Advanced Stereoselective Synthesis of Chiral Cyclopentanones for High-Purity Gabapentin Analogs
Advanced Stereoselective Synthesis of Chiral Cyclopentanones for High-Purity Gabapentin Analogs
The pharmaceutical industry's relentless pursuit of novel neurological therapeutics has placed a premium on the availability of high-purity chiral building blocks, particularly those serving as precursors for gabapentinoids. Patent CN1438985A introduces a groundbreaking methodology for the preparation of enantiomerically pure compounds of formula (I) and (II), which serve as critical intermediates in the synthesis of gabapentin analogs used for treating epilepsy, neuropathic pain, and neurodegenerative disorders. This technology represents a paradigm shift from reliance on scarce natural sources to a robust, fully synthetic pathway that guarantees stereochemical integrity. By leveraging chiral 4-acetoxycyclopent-2-en-1-one derivatives, manufacturers can now access a broad spectrum of 3-substituted and 3,4-disubstituted cyclopentanones with precise control over absolute configuration. 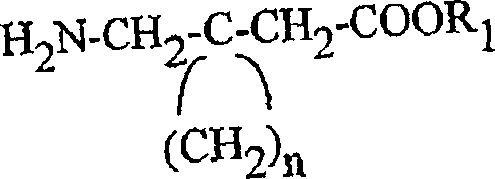 For R&D directors and procurement strategists, this patent offers a viable solution to the supply chain bottlenecks often associated with complex chiral intermediates, enabling the reliable production of high-purity pharmaceutical intermediates required for next-generation anticonvulsant therapies.
For R&D directors and procurement strategists, this patent offers a viable solution to the supply chain bottlenecks often associated with complex chiral intermediates, enabling the reliable production of high-purity pharmaceutical intermediates required for next-generation anticonvulsant therapies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral cyclopentanones required for neurological drug candidates has been plagued by significant inefficiencies and supply chain vulnerabilities. Traditional approaches often relied heavily on the isolation of chiral pools from natural sources, such as terpenes or amino acids, which inherently limits the structural diversity and scalability of the final product. For instance, while (R)-3-methyl-cyclopentanone might be accessible from natural origins, its (S)-enantiomer is notoriously difficult to obtain in high optical purity without complex and costly resolution processes. Furthermore, existing synthetic routes described in prior art, such as those referenced in WO 99/21824, frequently involve multi-step sequences with poor atom economy, requiring harsh conditions or expensive chiral auxiliaries that drive up the cost of goods sold (COGS). These conventional methods often struggle to introduce diverse alkyl or alkenyl substituents at the 3 and 4 positions of the cyclopentane ring without compromising stereochemical fidelity, leading to batches with unacceptable levels of diastereomeric impurities that complicate downstream purification and regulatory approval.
The Novel Approach
In stark contrast, the methodology disclosed in CN1438985A utilizes a highly efficient stereoselective conjugate addition strategy that bypasses the need for natural chiral pools. The core innovation lies in the use of readily available chiral enones, specifically (R)- or (S)-4-acetoxycyclopent-2-en-1-one, as the foundational scaffolds.  This approach allows for the direct introduction of carbon-based nucleophiles, such as organomagnesium or organozinc reagents, with exceptional stereocontrol dictated by the existing chiral center and the acetoxy leaving group. The process is modular, enabling the sequential addition of different R and R' groups (ranging from C1-C10 alkyl to cycloalkyl) to generate a vast library of 3,4-disubstituted intermediates. By eliminating the dependency on resolution steps and utilizing commodity chemicals for the carbon framework construction, this novel approach drastically simplifies the manufacturing workflow. It provides a direct route to both single substituted and disubstituted cyclopentanones, offering pharmaceutical manufacturers unprecedented flexibility in designing analog libraries while maintaining rigorous control over the stereochemical outcome, thereby ensuring consistent quality for cost reduction in API manufacturing.
This approach allows for the direct introduction of carbon-based nucleophiles, such as organomagnesium or organozinc reagents, with exceptional stereocontrol dictated by the existing chiral center and the acetoxy leaving group. The process is modular, enabling the sequential addition of different R and R' groups (ranging from C1-C10 alkyl to cycloalkyl) to generate a vast library of 3,4-disubstituted intermediates. By eliminating the dependency on resolution steps and utilizing commodity chemicals for the carbon framework construction, this novel approach drastically simplifies the manufacturing workflow. It provides a direct route to both single substituted and disubstituted cyclopentanones, offering pharmaceutical manufacturers unprecedented flexibility in designing analog libraries while maintaining rigorous control over the stereochemical outcome, thereby ensuring consistent quality for cost reduction in API manufacturing.
