Scalable Synthesis of High-Purity Carotenoid Intermediates via Selective Sulfone Chemistry
The global demand for natural carotenoids, including beta-carotene, lycopene, and astaxanthin, continues to surge across the pharmaceutical and nutritional supplement sectors due to their critical role as Vitamin A precursors and potent antioxidants. Patent CN1264815C introduces a groundbreaking methodology for synthesizing the essential C5 and C10 building blocks required for these complex molecules via the Julia reaction. Unlike traditional extraction methods which are limited by low yields from plant sources, this chemical synthesis approach offers a robust pathway for consistent supply. The innovation lies in the preparation of 4-chloro-3-methyl-2-butenylphenyl sulfide and its corresponding sulfone derivatives, which serve as stable intermediates for chain extension. 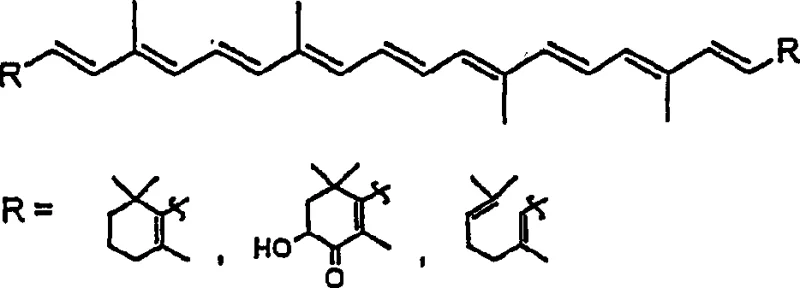 This technology addresses the longstanding challenges of stereochemical control and purification efficiency, positioning it as a vital asset for any reliable carotenoid intermediate supplier seeking to optimize their production capabilities for high-value fine chemicals.
This technology addresses the longstanding challenges of stereochemical control and purification efficiency, positioning it as a vital asset for any reliable carotenoid intermediate supplier seeking to optimize their production capabilities for high-value fine chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the organic synthesis of carotenoid compounds has relied on methods such as the Roche acetylide approach or the BASF Wittig reaction, both of which present significant manufacturing hurdles. The Roche method often results in the formation of cis-double bonds which possess lower biochemical activity, necessitating complex isomerization steps to achieve the desired all-trans configuration. Furthermore, the Wittig reaction, while effective for double bond formation, involves the preparation of polyene-containing phosphonium salts that are notoriously difficult to handle and purify on a large scale. Conventional sulfone-based routes also suffer from inefficiencies, particularly during the ring-opening of isoprene monoxide where substantial amounts of phenyl disulfide by-products are generated. These impurities typically require additional silica gel column chromatography for removal, a process that is cost-prohibitive and environmentally burdensome in an industrial setting, severely impacting cost reduction in fine chemical manufacturing.
The Novel Approach
The methodology disclosed in the patent data revolutionizes this landscape by introducing a streamlined synthesis route starting directly from isoprene and N-chlorosuccinimide (NCS) without the need for polar solvents. This novel approach facilitates the formation of chlorohydrin intermediates with high yield and purity, effectively bypassing the need for extensive chromatographic purification in the early stages. By utilizing specific copper(I) salt catalysts during the halogenation step, the process achieves exceptional trans-selectivity, with trans-to-cis ratios exceeding 8:1, thereby minimizing downstream isomerization requirements. The coupling reactions with benzenethiol or sodium sulfide are optimized to selectively leave specific halogen substituents, ensuring the structural integrity of the C5 and C10 units. This strategic redesign of the synthetic pathway not only simplifies the operational workflow but also significantly enhances the overall preparation efficiency, making it an ideal candidate for the commercial scale-up of complex carotenoid intermediates.
Mechanistic Insights into Selective Sulfide Oxidation
A critical bottleneck in the synthesis of carotenoid precursors via the Julia reaction is the selective oxidation of the thioether moiety to a sulfone group without compromising the sensitive polyene chain. The patent details a sophisticated catalytic system employing metal oxides such as vanadium pentoxide (V2O5) or sodium metavanadate (NaVO3) in conjunction with hydrogen peroxide. This catalytic environment ensures that the oxidation is strictly confined to the sulfur atom, preventing the unwanted epoxidation or cleavage of the adjacent carbon-carbon double bonds. 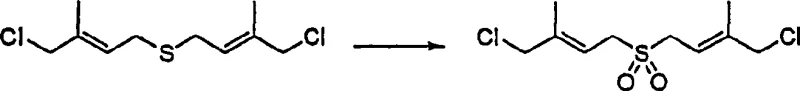 The ability to perform this transformation on the simpler C10 sulfide structure prior to the final coupling step is a masterstroke in process chemistry, as it avoids the non-selective oxidation issues that plague the larger C40 coupled products. This mechanistic precision is paramount for maintaining the high-purity carotenoid precursors required by regulatory standards in the food and pharma industries.
The ability to perform this transformation on the simpler C10 sulfide structure prior to the final coupling step is a masterstroke in process chemistry, as it avoids the non-selective oxidation issues that plague the larger C40 coupled products. This mechanistic precision is paramount for maintaining the high-purity carotenoid precursors required by regulatory standards in the food and pharma industries.
