, or Pt/C within an organic solvent matrix like methanol, the reaction proceeds under hydrogen pressures ranging from 0.5 to 1.5 MPa at temperatures between 50 and 100°C. This heterogeneous catalysis facilitates the cleavage of nitrogen-oxygen bonds while preserving the sensitive carbon-chlorine bond on the aromatic ring, a critical selectivity challenge in halogenated nitroarene reductions. The mechanism involves the adsorption of hydrogen and the nitro compound onto the metal surface, where sequential electron and proton transfers convert the nitro functionality into a primary amine, yielding 2-chloro-3-methylaniline with reported efficiencies reaching up to 99%. The choice of solvent and catalyst loading is optimized to prevent over-reduction or dehalogenation, ensuring the structural integrity of the intermediate for the subsequent fluorination step.
Following the reduction, the process advances to the diazotization and thermal decomposition phase, commonly known as the Balz-Schiemann reaction, which is the cornerstone for introducing fluorine into the aromatic system. The freshly synthesized 2-chloro-3-methylaniline reacts with fluoroboric acid to form a stable salt, which is then treated with sodium nitrite at low temperatures, typically between -15 and 0°C, to generate the diazonium fluoroborate species. This unstable intermediate is isolated, washed, and dried before undergoing thermal cracking at elevated temperatures of 160 to 250°C. During this pyrolysis, the diazonium group decomposes to release nitrogen gas, leaving behind a highly reactive aryl cation that immediately captures a fluoride ion from the tetrafluoroborate counterion. This mechanism ensures the regio-specific installation of the fluorine atom ortho to the chlorine substituent, producing 2-chloro-3-fluorotoluene with high purity and minimizing the formation of phenolic or chloro-by-products often seen in nucleophilic aromatic substitution alternatives.
How to Synthesize 2-Chloro-3-Fluorotoluene Efficiently
Implementing this synthesis protocol requires adherence to specific operational guidelines to maximize yield and safety, particularly given the involvement of high-pressure hydrogenation and thermally unstable diazonium salts. The process begins with the charging of 2-chloro-3-nitrotoluene and the selected catalyst into a pressure vessel, followed by purging with hydrogen to establish an inert atmosphere before pressurization. Detailed standard operating procedures regarding temperature ramping, pressure maintenance, and filtration techniques are critical to reproducing the high yields documented in the patent examples. For the second stage, strict temperature control during diazotization is paramount to prevent premature decomposition, while the thermal cracking step requires efficient gas handling systems to manage nitrogen evolution and product distillation. The detailed standardized synthesis steps see the guide below for a comprehensive breakdown of reagent ratios and processing conditions.
- Perform catalytic hydrogenation of 2-chloro-3-nitrotoluene using Raney Nickel or Pd/C in methanol at 50-100°C and 0.5-1.5 MPa pressure to obtain 2-chloro-3-methylaniline.
- React the resulting amine with fluoroboric acid and sodium nitrite at low temperatures (-15 to 0°C) to form the diazonium fluoroborate salt.
- Thermally decompose the dried diazonium salt at 160-250°C and collect the distilled liquid to isolate the final 2-chloro-3-fluorotoluene product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology represents a strategic opportunity to optimize cost structures and enhance supply security for fluorinated intermediates. The fundamental economic driver of this process is the utilization of 2-chloro-3-nitrotoluene, a by-product that is often available at a fraction of the cost of dedicated synthetic starting materials. By converting waste into value, manufacturers can significantly reduce the raw material cost basis, insulating the supply chain from volatility in the pricing of primary petrochemical feedstocks. Furthermore, the elimination of multiple synthetic steps such as sulfonation and desulfonation drastically simplifies the production workflow, leading to reduced utility consumption, lower labor requirements, and decreased capital expenditure on reactor capacity. This streamlined operation translates into substantial cost savings in agrochemical intermediate manufacturing without compromising on the quality or purity of the final output.
- Cost Reduction in Manufacturing: The economic model of this synthesis is predicated on the valorization of low-cost by-products, effectively turning a waste disposal liability into a revenue-generating asset. By bypassing the need for expensive blocking groups and the associated reagents for sulfonation and desulfonation, the overall chemical consumption per kilogram of product is markedly lower. Additionally, the high yields achieved in both the hydrogenation and fluorination steps minimize material loss, ensuring that a greater proportion of input mass is converted into saleable product. This efficiency gain allows for competitive pricing strategies in the global market for herbicide intermediates, providing a distinct margin advantage over competitors relying on legacy multi-step syntheses.
- Enhanced Supply Chain Reliability: Sourcing raw materials from established nitration lines ensures a consistent and abundant supply of the key precursor, 2-chloro-3-nitrotoluene, reducing the risk of shortages associated with niche starting materials. The robustness of the catalytic hydrogenation step, which tolerates standard industrial catalysts like Raney Nickel or Pd/C, further enhances operational reliability by allowing flexibility in catalyst procurement. Moreover, the simplified process flow reduces the number of potential failure points in the manufacturing line, leading to more predictable production schedules and shorter lead times for high-purity agrochemical intermediates. This stability is crucial for downstream customers who require just-in-time delivery to maintain their own herbicide production schedules.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing unit operations such as high-pressure hydrogenation and distillation that are standard in fine chemical facilities, facilitating easy transition from pilot to commercial scale. From an environmental perspective, the reduction in synthetic steps leads to a significant decrease in solvent usage and waste generation, aligning with increasingly stringent global regulations on industrial emissions. The ability to utilize a by-product also contributes to a circular economy model, improving the overall sustainability profile of the manufacturing site. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the brand value of the supply chain for environmentally conscious end-users.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented synthesis route. These insights are derived directly from the experimental data and process descriptions within CN116120145A, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding these nuances is essential for making informed decisions about process adoption and supplier qualification.
Q: What is the primary raw material source for this synthesis method?
A: The process utilizes 2-chloro-3-nitrotoluene, which is a by-product generated during the nitration of o-chlorotoluene for other industrial intermediates, ensuring low raw material costs and waste valorization.
Q: How does this method improve upon conventional synthesis routes?
A: Unlike traditional multi-step routes involving sulfonation and desulfonation, this method achieves the target molecule in just two main steps with high yields, significantly reducing energy consumption and processing time.
Q: What catalysts are suitable for the reduction step?
A: The patent specifies the use of Raney Nickel, Pd/C, or Pt/C catalysts, with Pd/C or Pt/C typically containing 0.5% to 2% metal content, offering flexibility in catalyst selection based on availability and cost.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloro-3-Fluorotoluene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient intermediate synthesis plays in the competitiveness of the global agrochemical industry. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like the one described in CN116120145A are successfully translated into robust industrial operations. Our state-of-the-art facilities are equipped to handle the specific requirements of catalytic hydrogenation and hazardous diazonium chemistry, adhering to stringent purity specifications and rigorous QC labs to guarantee product consistency. We are committed to delivering high-purity 2-chloro-3-fluorotoluene that meets the exacting standards required for the synthesis of next-generation herbicides.
We invite you to collaborate with us to leverage this advanced technology for your supply chain needs. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this by-product valorization route can optimize your total cost of ownership. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply of this vital chemical building block.