Mechanistic Insights into Stereoselective Conjugate Addition
The chemical elegance of this process is rooted in the precise manipulation of organometallic reactivity to achieve 1,4-addition selectivity. The reaction initiates with the conjugate addition of an organometallic nucleophile, typically generated in situ by mixing a Grignard reagent (e.g., n-propylmagnesium chloride) with dimethylzinc, to the chiral enone substrate. The presence of dimethylzinc is crucial as it moderates the reactivity of the Grignard reagent, suppressing competing 1,2-addition to the carbonyl group and favoring the desired conjugate addition to the beta-position. 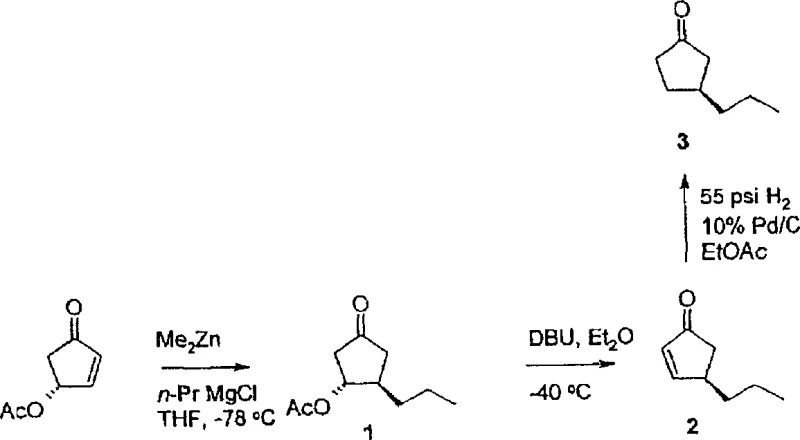 The acetoxy group at the 4-position plays a dual role: it acts as a directing group to influence the facial selectivity of the incoming nucleophile, ensuring the formation of the trans-3,4-disubstituted intermediate, and subsequently serves as a leaving group. Following the initial addition, the intermediate undergoes an elimination reaction mediated by a non-nucleophilic base such as 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU) at controlled low temperatures ranging from -40°C to -30°C. This elimination step regenerates the alpha,beta-unsaturated system, setting the stage for either catalytic hydrogenation to yield the saturated 3-substituted ketone or a second round of stereoselective conjugate addition to install a second substituent. The entire sequence is designed to minimize epimerization, preserving the optical integrity established by the starting enone.
The acetoxy group at the 4-position plays a dual role: it acts as a directing group to influence the facial selectivity of the incoming nucleophile, ensuring the formation of the trans-3,4-disubstituted intermediate, and subsequently serves as a leaving group. Following the initial addition, the intermediate undergoes an elimination reaction mediated by a non-nucleophilic base such as 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU) at controlled low temperatures ranging from -40°C to -30°C. This elimination step regenerates the alpha,beta-unsaturated system, setting the stage for either catalytic hydrogenation to yield the saturated 3-substituted ketone or a second round of stereoselective conjugate addition to install a second substituent. The entire sequence is designed to minimize epimerization, preserving the optical integrity established by the starting enone.
Impurity control is intrinsic to this mechanistic design. The use of chirally pure starting materials ensures that the resulting products possess high enantiomeric excess, typically exceeding 98% as confirmed by NMR analysis of derived chiral ketals. The patent details a robust verification method where the final ketones are reacted with (2R,3R)-(-)-2,3-butanediol to form diastereomeric ketals. In the event of any enantiomeric contamination, distinct peaks would appear in the NMR spectrum; however, experimental data confirms the absence of such peaks, validating the high fidelity of the stereochemical transfer. This level of purity is critical for pharmaceutical applications, as it reduces the burden on downstream purification processes like chromatography or crystallization, directly impacting the overall yield and environmental footprint of the synthesis. The ability to predict and control the stereochemistry at each step allows process chemists to design scalable routes with minimal risk of generating hard-to-remove stereoisomeric impurities.
How to Synthesize Enantiomerically Pure Cyclopentanones Efficiently
The practical implementation of this synthesis involves a series of well-defined unit operations that can be adapted for both laboratory and pilot-scale production. The process begins with the preparation of the organometallic reagent mixture under an inert atmosphere, followed by the controlled addition to the chiral enone at cryogenic temperatures to manage exotherms and ensure selectivity. Subsequent workup involves standard aqueous quenching and extraction protocols, while the elimination step requires careful temperature management to prevent side reactions.
- Perform conjugate addition of an organometallic nucleophile (e.g., Grignard reagent with dimethylzinc) to chiral 4-acetoxycyclopent-2-en-1-one at low temperature (-78°C) to generate trans-3,4-disubstituted intermediates.
- Execute an elimination reaction using a strong base like DBU at controlled temperatures (-40°C to -30°C) to remove the acetoxy leaving group and form the corresponding 4-alkyl-cyclopent-2-en-1-one.