Furthermore, the impurity control mechanism is deeply embedded in the physical properties of the resulting sulfone compounds. Unlike the corresponding sulfides which are often oils requiring chromatography, the trans-isomer of the bis(4-chloro-3-methyl-2-butenyl) sulfone can be isolated through simple recrystallization from methanol at low temperatures. This phase change from oil to solid crystal allows for the mechanical separation of the desired trans-isomer from cis-impurities and other reaction by-products. The process leverages the stability of the allylic chloride and double bonds under the specific oxidative conditions, ensuring that the final intermediate possesses the rigorous stereochemical definition needed for the subsequent Ramberg-Böcklund elimination. This level of control over the impurity profile is what distinguishes a laboratory curiosity from a viable industrial process for reducing lead time for high-purity carotenoid intermediates.
How to Synthesize Bis(4-chloro-3-methyl-2-butenyl) Sulfone Efficiently
The synthesis of these critical C10 units begins with the chlorohydroxylation of isoprene, followed by a copper-catalyzed allylic rearrangement to install the necessary halogen handles with high stereoselectivity. The subsequent coupling with sodium sulfide and selective oxidation forms the core of the process, allowing for the accumulation of material suitable for final assembly. Detailed standardized synthesis steps see the guide below. This sequence is designed to maximize throughput while minimizing waste, aligning with modern green chemistry principles.
- React isoprene with N-chlorosuccinimide (NCS) in water to form chlorohydrin intermediates without polar solvents.
- Perform allylic rearrangement and halogenation using copper(I) catalysts to achieve high trans-selectivity.
- Execute selective oxidation of the sulfide to sulfone using metal oxide catalysts and hydrogen peroxide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers tangible benefits that extend beyond mere technical feasibility. The elimination of silica gel column chromatography for the purification of key intermediates represents a drastic simplification of the manufacturing workflow, directly translating to reduced operational expenditures and faster batch turnover. By relying on readily available bulk commodities like isoprene and avoiding exotic or unstable reagents, the supply chain becomes more resilient to market fluctuations and raw material shortages. The ability to purify the final sulfone intermediate via crystallization rather than chromatography further reduces solvent consumption and waste disposal costs, contributing to substantial cost savings and enhanced environmental compliance. This process stability ensures that production schedules can be met consistently, addressing the critical need for supply continuity in the competitive nutraceutical market.
- Cost Reduction in Manufacturing: The process logic dictates significant economic advantages by removing the most expensive purification step, silica gel chromatography, from the production of the C5 and C10 building blocks. The use of inexpensive oxidants like hydrogen peroxide and recoverable metal oxide catalysts further drives down the variable cost per kilogram. Additionally, the high trans-selectivity achieved during the halogenation step reduces the loss of material associated with isomer correction, ensuring that a greater proportion of the input raw materials are converted into saleable product. These factors combine to create a leaner manufacturing model that supports aggressive pricing strategies without compromising margin integrity.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved as the primary feedstock, isoprene, is a widely produced petrochemical derivative with a mature global supply network. The synthetic route avoids reliance on specialized reagents that might have single-source suppliers or long lead times, thereby mitigating the risk of production stoppages. The robustness of the reaction conditions, which tolerate slight variations in temperature and stoichiometry without catastrophic failure, ensures high first-pass yields and predictable output volumes. This reliability is crucial for partners who require just-in-time delivery of high-quality intermediates to maintain their own downstream production schedules.
- Scalability and Environmental Compliance: The transition from chromatographic purification to crystallization is a key enabler for scaling this chemistry from pilot plants to multi-ton reactors. Crystallization is inherently easier to control and automate in large vessels compared to column chromatography, facilitating the commercial scale-up of complex sulfone intermediates. Moreover, the reduction in organic solvent usage and the avoidance of silica waste align with increasingly stringent environmental regulations regarding hazardous waste disposal. The process generates fewer by-products that require complex treatment, simplifying the effluent management strategy and reducing the overall environmental footprint of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this sulfone-based synthesis technology. These answers are derived directly from the experimental data and process descriptions found within the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the fit of this technology within their existing R&D pipelines and supply chain frameworks. We encourage technical teams to review these points when assessing process viability.
Q: How does this method improve purity compared to conventional Wittig reactions?
A: This method utilizes stable sulfone compounds and avoids the difficult handling of polyene-containing Wittig salts, resulting in easier by-product removal and higher stereochemical control.
Q: Can the sulfone oxidation be controlled without affecting double bonds?
A: Yes, by using specific metal oxide catalysts like NaVO3 with hydrogen peroxide, the thioether moiety is selectively oxidized to a sulfone while leaving the sensitive conjugated double bonds intact.
Q: Is this process suitable for large-scale industrial production?
A: The process eliminates the need for silica gel column chromatography for key intermediates and allows for purification via crystallization, making it highly viable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bis(4-chloro-3-methyl-2-butenyl) Sulfone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic pathways in the production of high-value carotenoids. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent can be realized in a practical manufacturing environment. We maintain stringent purity specifications and operate rigorous QC labs to verify that every batch of intermediate meets the exacting standards required for pharmaceutical and food-grade applications. Our commitment to quality assurance means that clients can trust our materials to perform consistently in their downstream Julia coupling reactions.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic impact of switching to this more efficient methodology. We are prepared to provide specific COA data and route feasibility assessments to support your internal validation processes, ensuring a smooth transition to a more reliable and cost-effective sourcing strategy for your carotenoid production needs.
