Advanced Pd-Catalyzed Regioselective Halogenation for High-Purity Pharmaceutical Intermediates
Advanced Pd-Catalyzed Regioselective Halogenation for High-Purity Pharmaceutical Intermediates
The landscape of fine chemical synthesis is constantly evolving, driven by the need for safer, more efficient, and environmentally sustainable processes. A pivotal advancement in this domain is detailed in Chinese Patent CN1209336C, which discloses a novel process for the production of 2-halobenzoic acids. These compounds serve as critical building blocks in the manufacture of high-value pharmaceuticals and agrochemicals. The patent introduces a transformative methodology that utilizes a palladium catalyst to achieve regioselective halogenation of benzoic acid derivatives under mild conditions. This technological breakthrough addresses long-standing challenges in the industry, such as the reliance on hazardous reagents and the difficulty in controlling substitution patterns on the aromatic ring. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediate supplier, understanding the nuances of this catalytic system is essential for optimizing supply chains and reducing manufacturing costs.
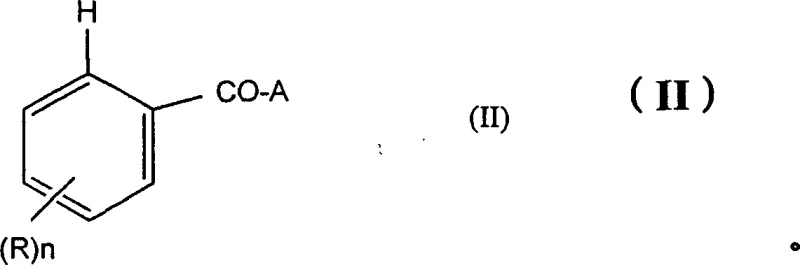
The core innovation lies in the ability to introduce a halogen atom—specifically chlorine, bromine, or iodine—precisely at the 2-position of the benzoic acid scaffold. The general formula (I) depicted above illustrates the structural versatility of the products, where the substituent A can vary from hydroxyl and alkali metal salts to complex amide groups. This flexibility allows for the synthesis of a wide array of intermediates required for drug discovery and development. By leveraging this patented technology, manufacturers can access high-purity OLED material precursors and specialty chemical intermediates that were previously difficult to produce with high fidelity. The implications for cost reduction in electronic chemical manufacturing and pharmaceutical synthesis are profound, as the process eliminates the need for stoichiometric amounts of toxic metals and harsh reaction conditions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-halobenzoic acids has been plagued by significant technical and safety hurdles. Traditional methods, such as the Sandmeyer reaction involving 2-aminobenzoic acid, carry an inherent risk of explosion and generate substantial quantities of acidic wastewater, posing severe environmental compliance challenges. Alternative approaches utilizing alkyllithium reagents require cryogenic conditions, often below 0°C, which drastically increases energy consumption and operational complexity. Furthermore, methods employing thallium(III) trifluoroacetate necessitate the use of stoichiometric amounts of highly toxic heavy metals, creating dangerous waste streams that are costly to treat and dispose of safely. Even earlier attempts at palladium-catalyzed halogenation often required substrates with specific heteroatom directing groups directly attached to the benzene ring or at the benzylic position, limiting the scope of applicable starting materials. Additionally, many of these prior art methods consumed palladium in near-stoichiometric quantities (5-20% based on substrate), negating the economic benefits typically associated with catalysis.
The Novel Approach
In stark contrast, the process described in CN1209336C offers a streamlined and robust solution. By reacting benzoic acid derivatives of general formula (II) with a halogenating agent in the presence of a catalytic amount of palladium, the invention achieves high yields and perfect regioselectivity. As illustrated in the reaction scheme below, the transformation proceeds efficiently without the need for extreme temperatures or hazardous reagents. The use of catalytic palladium, potentially at loadings as low as 1/10,000 equivalent, represents a paradigm shift in resource efficiency. This approach not only mitigates the safety risks associated with diazonium salts and cryogenic operations but also significantly simplifies the downstream purification process. The ability to utilize common solvents such as acetic acid, DMF, or THF further enhances the practicality of this method for commercial scale-up of complex polymer additives and pharmaceutical intermediates.
Mechanistic Insights into Pd-Catalyzed Regioselective Halogenation
The success of this methodology hinges on the unique interaction between the palladium catalyst and the benzoic acid substrate. Unlike traditional electrophilic aromatic substitution which often yields mixtures of ortho, meta, and para isomers, this palladium-catalyzed system exhibits exceptional control over the site of halogenation. The mechanism likely involves the coordination of the palladium species to the carbonyl oxygen or the nitrogen atom of the amide group in the substrate. This coordination directs the metal center to the adjacent ortho-position, facilitating the activation of the C-H bond at that specific site. Once the C-H bond is activated, the halogenating agent—whether it be molecular halogens like I2 and Br2 or N-halo succinimides like NIS and NCS—reacts with the organopalladium intermediate to install the halogen atom. This directed C-H activation strategy ensures that the halogen is introduced exclusively at the 2-position, adjacent to the carboxyl or amide functionality, thereby eliminating the formation of unwanted regioisomers that complicate purification.
