Optimized Synthesis of 2-Chloro-5-Nitrophenyl-4-Methoxybenzophenone for Scalable Pharmaceutical Manufacturing
Optimized Synthesis of 2-Chloro-5-Nitrophenyl-4-Methoxybenzophenone for Scalable Pharmaceutical Manufacturing
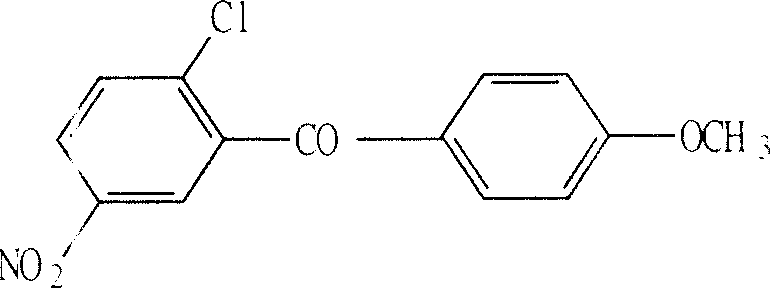
The pharmaceutical industry constantly seeks robust synthetic routes for critical intermediates that balance high purity with operational simplicity. Patent CN100386305C introduces a significant advancement in the preparation of 2-chloro-5-nitrophenyl-4'-methoxybenzophenone, a vital precursor for liver mitochondrial enzyme inhibitors and antipyretics. This technology leverages a refined Friedel-Crafts acylation strategy that addresses historical challenges regarding yield and isomer separation. By utilizing 2-chloro-5-nitrobenzoyl chloride and anisole under strictly controlled low-temperature conditions with anhydrous aluminum trichloride, the process achieves a purity exceeding 98% and a yield surpassing 58%. For R&D directors and procurement specialists, this represents a shift towards more predictable and efficient manufacturing protocols. The elimination of cumbersome batch-wise catalyst addition not only streamlines the workflow but also enhances the safety profile of the reaction, making it an attractive candidate for reliable pharmaceutical intermediate supplier partnerships aiming for consistent quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those disclosed in WO 03-0228042, relied on reacting 2-chloro-5-nitrobenzoic acid with phosphorus pentachloride followed by acylation. A critical bottleneck in these legacy processes was the requirement for batch-wise addition of the aluminum trichloride catalyst. This incremental approach made precise temperature regulation exceptionally difficult, often leading to thermal runaways or insufficient activation. Consequently, the reaction mixture would generate a high content of unwanted ortho-position isomers alongside the desired para-product. This lack of selectivity necessitated multiple recrystallization steps to achieve acceptable purity levels, drastically reducing the overall yield to approximately 41%. For supply chain heads, such inefficiencies translate into higher raw material consumption, increased waste generation, and prolonged production cycles, rendering the conventional route economically unviable for cost reduction in pharma intermediates manufacturing.
The Novel Approach
The innovative methodology described in CN100386305C fundamentally reengineers the acylation step to overcome these kinetic and thermodynamic barriers. Instead of分批 adding the catalyst, the novel protocol dictates a single,一次性 addition of anhydrous aluminum trichloride into the reaction mixture maintained at a cryogenic range of -20°C to -5°C. This precise thermal window suppresses the formation of ortho-substituted byproducts, thereby maximizing the selectivity for the target 4'-methoxybenzophenone derivative. The result is a dramatic improvement in process efficiency, where the crude product already possesses high purity, requiring only a single recrystallization to reach specifications greater than 98%. This streamlined approach not only boosts the yield to over 58% but also simplifies the downstream processing, offering a clear pathway for the commercial scale-up of complex pharmaceutical intermediates without the burden of repetitive purification loops.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The core of this synthesis relies on a classic yet meticulously optimized Friedel-Crafts acylation mechanism. In this electrophilic aromatic substitution, the 2-chloro-5-nitrobenzoyl chloride acts as the acylating agent, activated by the Lewis acid catalyst, anhydrous aluminum trichloride. The interaction between the carbonyl oxygen of the acid chloride and the aluminum center generates a highly reactive acylium ion complex. This electrophile then attacks the electron-rich aromatic ring of the anisole substrate. The methoxy group on the anisole ring serves as a strong ortho/para-directing activator; however, steric hindrance at the ortho positions favors attack at the para position. The innovation lies in the kinetic control exerted by the low temperature (-20°C to -5°C), which ensures that the thermodynamic product (the para-isomer) is formed exclusively while minimizing the kinetic trap of ortho-substitution.
Furthermore, the choice of solvent plays a pivotal role in stabilizing the transition states and managing the exothermic nature of the complexation. The patent specifies the use of halogenated hydrocarbons, with dichloromethane being the preferred medium due to its excellent solubility profile and low freezing point, which is essential for maintaining the sub-zero reaction conditions. Following the acylation, the reaction mixture undergoes hydrolysis with hydrochloric acid to decompose the aluminum complex and liberate the free ketone. This step is critical for breaking the strong coordination bond between the product carbonyl and the aluminum species. The subsequent workup involves simple phase separation and solvent removal, followed by recrystallization from an ethanol and ethyl acetate mixture. This specific solvent system is chosen to maximize the solubility difference between the target molecule and any remaining impurities, ensuring the final high-purity specification required for high-purity OLED material or pharmaceutical applications.
