Industrial Scale-Up of 1,2-cis-2-fluorocyclopropane-1-carboxylic Ester via Lewis Acid Catalysis
The pharmaceutical and agrochemical industries constantly seek robust synthetic routes for key building blocks, particularly fluorinated cyclopropanes which serve as critical scaffolds for next-generation quinolone antibiotics. Patent CN1930112A discloses a groundbreaking methodology for the production of 1,2-cis-2-fluorocyclopropane-1-carboxylic ester compounds, addressing long-standing inefficiencies in reductive dehalogenation processes. This innovation replaces problematic conventional solvents with a sophisticated system utilizing aprotic polar solvents and specific Lewis acid catalysts to achieve superior stereocontrol. The technical breakthrough lies in the ability to convert halogenated precursors into the desired cis-isomer with exceptional speed and purity, fundamentally altering the economic landscape for reliable pharmaceutical intermediate supplier networks. By mitigating the formation of toxic byproducts and resolving mixing limitations, this patent provides a blueprint for sustainable and high-efficiency manufacturing.
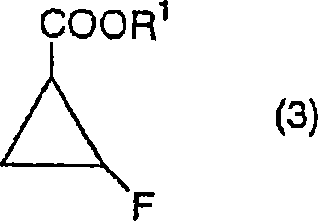
Traditional synthetic approaches for constructing the 1,2-cis-2-fluorocyclopropyl moiety have historically relied on dechlorination reactions in dimethyl sulfoxide (DMSO) using sodium borohydride. While chemically feasible, these legacy methods suffer from severe engineering drawbacks that hinder commercial scale-up of complex polymer additives and pharmaceutical intermediates alike. As the reaction progresses in DMSO, the viscosity of the reaction mass increases dramatically, leading to poor stirring efficiency and heterogeneous heat transfer. This physical limitation necessitates reaction times extending over several days to reach completion, creating a bottleneck in production schedules. Furthermore, the use of DMSO in reductive environments often generates dimethyl sulfide as a malodorous byproduct, imposing strict environmental controls and increasing operational costs for waste management. These factors collectively degrade the overall process economics and pose significant safety challenges for plant operators.
In stark contrast, the novel approach detailed in the patent utilizes a synergistic combination of a metal borohydride reducing agent, a catalytic amount of a Lewis acid, and an aprotic polar solvent such as N,N-dimethylacetamide (DMAc) or N-methyl-2-pyrrolidone (NMP). This triad of reagents creates a homogeneous reaction environment that maintains low viscosity throughout the transformation, ensuring consistent mixing and thermal control. Experimental data from the patent demonstrates that this system can reduce reaction times from days to merely a few hours while achieving yields as high as 98% to 100%. The inclusion of Lewis acids like cobalt chloride or indium chloride is pivotal, as they activate the carbon-halogen bond without promoting unwanted side reactions. This methodological shift not only enhances throughput but also eliminates the foul-smelling sulfur byproducts, representing a significant advancement in cost reduction in API manufacturing by simplifying downstream processing and odor control requirements.
Mechanistic Insights into Lewis Acid-Catalyzed Reductive Dehalogenation
The core mechanistic advantage of this process stems from the specific interaction between the Lewis acid catalyst and the halogenated cyclopropane substrate. In the absence of a Lewis acid, the reduction of the carbon-chlorine or carbon-bromine bond by borohydride species is kinetically sluggish and non-selective. The Lewis acid, acting as a soft electrophile, coordinates with the halogen atom or the carbonyl oxygen, thereby lowering the activation energy for the hydride transfer. This coordination facilitates a concerted or stepwise elimination of the halide ion, preserving the delicate stereochemistry of the cyclopropane ring. The patent highlights that different Lewis acids exert varying degrees of influence on the cis/trans selectivity, with cobalt and indium salts showing particular efficacy in favoring the thermodynamically stable cis-isomer. This precise control over the reaction pathway is essential for producing high-purity OLED material precursors and pharmaceutical intermediates where isomeric purity dictates biological activity.
Furthermore, the choice of solvent plays a dual role as both a reaction medium and a participant in stabilizing the transition state. Aprotic polar solvents possess high dielectric constants that stabilize the ionic intermediates formed during the dehalogenation, preventing the collapse of the cyclopropane ring which is prone to strain-induced opening. Comparative examples in the patent reveal that switching to protic solvents like ethanol or non-polar solvents like toluene results in negligible yields or complete reaction failure. This underscores the necessity of the specific solvation shell provided by amides or cyclic ureas to support the catalytic cycle. Additionally, the solvent system effectively suppresses the formation of radical species that could lead to polymerization or dimerization impurities. By maintaining a clean reaction profile, the process minimizes the burden on purification steps, directly contributing to reducing lead time for high-purity pharmaceutical intermediates and ensuring a consistent supply of quality material for downstream drug synthesis.
How to Synthesize 1,2-cis-2-fluorocyclopropane-1-carboxylic Ester Efficiently
The implementation of this synthetic route requires careful attention to reagent addition order and temperature control to maximize the benefits of the Lewis acid catalysis. The general protocol involves dissolving the reducing agent in the chosen aprotic polar solvent, followed by the introduction of the halogenated precursor. The Lewis acid is typically added last, often as a solution, to initiate the rapid dehalogenation. Maintaining the temperature between 0°C and 60°C is critical; too low a temperature slows the kinetics, while excessive heat may compromise stereoselectivity. The detailed standardized synthesis steps see the guide below for specific molar ratios and workup procedures tailored to different substrate scales.
