Advanced Synthesis of Oxophthalazinyl Acetic Acids for Aldose Reductase Inhibitors
Advanced Synthesis of Oxophthalazinyl Acetic Acids for Aldose Reductase Inhibitors
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for complex heterocyclic intermediates, particularly those serving as critical building blocks for treating chronic metabolic disorders. Patent CN1024347C discloses a sophisticated methodology for the preparation of oxophthalazinyl acetic acids featuring benzothiazole or other heterocyclic side chains. These specific molecular architectures are identified as potent aldose reductase inhibitors, offering significant therapeutic potential for managing severe diabetic complications including cataracts, retinopathy, and neuropathy. The technical disclosure provides a comprehensive framework for constructing the phthalazinone core through a sequence involving Reformatsky condensation, acid-catalyzed dehydration, and hydrazine-mediated cyclization. For R&D directors and procurement specialists evaluating reliable pharmaceutical intermediate supplier options, understanding the mechanistic nuances and operational feasibility of this patented route is essential for securing a stable supply of high-purity active pharmaceutical ingredient (API) precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for constructing substituted phthalazinone scaffolds often encounter significant hurdles regarding regioselectivity, yield optimization, and the handling of hazardous reagents. Conventional methods frequently rely on harsh cyclization conditions that can degrade sensitive functional groups or lead to complex mixtures of isomers, thereby complicating downstream purification processes. Furthermore, earlier approaches to introducing heterocyclic side chains, such as benzothiazole moieties, often required multi-step sequences with poor atom economy and the use of expensive transition metal catalysts that necessitate rigorous removal to meet regulatory standards. These inefficiencies translate directly into increased manufacturing costs and extended lead times, creating bottlenecks for cost reduction in API manufacturing. The inability to easily control the substitution pattern on the phthalazinone ring without protecting group strategies further limits the versatility of older methodologies, making them less attractive for the rapid development of diverse analog libraries required in modern drug discovery programs.
The Novel Approach
The methodology outlined in the patent data presents a streamlined and chemically elegant solution by leveraging a versatile nitrile-containing intermediate that undergoes efficient coupling with aniline derivatives. This novel approach utilizes a pre-formed acid addition salt strategy or in situ salt formation to drive the condensation reaction between the oxophthalazinyl acetic acid ester and the aniline component under controlled thermal conditions. By employing strong acid catalysis during the coupling phase, the reaction kinetics are significantly enhanced, allowing for the formation of the desired carbon-nitrogen bonds with high fidelity. This strategy effectively bypasses the need for exotic catalysts and simplifies the workup procedure, as the acid salts can often be managed through standard aqueous extraction or crystallization techniques. The flexibility of this route allows for the introduction of various heterocyclic side chains, providing a modular platform for generating diverse biological candidates while maintaining a consistent and reproducible core synthesis.