From an impurity control perspective, this mechanistic pathway offers distinct advantages. The high regioselectivity (often reported as 100% in the patent examples) means that the crude reaction mixture contains minimal structural impurities, reducing the burden on chromatographic separation steps. Furthermore, the mild reaction conditions (typically 50°C to 120°C) prevent the degradation of sensitive functional groups that might be present on the aromatic ring or the side chains. For instance, substrates containing nitro, cyano, or trifluoromethyl groups remain intact throughout the process. The choice of halogenating agent also plays a crucial role; N-iodosuccinimide (NIS) and N-chlorosuccinimide (NCS) are particularly effective, providing a controlled source of electrophilic halogen that complements the palladium cycle. This precise control over the reaction trajectory ensures that the final product meets the stringent purity specifications required for active pharmaceutical ingredients (APIs) and high-performance agrochemicals.
How to Synthesize 2-Halobenzoic Acid Efficiently
The synthesis of these valuable intermediates is straightforward and adaptable to various scales of production. The process begins with the selection of an appropriate benzoic acid derivative, which serves as the substrate. This substrate is then combined with a palladium catalyst, such as palladium acetate or palladium chloride, and a halogenating agent in a suitable inert solvent. The reaction mixture is heated to a moderate temperature, typically ranging from room temperature to the boiling point of the solvent, with a preferred range of 50°C to 120°C. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining the benzoic acid derivative substrate with a catalytic amount of palladium catalyst (e.g., Pd(OAc)2) and a suitable halogenating agent in an inert solvent.
- Heat the mixture to a temperature between 40°C and 200°C, preferably 50°C to 120°C, while stirring to facilitate the regioselective halogenation at the ortho-position.
- Upon completion, cool the reaction, perform aqueous workup including extraction with organic solvents like ethyl acetate, and purify the crude product to obtain high-purity 2-halobenzoic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process translates into tangible strategic benefits. The shift from stoichiometric toxic reagents to a catalytic system fundamentally alters the cost structure and risk profile of manufacturing 2-halobenzoic acids. By eliminating the need for expensive and hazardous materials like thallium salts or large quantities of alkyllithium reagents, the process inherently lowers the raw material costs. Moreover, the mild operating conditions reduce energy consumption, as there is no need for energy-intensive cryogenic cooling or high-temperature furnaces. This efficiency gain is compounded by the simplified waste management protocol; the absence of heavy metal sludge and acidic wastewater significantly reduces disposal costs and regulatory compliance burdens. Consequently, this technology enables substantial cost savings in the production of key intermediates used across the pharmaceutical and agrochemical sectors.
- Cost Reduction in Manufacturing: The utilization of palladium in truly catalytic amounts, potentially as low as 1/10,000 equivalent, drastically reduces the dependency on precious metals compared to prior art methods requiring 5-20% loading. This reduction in catalyst loading, combined with the use of inexpensive halogenating agents like N-halosuccinimides, leads to a significantly lower cost of goods sold (COGS). Furthermore, the high regioselectivity minimizes the loss of valuable starting materials to side products, improving the overall atom economy of the process. The elimination of complex purification steps required to remove stoichiometric metal waste further contributes to operational cost reductions, making the final product more competitive in the global market.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures a more stable and predictable supply of critical intermediates. Unlike methods reliant on unstable diazonium salts or moisture-sensitive organolithium reagents, the reagents used in this process are commercially available and stable under standard storage conditions. This stability reduces the risk of production delays caused by reagent degradation or handling difficulties. Additionally, the compatibility of the process with a wide range of solvents and substrates provides flexibility in sourcing raw materials, allowing manufacturers to adapt quickly to market fluctuations. This resilience is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard reactor equipment and common solvents that are easily handled on a multi-ton scale. The mild reaction conditions and lack of explosive hazards make it safer to operate at large volumes, reducing the need for specialized containment facilities. From an environmental standpoint, the significant reduction in hazardous waste generation aligns with increasingly strict global regulations on chemical manufacturing. By minimizing the discharge of toxic heavy metals and acidic effluents, manufacturers can achieve better environmental, social, and governance (ESG) ratings. This compliance not only avoids potential fines but also enhances the brand reputation of the supplier as a responsible partner in the sustainable chemistry movement.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed halogenation technology. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these details is vital for technical teams evaluating the feasibility of integrating this process into their existing manufacturing workflows.
Q: What are the advantages of this Pd-catalyzed method over traditional Sandmeyer reactions?
A: Unlike Sandmeyer reactions which pose explosion risks and generate large amounts of acidic wastewater, this Pd-catalyzed process operates under mild conditions with high regioselectivity and significantly reduced environmental impact.
Q: Can this process be scaled for commercial production of agrochemical intermediates?
A: Yes, the use of catalytic amounts of palladium (as low as 1/10,000 equivalent) and common solvents like DMF or acetic acid makes the process highly scalable and cost-effective for industrial manufacturing.
Q: What is the regioselectivity of the halogenation reaction?
A: The process demonstrates excellent regioselectivity, specifically targeting the 2-position (ortho-position) relative to the carboxyl or amide group, often achieving 100% selectivity as shown in patent examples.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Halobenzoic Acid Supplier
The technological potential of the Pd-catalyzed regioselective halogenation process described in CN1209336C is immense, offering a pathway to high-quality intermediates with superior efficiency. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order size. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 2-halobenzoic acid meets the exacting standards required for API synthesis. We are committed to leveraging advanced catalytic technologies to deliver value-driven solutions for the global fine chemical industry.
We invite you to collaborate with us to optimize your supply chain for these critical building blocks. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. Please contact us to request specific COA data and route feasibility assessments for your target molecules. By partnering with NINGBO INNO PHARMCHEM, you secure a reliable source of high-purity intermediates while benefiting from our expertise in process optimization and regulatory compliance.