- Dissolve a metal borohydride reducing agent, such as sodium borohydride, in an aprotic polar solvent like NMP or DMAc under stirring.
- Add the halogenated fluoro-cyclopropane precursor (Formula 1) to the reaction mixture, followed by the slow addition of a catalytic Lewis acid solution.
- Maintain the reaction temperature between 0°C and 60°C for a short duration, then perform aqueous workup and extraction to isolate the high-purity cis-ester product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology offers tangible strategic benefits beyond simple chemical yield improvements. The primary advantage lies in the drastic simplification of the manufacturing workflow, which translates directly into operational cost savings. By eliminating the need for DMSO and the associated handling of malodorous dimethyl sulfide, facilities can avoid the capital expenditure required for specialized scrubbing systems and extensive ventilation upgrades. This reduction in environmental compliance complexity allows for faster permitting and more flexible site selection for production units. Moreover, the significant reduction in reaction time from multiple days to mere hours increases the asset turnover rate of existing reactor vessels, effectively expanding production capacity without the need for new hardware investments. This efficiency gain is a powerful driver for cost reduction in electronic chemical manufacturing and pharma sectors alike.
- Cost Reduction in Manufacturing: The elimination of expensive and difficult-to-remove solvents like DMSO, combined with the use of catalytic amounts of relatively inexpensive Lewis acids, lowers the raw material cost per kilogram. The high selectivity of the reaction minimizes the loss of valuable starting materials to byproducts, ensuring that a greater proportion of input costs are converted into saleable product. Furthermore, the simplified workup procedure, which often involves straightforward aqueous extraction rather than complex chromatographic separations, reduces solvent consumption and waste disposal fees. These cumulative effects result in a leaner cost structure that enhances competitiveness in the global market for fine chemicals.
- Enhanced Supply Chain Reliability: The robustness of the new solvent system, utilizing widely available industrial solvents like DMAc and NMP, mitigates the risk of supply disruptions associated with specialty reagents. The process tolerance to variations in reaction conditions ensures consistent batch-to-batch quality, reducing the incidence of out-of-specification materials that can delay shipments. By shortening the production cycle time, manufacturers can respond more agilely to fluctuations in demand, maintaining optimal inventory levels and preventing stockouts. This reliability is crucial for maintaining the continuity of supply chains for critical pharmaceutical intermediates where delays can impact the launch of life-saving medications.
- Scalability and Environmental Compliance: The low viscosity of the reaction mixture ensures that heat and mass transfer remain efficient even at large scales, addressing a common failure point in scaling up exothermic reductions. This physical property allows for safe operation in large-volume reactors without the risk of thermal runaway or mixing dead zones. Additionally, the absence of sulfur-based byproducts aligns with increasingly stringent global environmental regulations regarding volatile organic compounds and odor emissions. This alignment future-proofs the manufacturing process against regulatory tightening, ensuring long-term operational viability and reducing the risk of shutdowns due to non-compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Lewis acid-catalyzed dehalogenation process. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering a clear understanding of the process boundaries and optimization opportunities. Understanding these nuances is vital for R&D teams planning technology transfer and for procurement specialists evaluating supplier capabilities.
Q: Why is the choice of solvent critical in this dehalogenation reaction?
A: According to patent CN1930112A, aprotic polar solvents like DMAc or NMP are essential because they prevent the formation of viscous reaction masses seen in DMSO and avoid the generation of malodorous dimethyl sulfide byproducts, ensuring high yields and selectivity.
Q: What role does the Lewis acid play in improving cis-selectivity?
A: The Lewis acid, such as cobalt chloride or indium chloride, activates the carbon-halogen bond for reduction. Comparative data indicates that without the Lewis acid, yields drop significantly (e.g., from 98% to 61%), and the reaction time extends drastically, compromising the cis/trans stereochemical ratio.
Q: Can this process be scaled for commercial API production?
A: Yes, the process is designed for industrial applicability. By eliminating the viscosity issues associated with traditional DMSO methods, the new protocol allows for efficient stirring and heat transfer in large reactors, facilitating the commercial scale-up of complex fluorinated cyclopropanes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2-cis-2-fluorocyclopropane-1-carboxylic Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of advanced therapeutic agents. Our technical team has extensively analyzed the methodologies described in patent CN1930112A and possesses the expertise to adapt this chemistry for diverse substrate scopes. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to pilot plant is seamless and efficient. Our facility is equipped with rigorous QC labs and stringent purity specifications to guarantee that every batch of 1,2-cis-2-fluorocyclopropane-1-carboxylic ester meets the exacting standards required for GMP manufacturing. We are committed to delivering not just a chemical product, but a reliable partnership that supports your long-term development goals.
We invite you to engage with our technical procurement team to discuss how this optimized synthetic route can benefit your specific project requirements. By leveraging our process optimization capabilities, we can provide a Customized Cost-Saving Analysis tailored to your volume needs and purity targets. Please contact us to request specific COA data and route feasibility assessments for your upcoming campaigns. Let us help you secure a stable and cost-effective supply chain for your most critical fluorinated building blocks.
