Scalable Synthesis of Potent Matrix Metalloproteinase Inhibitors for Commercial API Production
Introduction to Advanced MMP Inhibitor Synthesis
The development of potent matrix metalloproteinase (MMP) inhibitors represents a critical frontier in the treatment of oncology and inflammatory diseases, as detailed in patent CN1380279A. This intellectual property discloses a sophisticated class of (4-alkynyl)-aromatic keto acid compounds that exhibit remarkable selectivity against specific MMP subtypes, offering a promising therapeutic avenue for conditions such as cancer metastasis, arthritis, and atherosclerosis. The core innovation lies in a versatile synthetic methodology that combines classical Friedel-Crafts acylation with modern palladium-catalyzed cross-coupling techniques. For pharmaceutical manufacturers, understanding the nuances of this chemistry is essential, as it provides a robust framework for producing high-value active pharmaceutical ingredient (API) intermediates. The structural diversity allowed by this method enables the fine-tuning of pharmacokinetic properties, making it a highly attractive candidate for commercial development by a reliable pharmaceutical intermediate supplier.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing aryl keto acids often suffer from significant drawbacks regarding regioselectivity and functional group tolerance. Conventional Friedel-Crafts reactions can lead to poly-acylation or rearrangement issues, particularly when dealing with sensitive substrates required for biological activity. Furthermore, introducing alkynyl moieties onto aromatic rings typically requires harsh conditions that may degrade other parts of the molecule, such as ester or amide functionalities present in complex drug candidates. Older methods often lack the modularity needed to rapidly generate libraries of analogs for structure-activity relationship (SAR) studies, leading to prolonged development timelines. The inability to efficiently install the critical 4-alkynyl group without compromising the integrity of the keto-acid backbone has historically been a bottleneck in the cost reduction in API manufacturing for this class of compounds.
The Novel Approach
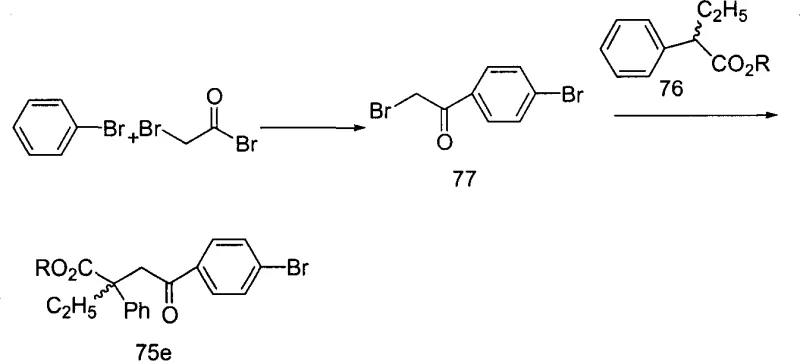
The methodology outlined in the patent overcomes these hurdles by employing a stepwise strategy that isolates the construction of the carbon skeleton from the installation of the pharmacophore. By first establishing the 4-halophenyl ketone scaffold through controlled acylation, the process ensures high regiochemical purity before subjecting the molecule to cross-coupling. This modular design allows for the late-stage introduction of various alkynes, significantly enhancing the flexibility of the synthesis. As illustrated in the reaction scheme below, the alkylation of alpha-halo ketones with substituted acetates provides a precise route to introduce steric bulk and chirality at the alpha-position, which is vital for enzyme binding affinity. This strategic decoupling of synthetic steps minimizes side reactions and simplifies purification, thereby streamlining the path to commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Pd-Catalyzed Sonogashira Coupling
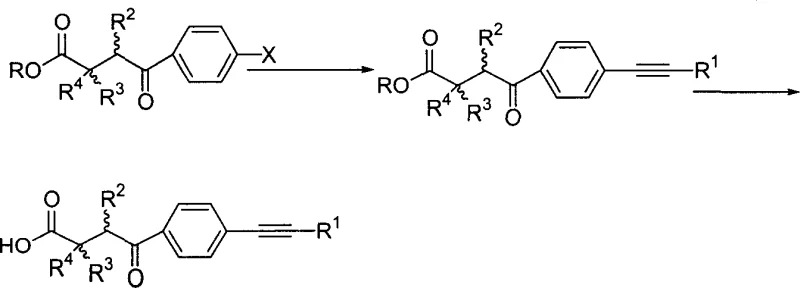
The heart of this synthetic innovation is the palladium-catalyzed Sonogashira coupling, which facilitates the formation of the carbon-carbon triple bond between the aryl halide and the terminal alkyne. Mechanistically, this cycle begins with the oxidative addition of the aryl bromide to the zero-valent palladium catalyst, forming an aryl-palladium(II) species. Simultaneously, a copper co-catalyst activates the terminal alkyne by forming a copper-acetylide complex, which is more nucleophilic than the free alkyne. Transmetallation then occurs, transferring the alkynyl group from copper to the palladium center, followed by reductive elimination to release the coupled product and regenerate the active catalyst. This mechanism operates under relatively mild thermal conditions (50-100°C), preserving sensitive functional groups like esters and carbamates that are essential for the biological activity of the final MMP inhibitor. The inclusion of amine bases serves to neutralize the hydrogen halide byproduct, driving the equilibrium forward and ensuring high conversion rates.