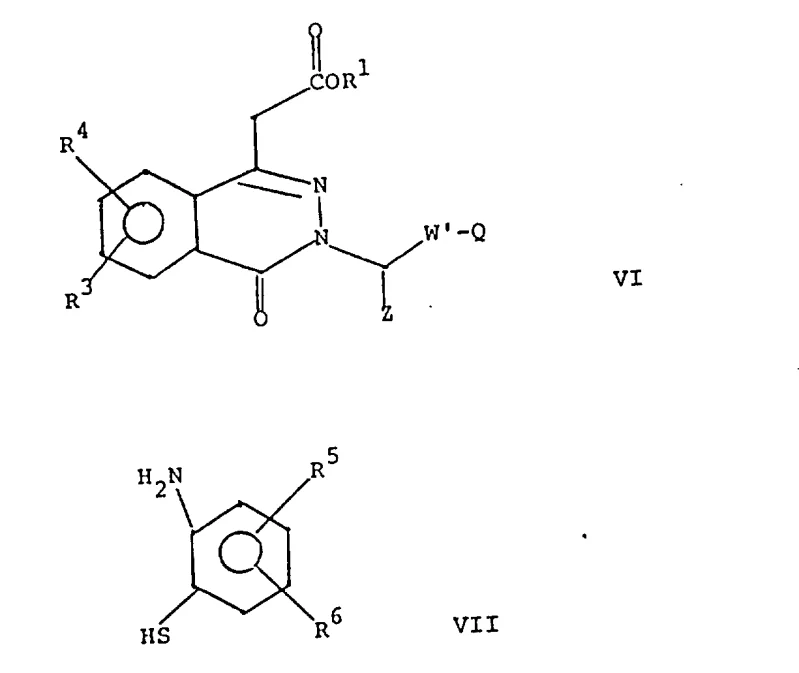
Mechanistic Insights into Reformatsky Condensation and Cyclization
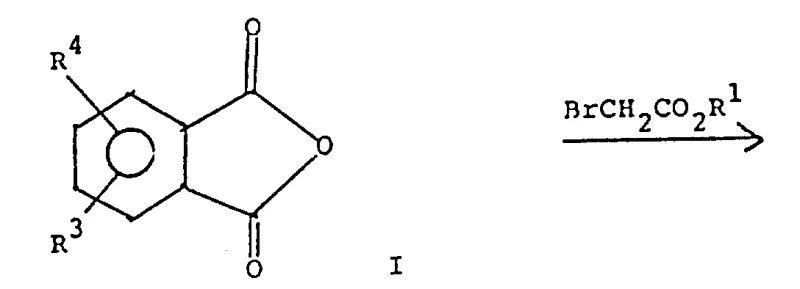
The foundational step of this synthesis involves the Reformatsky reaction, where an alpha-halo ester reacts with phthalic anhydride in the presence of activated zinc or a zinc-copper couple. This organometallic transformation generates a zinc enolate intermediate that nucleophilically attacks the carbonyl carbon of the anhydride, leading to the formation of a beta-hydroxy ester which spontaneously lactonizes to form the isobenzofuranone derivative. The choice of solvent, typically benzene or tetrahydrofuran, and the maintenance of reflux temperatures are critical parameters that ensure the complete consumption of the zinc metal and the stabilization of the organozinc species. Following this, the hydroxy-lactone intermediate undergoes acid-catalyzed dehydration, often utilizing p-toluenesulfonic acid or sulfuric acid, to generate an exocyclic double bond. This unsaturated lactone is highly reactive towards nucleophiles, setting the stage for the subsequent ring-expansion reaction.
The cyclization to the phthalazinone core is achieved through the reaction of the unsaturated lactone with hydrazine, which acts as a bidentate nucleophile attacking both the ester carbonyl and the lactone carbonyl centers. This double addition-elimination sequence results in the expulsion of alcohol and water molecules, driving the equilibrium towards the formation of the stable six-membered diazine ring fused to the benzene system. The resulting 4-oxo-3,4-dihydrophthalazine-1-acetic acid derivative serves as a pivotal scaffold for further functionalization. Subsequent alkylation at the nitrogen or carbon positions allows for the attachment of various side chains, such as cyanomethyl or hydroxymethyl groups, which can be further transformed into the final heterocyclic substituents. This mechanistic pathway ensures high structural integrity and minimizes the formation of regioisomeric impurities, which is paramount for meeting stringent purity specifications in pharmaceutical manufacturing.
How to Synthesize Oxophthalazinyl Acetic Acid Efficiently
The synthesis of these valuable intermediates requires precise control over reaction parameters to maximize yield and minimize impurity profiles. The process begins with the careful activation of zinc dust and the slow addition of the halo-ester to control the exotherm during the Reformatsky step. Subsequent steps involve standard organic transformations such as dehydration, hydrazinolysis, and nucleophilic substitution, all of which are well-suited for batch processing in standard glass-lined or stainless steel reactors. The detailed standardized synthesis steps see the guide below for specific stoichiometric ratios and temperature profiles optimized for reproducibility.
- Perform a Reformatsky reaction between phthalic anhydride and alpha-halo ester using zinc or zinc-copper couple in refluxing benzene or THF to form the hydroxy-lactone intermediate.
- Dehydrate the hydroxy-lactone using strong acid catalysis to generate the unsaturated lactone, followed by cyclization with hydrazine to form the phthalazinone core.
