Scalable Synthesis of Phosphine Oxide Vitamin D Precursors for Global Pharma Supply Chains
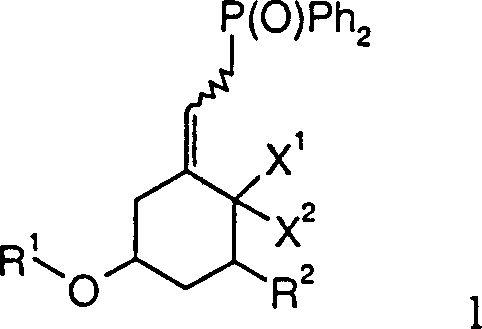
The pharmaceutical industry continuously demands more efficient and scalable routes for the synthesis of complex bioactive molecules, particularly Vitamin D analogs which are critical for treating metabolic bone diseases and psoriasis. Patent CN1176091C introduces a robust methodology for preparing phosphine oxide compounds of Formula 1, which serve as essential A-ring synthons in the total synthesis of these vital therapeutics. This technology addresses long-standing challenges in intermediate manufacturing by utilizing a streamlined two-step sequence involving triphosgene-mediated chlorination followed by nucleophilic substitution. By leveraging this specific chemical architecture, manufacturers can achieve significant improvements in process reliability and product consistency. The strategic implementation of this pathway allows for the precise installation of the phosphine oxide moiety, a critical functionality for subsequent Wittig-type couplings that construct the final Vitamin D skeleton. As a leading entity in fine chemical manufacturing, understanding the nuances of such patented processes is paramount for maintaining a competitive edge in the global supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of allylic phosphine oxides for Vitamin D synthesis has been plagued by inefficiencies inherent in older chlorination and substitution protocols. Traditional methods often relied on hazardous gaseous reagents or harsh acidic conditions that compromised the integrity of sensitive protecting groups, such as silyl ethers, which are ubiquitous in steroid chemistry. These conventional routes frequently suffered from poor regioselectivity and significant formation of elimination by-products, leading to complex purification burdens and reduced overall yields. Furthermore, the instability of intermediate allylic halides under standard conditions often necessitated immediate usage without isolation, complicating logistics and quality control in a multi-kilogram production environment. The reliance on stoichiometric amounts of toxic heavy metals or difficult-to-remove catalysts in alternative pathways further exacerbated environmental compliance issues and increased the cost of goods sold due to extensive waste treatment requirements.
The Novel Approach
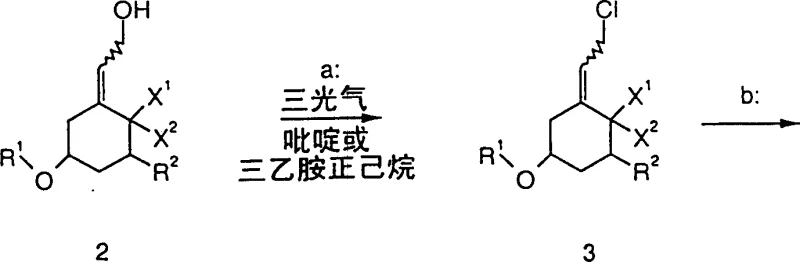
The innovative process detailed in the patent data circumvents these historical bottlenecks by employing triphosgene as a safe, solid-source equivalent of phosgene for the activation of allylic alcohols. This transformation proceeds under mild basic conditions using organic amines like pyridine or triethylamine, ensuring compatibility with acid-labile protecting groups such as tert-butyldimethylsilyl (TBS) ethers. The resulting allylic chloride intermediate is generated with high fidelity and can be efficiently displaced by diphenylphosphine oxide salts without significant erosion of stereochemical integrity. This approach not only simplifies the operational workflow by avoiding cryogenic extremes in the initial activation step but also enhances the safety profile of the manufacturing process by eliminating the need for handling gaseous toxins. The modularity of this synthesis allows for the accommodation of various substituents at the C-1 and C-5 positions, making it a versatile platform for generating diverse libraries of Vitamin D analogs.
Mechanistic Insights into Triphosgene-Mediated Chlorination and Substitution
The core of this synthetic strategy lies in the mechanistic elegance of converting an allylic alcohol into a reactive electrophile using triphosgene. Upon interaction with the organic base, triphosgene decomposes to generate phosgene in situ, which reacts with the hydroxyl group of the precursor to form a chloroformate intermediate. This activated species undergoes rapid nucleophilic attack by chloride ions, facilitated by the base, to yield the corresponding allylic chloride with inversion or retention depending on the specific ion-pairing dynamics. The choice of solvent, typically a proton-inert hydrocarbon like hexane, is critical to prevent solvolysis and ensure the stability of the reactive chloride intermediate. This controlled activation prevents the rearrangement of the double bond, preserving the crucial Z-configuration required for the biological activity of the final Vitamin D metabolite.