Following the coupling reaction, the final step involves the hydrolysis of the ester moiety to reveal the free carboxylic acid, which acts as the zinc-binding group in the MMP active site. This hydrolysis is typically performed under basic conditions using sodium or potassium hydroxide, followed by careful acidification to precipitate the product. The choice of hydrolysis conditions is critical; overly aggressive conditions could lead to decarboxylation or degradation of the alkynyl linkage. However, the patent demonstrates that standard aqueous base hydrolysis is sufficient to cleave the ester without affecting the newly formed alkyne or the ketone functionality. This robustness in the final deprotection step is a key factor in reducing lead time for high-purity pharmaceutical intermediates, as it eliminates the need for specialized protecting group strategies that add cost and complexity to the manufacturing process.
How to Synthesize 4-Alkynyl-Aryl Keto Acids Efficiently
The synthesis of these valuable intermediates follows a logical progression starting from commercially available halobenzenes and succinic derivatives. The initial acylation step establishes the four-carbon chain attached to the aromatic ring, setting the stage for subsequent functionalization. Operators must maintain strict anhydrous conditions during the Lewis acid catalyzed steps to prevent catalyst deactivation and ensure optimal yields. Following the formation of the bromo-ketone intermediate, the process moves to the crucial cross-coupling phase where the alkyne is introduced. Detailed standard operating procedures regarding catalyst loading, temperature ramping, and workup protocols are essential for reproducibility. For a comprehensive guide on the specific molar ratios, solvent choices, and purification techniques validated in the patent examples, please refer to the standardized synthesis steps provided below.
- Perform Friedel-Crafts acylation of halobenzene with succinic anhydride derivatives or haloacetyl halides using Lewis acids like AlCl3 to form the keto-acid backbone.
- Execute alkylation reactions using organolithium reagents at low temperatures to introduce chiral or substituted side chains at the alpha-position.
- Conduct Sonogashira coupling with terminal alkynes using Pd/Cu catalysts followed by ester hydrolysis to yield the final carboxylic acid product.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this synthetic route offers distinct advantages rooted in the availability of starting materials and the simplicity of unit operations. The primary building blocks, such as bromobenzene, succinic anhydride, and various terminal alkynes, are commodity chemicals available from multiple global sources, mitigating the risk of single-source dependency. This abundance of raw materials translates directly into enhanced supply chain reliability, as procurement teams can easily secure feedstock even during market fluctuations. Furthermore, the reaction conditions, while requiring standard inert atmosphere techniques, do not demand exotic high-pressure equipment or cryogenic infrastructure beyond standard laboratory capabilities, making the technology transfer to pilot and production plants straightforward and cost-effective.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for expensive transition metal removal steps often associated with more complex catalytic cycles, as the palladium loading is kept minimal relative to the substrate. By utilizing a convergent synthesis strategy where the alkyne and the keto-acid fragments are prepared separately and joined late in the sequence, the overall process efficiency is maximized, reducing waste generation and solvent consumption. This streamlined approach inherently lowers the cost of goods sold (COGS) by minimizing the number of isolation and purification stages required to achieve pharmaceutical grade purity, delivering substantial cost savings without compromising quality.
- Enhanced Supply Chain Reliability: The robustness of the chemistry ensures consistent batch-to-batch quality, which is paramount for maintaining uninterrupted API production schedules. Since the key intermediates are stable and can be stored for extended periods, manufacturers can build strategic inventory buffers to absorb demand shocks. The reliance on well-understood reaction mechanisms like Friedel-Crafts and Sonogashira coupling means that technical support and troubleshooting expertise are widely available within the industry, further securing the supply chain against technical obsolescence or operational failures.
- Scalability and Environmental Compliance: The process generates manageable waste streams primarily consisting of aqueous salts and organic solvents that can be recovered and recycled through standard distillation protocols. The absence of highly toxic reagents or persistent organic pollutants simplifies the environmental permitting process for new manufacturing facilities. Additionally, the high atom economy of the coupling reaction ensures that a significant proportion of the reactant mass ends up in the final product, aligning with green chemistry principles and reducing the environmental footprint associated with large-scale chemical production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these MMP inhibitor intermediates. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these details is crucial for evaluating the feasibility of integrating this chemistry into your existing product portfolio or R&D pipeline. We encourage stakeholders to review these insights to fully appreciate the strategic value of this technology.
Q: What is the primary advantage of the Sonogashira coupling route in this patent?
A: The Sonogashira coupling allows for the mild and selective introduction of diverse alkynyl groups at the para-position of the phenyl ring, which is critical for tuning the selectivity against specific MMP subtypes like MMP-3 and MMP-12.
Q: How does the process handle chiral centers in the intermediate synthesis?
A: The patent details the use of chiral succinic lactones or asymmetric alkylation with organolithium reagents at low temperatures (-78°C) to establish stereochemistry early in the synthesis, ensuring high optical purity in the final inhibitor.
Q: Are the reaction conditions suitable for large-scale manufacturing?
A: Yes, the process utilizes standard industrial reagents like aluminum trichloride and palladium catalysts. While some steps require anhydrous conditions, the overall pathway avoids exotic reagents, facilitating scale-up from kilogram to tonnage production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Alkynyl-Aryl Keto Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of these (4-alkynyl)-aromatic keto acid compounds in the next generation of therapeutics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our facility is equipped with state-of-the-art rigorous QC labs capable of meeting stringent purity specifications required for oncology and anti-inflammatory drug candidates. We understand that consistency is key in pharmaceutical supply, and our dedicated process chemistry team is ready to optimize this specific route to maximize yield and minimize impurities for your specific needs.
We invite you to engage with our technical procurement team to discuss how we can support your supply chain goals. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of sourcing these intermediates through our optimized channels. We are prepared to provide specific COA data and route feasibility assessments tailored to your volume requirements, ensuring that you have all the necessary information to make informed sourcing decisions. Let us be your partner in bringing these life-saving medicines to market faster and more efficiently.
