Advanced Cerium-Catalyzed Synthesis of Tetrahydrofuran-3-ones for Industrial Scale Production
Advanced Cerium-Catalyzed Synthesis of Tetrahydrofuran-3-ones for Industrial Scale Production
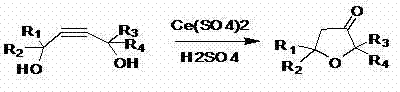
The landscape of fine chemical manufacturing is undergoing a significant transformation driven by the urgent need for greener, more efficient synthetic pathways. A pivotal development in this domain is documented in Chinese Patent CN102321054B, which discloses a robust preparation method for tetrahydrofuran-3-one compounds. This technology represents a major leap forward for manufacturers seeking a reliable tetrahydrofuran-3-one supplier, as it replaces hazardous traditional reagents with a benign cerium-based catalytic system. By utilizing 2-butyne-1,4-diol derivatives as starting materials and employing ceric sulfate in an aqueous sulfuric acid medium, the process achieves high conversion rates through a concerted dehydration and hydration mechanism. The implications for the global supply chain are profound, offering a route that is not only environmentally friendly but also economically superior due to the recyclability of the catalyst system. For R&D directors and procurement managers alike, this patent offers a blueprint for cost reduction in pharmaceutical intermediate manufacturing that aligns perfectly with modern sustainability goals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of tetrahydrofuran-3-one scaffolds has been plagued by severe safety and efficiency bottlenecks that hinder large-scale commercial adoption. Prior art, such as the method described in Japanese Patent JP2006206463, relies heavily on the use of potassium cyanide, a substance known for its extreme toxicity and the stringent regulatory burdens associated with its handling and disposal. Furthermore, alternative routes like those found in Chinese Patent CN101712664A necessitate the use of metallic sodium and highly flammable ethers, creating significant explosion hazards and requiring expensive inert atmosphere conditions. Another approach cited in US Patent No. 4464409 utilizes potassium gluconate but suffers from abysmally low yields, often falling below 25%, which renders it completely unsuitable for industrial application. These legacy methods collectively contribute to high production costs, complex multi-step sequences, and substantial environmental pollution, creating a critical need for innovation in this sector.
The Novel Approach
In stark contrast to these hazardous legacy protocols, the novel approach detailed in CN102321054B introduces a streamlined, one-pot transformation that dramatically simplifies the operational workflow. By leveraging the unique oxidative and acidic properties of ceric sulfate in conjunction with sulfuric acid, the process effectively converts readily available 2-butyne-1,4-diols directly into the desired furanone rings. This methodology eliminates the need for toxic cyanides or pyrophoric metals, thereby removing the most dangerous steps from the manufacturing line. The reaction conditions are remarkably mild, operating effectively between 50°C and 90°C, which reduces energy consumption compared to high-temperature reflux methods. Moreover, the simplicity of the workup procedure, which often involves mere phase separation and distillation, allows for rapid turnover and higher throughput. This shift towards a safer, more direct synthesis pathway is essential for any organization aiming to secure a stable supply of high-purity fine chemical intermediates.
Mechanistic Insights into Cerium-Catalyzed Cyclization
The core of this technological breakthrough lies in the dual functionality of the ceric sulfate and sulfuric acid system, which facilitates a complex cascade of dehydration and hydration reactions. The mechanism initiates with the activation of the alkyne moiety within the 2-butyne-1,4-diol substrate by the acidic medium, promoting a hydration event that generates an enol or ketone intermediate. Simultaneously, the hydroxyl groups undergo acid-catalyzed dehydration, driving the intramolecular cyclization that forms the tetrahydrofuran ring structure. The presence of Cerium(IV) acts as a potent Lewis acid and oxidant, stabilizing transition states and ensuring that the reaction proceeds with high regioselectivity towards the 3-ketone product. This precise control over the reaction trajectory is what allows the process to avoid the formation of complex polymeric byproducts that typically plague alkyne hydration reactions in strong acids.
Furthermore, the mechanistic pathway inherently supports excellent impurity control, a critical factor for R&D directors focused on product quality. Because the reaction occurs in a homogeneous aqueous phase with a highly selective catalyst, side reactions such as over-oxidation or random polymerization are minimized. The patent data indicates that by maintaining the sulfuric acid mass fraction between 1% and 20%, the system avoids the harsh conditions that lead to charring or degradation of the sensitive furanone ring. Additionally, the ability to tune the molar ratio of the diol to the cerium catalyst allows operators to balance reaction speed against selectivity, ensuring that the final crude product requires minimal purification. This level of mechanistic understanding translates directly into a more robust manufacturing process capable of delivering consistent batch-to-batch quality.