How to Synthesize 2-Chloro-5-Nitrophenyl-4'-Methoxybenzophenone Efficiently
Implementing this synthesis requires strict adherence to the molar ratios and thermal parameters defined in the patent to ensure reproducibility and safety. The process begins with the preparation of the reaction vessel, typically a four-necked flask equipped with efficient stirring and cooling capabilities to handle the exotherm upon catalyst addition. The stoichiometry is precise, utilizing a molar ratio of 2-chloro-5-nitrobenzoyl chloride to anisole to aluminum trichloride of approximately 1.0:1.6-2.0:1.4-1.5. This excess of anisole helps drive the reaction to completion while the specific catalyst loading ensures full activation without excessive waste. The detailed standardized synthesis steps, including specific addition rates and quenching procedures, are outlined below to guide process engineers in replicating this high-yield route.
- Combine 2-chloro-5-nitrobenzoyl chloride and anisole in a halogenated hydrocarbon solvent such as dichloromethane.
- Cool the reaction mixture to between -20°C and -5°C and add anhydrous aluminum trichloride in a single portion.
- Maintain temperature for 5-15 hours, hydrolyze with hydrochloric acid, and purify via recrystallization using ethanol and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this patented process offers tangible benefits that extend beyond mere chemical yield. The transition from a multi-step, low-yield legacy process to this streamlined single-addition protocol directly impacts the cost of goods sold (COGS). By eliminating the need for multiple recrystallizations and reducing the formation of difficult-to-separate isomers, the process significantly reduces solvent consumption and energy usage associated with extended heating and cooling cycles. This operational simplicity translates into substantial cost savings in manufacturing overheads. Furthermore, the robustness of the reaction conditions allows for more predictable batch times, enabling better production planning and inventory management. The ability to consistently achieve yields over 58% means that less raw material is required per kilogram of finished product, effectively lowering the input cost basis for this critical pharmaceutical intermediate.
- Cost Reduction in Manufacturing: The elimination of batch-wise catalyst addition removes a major source of operational variability and labor intensity. In traditional methods, the slow addition of catalyst requires constant monitoring and extends the batch cycle time significantly. By switching to a single-addition method at controlled low temperatures, the plant can reduce man-hours per batch and minimize the risk of human error. Additionally, the higher selectivity means that valuable starting materials like 2-chloro-5-nitrobenzoyl chloride are converted more efficiently into the desired product rather than wasted as ortho-isomer byproducts. This improved atom economy directly contributes to a lower cost per unit, providing a competitive edge in pricing strategies for bulk chemical procurement without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by complex processes that are prone to failure or deviation. The simplified workflow described in this patent reduces the number of critical control points where a batch could be rejected. With fewer purification steps required—specifically the reduction to a single recrystallization—the lead time for producing high-purity pharmaceutical intermediates is drastically shortened. This agility allows suppliers to respond more quickly to fluctuating market demands. Moreover, the use of common, commercially available solvents like dichloromethane and ethanol ensures that raw material sourcing remains stable and unaffected by niche supply shortages. This reliability is crucial for maintaining uninterrupted production schedules for downstream API manufacturers who depend on timely deliveries.
- Scalability and Environmental Compliance: Scaling chemical processes from the lab to the plant floor often reveals hidden thermal hazards, but this method's emphasis on low-temperature control mitigates such risks. The exothermic nature of the Friedel-Crafts reaction is managed effectively by the cryogenic conditions, making the process safer to operate at the 100 kg to 100 MT scale. From an environmental perspective, the reduction in solvent volume and the minimization of byproduct waste align with green chemistry principles. Less waste generation means lower costs for hazardous waste disposal and a reduced environmental footprint. This compliance with stringent environmental regulations facilitates smoother regulatory approvals and enhances the sustainability profile of the supply chain, a key metric for modern corporate responsibility goals.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is essential for stakeholders evaluating its implementation. The following questions address common concerns regarding the practical application of this technology, focusing on the specific improvements over prior art and the quality metrics achieved. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, ensuring that all answers reflect the verified capabilities of the method.
Q: What is the primary advantage of the single-addition catalyst method?
A: The single addition of anhydrous aluminum trichloride at low temperatures significantly simplifies operation compared to batch addition, ensuring better temperature control and higher para-selectivity.
Q: How does this process improve product purity?
A: By minimizing ortho-position byproducts through precise thermal management, the process achieves over 98% purity after just one recrystallization step.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the simplified workflow, easy separation of products, and robust yield exceeding 58% make it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloro-5-Nitrophenyl-4'-Methoxybenzophenone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications. Our technical team has thoroughly analyzed the pathway described in CN100386305C and validated its potential for industrial application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop to reactor is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 2-chloro-5-nitrophenyl-4'-methoxybenzophenone meets the >98% purity threshold required for sensitive pharmaceutical applications. We are committed to delivering consistency and reliability in every shipment.
We invite global partners to collaborate with us to leverage this advanced synthetic route for their supply chains. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our expertise in fine chemical intermediates can optimize your production costs and secure your supply of this essential building block.