Subsequent substitution involves the generation of a potent nucleophile via the deprotonation of diphenylphosphine oxide using sodium hydride in a polar aprotic solvent such as DMF. The resulting phosphine oxide anion attacks the allylic chloride intermediate in an SN2-prime or direct SN2 fashion, displacing the chloride leaving group. The use of DMF is instrumental here as it effectively solvates the cation, increasing the nucleophilicity of the phosphorus species. Temperature control during this exothermic step is vital; maintaining the reaction between -80°C and 50°C prevents competing elimination reactions that could lead to diene impurities. This mechanistic precision ensures that the final phosphine oxide product retains the necessary stereochemistry for downstream coupling reactions, thereby securing the structural fidelity of the complex steroid framework.
How to Synthesize Phosphine Oxide Vitamin D Precursor Efficiently
Executing this synthesis requires strict adherence to the optimized parameters regarding reagent stoichiometry and thermal management to maximize yield and purity. The process begins with the careful addition of triphosgene to a solution of the allylic alcohol precursor in hexane, followed by the controlled introduction of the organic base to manage the exotherm. Once the chlorination is complete, the intermediate is isolated or telescoped directly into the substitution phase where the phosphine oxide salt is prepared separately. Detailed standard operating procedures regarding quenching, extraction, and crystallization are essential to remove inorganic salts and residual solvents effectively. For a comprehensive breakdown of the specific experimental conditions and workup protocols, please refer to the standardized guide below.
- Chlorinate the allylic alcohol precursor (Formula 2) using triphosgene and an organic base like pyridine or triethylamine in an aprotic solvent.
- Generate the alkali metal salt of diphenylphosphine oxide in situ using sodium hydride in DMF.
- React the intermediate allylic chloride (Formula 3) with the phosphine oxide salt at controlled low temperatures to yield the target phosphine oxide (Formula 1).
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this triphosgene-based methodology offers substantial strategic benefits for procurement and supply chain management within the pharmaceutical sector. The shift from gaseous chlorinating agents to solid triphosgene drastically simplifies logistics, reducing the regulatory burden and safety infrastructure costs associated with storing and transporting hazardous gases. This operational simplification translates directly into enhanced supply chain reliability, as the risk of shipment delays due to hazardous material restrictions is significantly mitigated. Furthermore, the high selectivity of the reaction minimizes the formation of difficult-to-separate impurities, which reduces the consumption of chromatography media and solvents during purification. This efficiency gain leads to a marked reduction in manufacturing costs and waste disposal fees, aligning with modern green chemistry initiatives and sustainability goals.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of commodity chemicals like triphosgene and sodium hydride drive down raw material costs significantly. By avoiding complex catalytic cycles that require rigorous metal scavenging steps, the process reduces both the cost of goods and the time required for quality assurance testing. The high yield observed in the exemplary data suggests that less starting material is wasted, optimizing the overall material balance and improving the economic viability of producing high-value Vitamin D intermediates at scale.
- Enhanced Supply Chain Reliability: The reliance on widely available, stable reagents ensures a consistent supply of critical inputs, shielding production schedules from the volatility often seen with specialized or custom-synthesized catalysts. The robustness of the reaction conditions allows for flexibility in manufacturing sites, enabling multi-site production strategies that mitigate the risk of single-point failures. This resilience is crucial for maintaining continuous supply to downstream API manufacturers who depend on timely delivery of these key building blocks for their own production timelines.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from gram to multi-kilogram scales without loss of efficiency or safety. The use of standard solvents and the absence of persistent organic pollutants or heavy metals simplify wastewater treatment and regulatory compliance. This environmental compatibility facilitates faster permitting for new production lines and reduces the long-term liability associated with hazardous waste management, making it an attractive option for sustainable chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the patent specifications and practical experience in scaling similar organic transformations. Understanding these nuances helps stakeholders make informed decisions regarding technology transfer and process validation. For further technical clarification or specific data requests, our team is available to provide detailed support.
Q: What are the key advantages of using triphosgene over traditional chlorinating agents?
A: Triphosgene offers superior handling safety as a solid compared to gaseous phosgene, while providing high atom economy and clean conversion to allylic chlorides under mild conditions.
Q: How does this process impact the purity profile of Vitamin D intermediates?
A: The method minimizes side reactions typical of harsh acidic conditions, resulting in cleaner crude products that require less rigorous purification, thereby enhancing overall yield and purity specifications.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the use of stable reagents like triphosgene and standard solvents like hexane and DMF makes the process highly scalable and compatible with existing industrial infrastructure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phosphine Oxide Vitamin D Precursor Supplier
At NINGBO INNO PHARMCHEM, we combine deep technical expertise with robust manufacturing capabilities to deliver high-quality pharmaceutical intermediates. Our facility is equipped to handle complex synthetic pathways, including the triphosgene-mediated processes described in CN1176091C, ensuring that your supply needs are met with precision and consistency. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, adhering to stringent purity specifications and operating within rigorous QC labs to guarantee product integrity. Our commitment to excellence extends beyond mere synthesis; we offer comprehensive process optimization services to enhance yield and reduce costs for our global partners.
We invite you to collaborate with us to leverage this advanced technology for your Vitamin D analog programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing prowess can accelerate your development timelines and secure your supply chain.