How to Synthesize Tetrahydrofuran-3-one Efficiently
Implementing this synthesis route in a commercial setting requires careful attention to the specific parameters outlined in the patent to maximize yield and catalyst longevity. The process begins with the preparation of the catalytic solution, where ceric sulfate is dissolved in dilute sulfuric acid, followed by the controlled addition of the alkyne-diol substrate. Operators must maintain the reaction temperature within the optimal window of 50°C to 90°C for a duration of 2 to 5 hours to ensure complete conversion without thermal degradation. Following the reaction, the mixture is cooled, allowing the organic product to separate from the aqueous catalyst phase, which can then be recovered for reuse.
- Prepare the reaction medium by mixing aqueous sulfuric acid with ceric sulfate catalyst, ensuring the acid concentration is between 1% and 20% for optimal activity.
- Introduce the 2-butyne-1,4-diol substrate into the acidic mixture and heat the system to a temperature range of 50°C to 90°C to initiate the cascade dehydration and hydration reactions.
- Upon completion, separate the organic crude product layer from the aqueous catalyst layer, which can be recycled for subsequent batches to minimize waste and cost.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this cerium-catalyzed technology offers tangible strategic benefits that extend far beyond simple chemical transformation. The most significant advantage is the drastic simplification of the supply chain regarding raw material safety and handling. By eliminating the need for regulated substances like potassium cyanide or sodium metal, facilities can reduce insurance premiums, lower compliance costs, and minimize the risk of production stoppages due to safety audits. Furthermore, the ability to recycle the aqueous catalyst layer, as demonstrated in the patent embodiments, means that the consumption of cerium salts and sulfuric acid is significantly reduced over time. This closed-loop aspect of the process directly contributes to substantial cost savings in raw material procurement and waste disposal fees, making the overall cost of goods sold much more competitive in the global market.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the elimination of expensive and hazardous reagents that require specialized storage and disposal protocols. Traditional methods involving cyanides or metallic sodium incur high hidden costs related to safety infrastructure, neutralization of toxic waste, and regulatory reporting. In contrast, the cerium sulfate system utilizes relatively inexpensive inorganic salts that can be recovered and reused multiple times without significant loss of activity. This recyclability drastically lowers the variable cost per kilogram of product, allowing manufacturers to offer more competitive pricing to their clients while maintaining healthy margins. Additionally, the high yields reported in the patent examples mean that less raw material is wasted, further enhancing the overall economic efficiency of the production line.
- Enhanced Supply Chain Reliability: From a logistics perspective, relying on common, non-hazardous starting materials like 2-butyne-1,4-diols ensures a more resilient supply chain. Unlike specialty reagents that may be subject to export controls or limited vendor availability, the inputs for this process are commodity chemicals available from multiple global sources. This diversification of supply reduces the risk of bottlenecks and ensures continuous production even during market fluctuations. Moreover, the simplified reaction protocol reduces the dependency on highly skilled labor for dangerous operations, making it easier to scale up production capacity rapidly to meet surging demand. This reliability is crucial for pharmaceutical customers who require uninterrupted delivery of critical intermediates to maintain their own drug manufacturing schedules.
- Scalability and Environmental Compliance: The environmental profile of this method aligns perfectly with increasingly strict global regulations on industrial emissions and waste discharge. The process generates minimal three wastes, and the aqueous waste stream is primarily composed of dilute acid and metal salts that are easier to treat than organic solvent-heavy waste from traditional methods. The near-zero discharge capability mentioned in the patent highlights the potential for this technology to be implemented in regions with rigorous environmental standards. Scalability is further supported by the fact that the reaction does not require extreme pressures or temperatures, allowing it to be run in standard glass-lined or stainless steel reactors commonly found in fine chemical plants. This ease of scale-up ensures that the transition from pilot plant to commercial tonnage production is smooth and predictable.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These answers are derived directly from the experimental data and claims presented in the patent documentation to provide clarity on process capabilities. Understanding these details is essential for partners evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary safety advantages of this cerium-catalyzed method over traditional routes?
A: Unlike conventional methods that rely on highly toxic potassium cyanide or hazardous sodium metal, this process utilizes ceric sulfate in dilute sulfuric acid, significantly reducing operational risks and eliminating the need for specialized hazardous waste disposal infrastructure.
Q: Can the catalyst system be reused to lower production costs?
A: Yes, the patent explicitly demonstrates that the aqueous layer containing the sulfuric acid and ceric sulfate can be separated and reused for multiple reaction cycles with minimal loss in catalytic efficiency, driving down raw material consumption.
Q: What purity levels can be achieved with this synthesis route?
A: Experimental data indicates that the crude products obtained via distillation consistently achieve purity levels exceeding 98%, with specific examples showing GC analysis content up to 99.4%, reducing the burden on downstream purification processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydrofuran-3-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the cerium-catalyzed synthesis route for producing high-value heterocyclic intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practice. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of tetrahydrofuran-3-one delivered meets the highest industry standards. We are committed to leveraging this green chemistry innovation to provide our clients with a sustainable and cost-effective supply solution that supports their long-term growth objectives.
We invite potential partners to engage with our technical procurement team to discuss how this advanced manufacturing method can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this catalytic system for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and profitability in your operations.