- Finalize the synthesis either by catalytic hydrogenation to yield 3-substituted cyclopentanones or by a second stereoselective conjugate addition to produce 3,4-disubstituted derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers transformative benefits regarding cost stability and sourcing security. By shifting away from natural extraction or complex resolution processes, manufacturers can decouple their supply of critical intermediates from agricultural variability and biological constraints. The reliance on commodity chemicals such as Grignard reagents, dimethylzinc, and palladium on carbon means that raw material costs are predictable and subject to standard market fluctuations rather than the volatility of niche chiral pool suppliers. Furthermore, the streamlined nature of the synthesis, which combines carbon-carbon bond formation and stereocontrol in fewer steps, significantly reduces the total processing time and solvent consumption. This efficiency translates into substantial cost savings in API manufacturing, allowing companies to remain competitive in the generic and branded drug markets. The robustness of the chemistry also implies a lower risk of batch failures, ensuring a more reliable supply of high-purity pharmaceutical intermediates to meet global demand.
- Cost Reduction in Manufacturing: The elimination of expensive chiral resolution agents and the reduction in total synthetic steps directly lower the operational expenditure. By avoiding the need for multiple recrystallizations or preparative chiral HPLC to separate enantiomers, the process achieves a leaner manufacturing profile. The use of catalytic hydrogenation for the final saturation step is a cost-effective alternative to stoichiometric reducing agents, further driving down the cost of goods. Additionally, the high yields reported in the patent examples, such as 91% for the hydrogenation step and 68% for the conjugate addition, indicate a highly efficient use of starting materials, minimizing waste disposal costs and maximizing throughput per reactor volume.
- Enhanced Supply Chain Reliability: Sourcing chiral intermediates from natural sources often involves long lead times and geopolitical risks associated with raw material harvesting. This synthetic route utilizes widely available petrochemical-derived starting materials, ensuring a stable and continuous supply chain. The ability to synthesize both (R) and (S) enantiomers from commercially available chiral enones means that manufacturers are not held hostage by the availability of a single natural enantiomer. This flexibility allows for rapid response to market demands for different stereoisomers, reducing lead time for high-purity pharmaceutical intermediates and preventing production stoppages due to raw material shortages. The scalability of the reaction conditions, which do not require exotic equipment beyond standard low-temperature reactors, further enhances supply security.
- Scalability and Environmental Compliance: The process is designed with scale-up in mind, utilizing reagents and conditions that are compatible with large-scale stainless steel reactors. The avoidance of heavy metal catalysts in the carbon-carbon bond forming steps (relying instead on zinc and magnesium) simplifies the removal of metal residues, a critical factor for meeting stringent regulatory limits on elemental impurities in drug substances. The waste stream is primarily composed of organic solvents and salts, which can be managed through standard recovery and treatment protocols. The high atom economy of the conjugate addition and the catalytic nature of the hydrogenation step contribute to a greener manufacturing process, aligning with modern sustainability goals and reducing the environmental compliance burden for chemical producers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and claims within CN1438985A, providing a clear understanding of the process capabilities and limitations.
Q: What is the primary advantage of this synthesis route over natural extraction?
A: Unlike natural sources which are limited in availability and stereoisomer diversity, this synthetic route allows for the production of specific (R) or (S) enantiomers and diverse alkyl substitutions (C1-C10) that are otherwise inaccessible or prohibitively expensive to isolate from nature.
Q: How is enantiomeric purity ensured in this process?
A: The process utilizes chirally pure starting materials ((R)- or (S)-4-acetoxycyclopent-2-en-1-one) where the acetoxy group directs the stereochemistry of the incoming nucleophile. Purity is verified by converting the product into diastereomeric ketals using (2R,3R)-2,3-butanediol and analyzing via NMR, confirming purity levels exceeding 98%.
Q: Can this method be scaled for industrial API production?
A: Yes, the methodology relies on standard organometallic reagents (Grignard, organozinc) and common catalysts (Pd/C) without requiring exotic transition metals or ultra-low cryogenic conditions below -78°C for extended periods, making it highly amenable to commercial scale-up in standard stainless steel reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gabapentin Analog Intermediates Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic pathways in the development of life-saving neurological medications. Our team of expert process chemists has extensively evaluated the technology described in CN1438985A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with state-of-the-art facilities capable of handling sensitive organometallic reactions and cryogenic processes, ensuring that every batch of chiral cyclopentanones meets stringent purity specifications. Our rigorous QC labs employ advanced analytical techniques, including chiral HPLC and NMR, to verify the enantiomeric excess and chemical identity of every intermediate, guaranteeing the quality required for GMP manufacturing.
We invite pharmaceutical partners to collaborate with us to leverage this advanced synthesis for their pipeline projects. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a sustainable and cost-effective supply of these vital building blocks, accelerating your journey from discovery to commercial launch.