- Alkylate the phthalazinone nitrogen or carbon position with a functionalized side chain precursor, followed by coupling with an aniline derivative to finalize the heterocyclic structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers substantial advantages regarding raw material availability and process safety, directly addressing key concerns for supply chain heads. The starting materials, including phthalic anhydride and common alpha-halo esters, are commodity chemicals available from multiple global sources, ensuring supply continuity and mitigating the risk of single-source dependency. The elimination of precious metal catalysts not only reduces the direct cost of goods sold but also simplifies the environmental compliance burden associated with heavy metal waste disposal. This translates into significant cost reduction in API manufacturing, as the downstream purification trains do not require specialized scavenging resins or complex filtration units to remove trace metals. Furthermore, the use of common solvents like ethanol, toluene, and DMF allows for efficient solvent recovery and recycling, enhancing the overall sustainability profile of the manufacturing process.
- Cost Reduction in Manufacturing: The process avoids the use of expensive palladium or rhodium catalysts, relying instead on inexpensive zinc and basic mineral acids, which drastically lowers the raw material expenditure per kilogram of product. The high conversion rates observed in the cyclization steps reduce the volume of mother liquor requiring treatment, thereby lowering waste management costs. Additionally, the ability to perform telescoped reactions, where intermediates are not isolated but carried forward in solution, can significantly reduce processing time and solvent consumption. These factors combine to create a highly cost-competitive manufacturing model that supports aggressive pricing strategies for the final API.
- Enhanced Supply Chain Reliability: Since the synthesis relies on widely available bulk chemicals rather than specialized custom synthons, the risk of supply disruption is minimized. The robustness of the reaction conditions, which tolerate moderate variations in temperature and stoichiometry, ensures consistent batch-to-batch quality even when scaling up production volumes. This reliability is crucial for maintaining uninterrupted production schedules for downstream drug products. The simplified purification protocols also mean that manufacturing cycles are shorter, allowing for faster turnaround times and improved responsiveness to market demand fluctuations without compromising on quality standards.
- Scalability and Environmental Compliance: The reaction steps are inherently scalable, utilizing standard unit operations that are easily transferred from pilot plant to commercial scale facilities. The absence of pyrophoric reagents or extreme high-pressure conditions enhances operational safety, reducing insurance and facility maintenance costs. Moreover, the aqueous workups and crystallization steps generate waste streams that are easier to treat compared to those containing heavy metals or persistent organic pollutants. This alignment with green chemistry principles facilitates smoother regulatory approvals and supports corporate sustainability goals, making the process attractive for long-term commercial partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these oxophthalazinyl derivatives. The answers are derived directly from the experimental data and process descriptions found within the patent literature, ensuring accuracy and relevance for technical decision-makers. Understanding these details helps in assessing the feasibility of integrating this chemistry into existing manufacturing portfolios.
Q: What are the primary therapeutic applications of these oxophthalazinyl compounds?
A: These compounds function as potent aldose reductase inhibitors, specifically designed to treat chronic diabetic complications such as cataracts, retinopathy, and neuropathy by blocking the polyol pathway.
Q: How does the Reformatsky reaction condition impact the purity of the intermediate?
A: Utilizing activated zinc or zinc-copper couples in anhydrous solvents like THF ensures efficient organozinc formation, minimizing side reactions and facilitating high-purity isolation of the hydroxy-lactone precursor.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the process relies on standard unit operations such as reflux, filtration, and crystallization using common solvents, making it highly amenable to commercial scale-up from kilogram to metric ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxophthalazinyl Acetic Acid Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and contract development, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at optimizing the specific reaction conditions described in patent CN1024347C to ensure stringent purity specifications are met for every batch. We utilize rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the identity and purity of complex heterocyclic intermediates, guaranteeing that our clients receive materials that are ready for immediate use in GMP API synthesis. Our commitment to quality and consistency makes us a trusted partner for pharmaceutical companies seeking to secure their supply chains for critical diabetes medication intermediates.
We invite you to engage with our technical procurement team to discuss your specific project requirements and explore how our manufacturing capabilities can support your development timelines. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can reduce your overall production expenses. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your unique molecular targets. Let us collaborate to bring your next generation of aldose reductase inhibitors from the laboratory bench to the global market efficiently and reliably.
