Revolutionizing Mandelic Acid Production: A Green Styrene-Based Route for Global Supply Chains
Revolutionizing Mandelic Acid Production: A Green Styrene-Based Route for Global Supply Chains
The landscape of fine chemical manufacturing is constantly evolving, driven by the dual imperatives of cost efficiency and environmental sustainability. A pivotal advancement in this domain is detailed in patent CN112321410A, which introduces a novel method for synthesizing mandelic acid, a critical intermediate widely utilized in the pharmaceutical, agrochemical, and cosmetic industries. Unlike traditional pathways that rely on hazardous cyanide reagents or generate excessive wastewater, this innovative approach leverages styrene as a foundational raw material. By employing trichloroisocyanuric acid (TCCA) as a dual-function oxidizing and chlorinating agent, the process achieves a direct transformation of styrene into 2,2-dichloroacetophenone, which is subsequently hydrolyzed to yield high-purity mandelic acid. This technological breakthrough not only simplifies the synthetic route but also aligns perfectly with modern green chemistry principles, offering a robust solution for a reliable pharmaceutical intermediates supplier seeking to optimize their production capabilities.
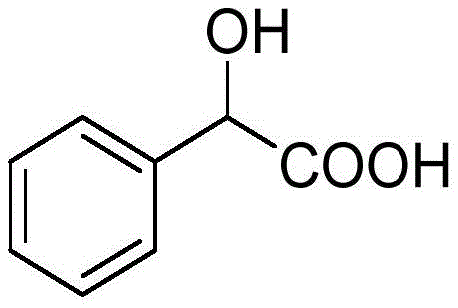
The structural integrity and purity of the final product are paramount for downstream applications, ranging from urinary tract bactericides to heterocyclic disperse dyes. The method outlined in the patent ensures that the resulting mandelic acid meets stringent quality specifications required by global regulatory bodies. By shifting away from the legacy benzaldehyde oxidation methods, which are plagued by low yields and severe toxicity issues, this new protocol establishes a safer operational environment. For R&D directors and procurement managers alike, understanding the mechanistic nuances of this styrene-based route is essential for evaluating its potential to reduce lead times and enhance supply chain resilience in the competitive market of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of mandelic acid has been dominated by three primary methodologies, each carrying significant drawbacks that hinder efficient large-scale production. The earliest method, the benzaldehyde oxidation route, involves the formation of hydroxyl phenylacetonitrile via a cyanide reaction followed by hydrolysis. While chemically straightforward, this process is fundamentally flawed due to the extreme toxicity of cyanide reagents, posing severe safety risks to personnel and creating complex waste disposal challenges that have led to its gradual elimination in regulated markets. Another prevalent technique is the phase transfer catalysis method, where benzaldehyde reacts with chloroform in a concentrated sodium hydroxide solution. Although this method offers mild reaction conditions and yields exceeding 76%, it necessitates multiple ether extraction steps, resulting in substantial solvent consumption and the generation of large volumes of saline wastewater that require expensive treatment protocols before discharge.
Furthermore, the acetophenone derivation method, which involves direct chlorination of acetophenone followed by hydrolysis, presents its own set of operational hazards. Acetophenone is known to be irritating to the eyes and possesses a strong, unpleasant odor, complicating workplace safety management. Additionally, like the phase transfer method, this route suffers from high wastewater generation, impacting the overall environmental footprint of the manufacturing facility. These conventional approaches collectively represent a bottleneck for cost reduction in pharmaceutical intermediates manufacturing, as the expenses associated with safety compliance, waste treatment, and raw material volatility erode profit margins. The industry urgently requires a paradigm shift towards a more streamlined, eco-friendly synthesis strategy that mitigates these inherent risks while maintaining high product quality.
The Novel Approach
The methodology disclosed in patent CN112321410A represents a transformative departure from these legacy processes by utilizing styrene, a commodity chemical that is inexpensive, low in toxicity, and non-irritating. The core innovation lies in the use of trichloroisocyanuric acid (TCCA) within a mixed solvent system comprising water and organic co-solvents such as ethylene glycol dimethyl ether, methanol, or ethanol. TCCA serves a dual role, acting as both a potent oxidant and a chlorinating agent to convert styrene directly into 2,2-dichloroacetophenone in a single step. This consolidation of reaction steps significantly shortens the production cycle, thereby enhancing throughput and reducing the energy consumption associated with multi-stage processing. The subsequent hydrolysis of the dichloro-intermediate under alkaline conditions proceeds smoothly to yield the target mandelic acid, bypassing the need for toxic cyanides or harsh chloroform reagents entirely.
Beyond the immediate reaction benefits, this novel approach introduces a closed-loop element regarding reagent usage that is rare in fine chemical synthesis. The reaction of TCCA generates cyanuric acid as a solid byproduct, which can be easily separated via filtration. Crucially, this cyanuric acid is not waste; it can be re-chlorinated to regenerate TCCA, allowing for a cyclical reuse of the chlorinating agent. This feature dramatically lowers the net consumption of reagents and minimizes solid waste discharge, addressing the critical pain points of environmental compliance and raw material costs. For supply chain leaders, this translates to a more predictable and sustainable sourcing model for commercial scale-up of complex pharmaceutical intermediates, ensuring that production capacity is not constrained by waste disposal limits or volatile reagent pricing.
Mechanistic Insights into TCCA-Mediated Oxidation-Chlorination
The chemical elegance of this synthesis lies in the specific reactivity of trichloroisocyanuric acid towards the vinyl group of styrene. In the initial oxidation-chlorination stage, TCCA functions as a source of electrophilic chlorine and oxygen species. When styrene is introduced into the solvent mixture at temperatures ranging from 50°C to 90°C, the electron-rich double bond of the styrene molecule undergoes attack by the active chlorine species generated from TCCA. This results in the formation of a chlorohydrin intermediate which rapidly rearranges and undergoes further oxidation to yield 2,2-dichloroacetophenone. The choice of solvent is critical here; the specific volume ratio of organic co-solvent to water (optimized at 1:4.5) ensures adequate solubility of the organic substrate while maintaining the necessary polarity for the ionic mechanisms involved in the hydrolysis of the N-Cl bonds in TCCA. This precise control over the reaction medium prevents side reactions such as polymerization of styrene, thereby maximizing the selectivity towards the desired dichloro-ketone intermediate.
Following the isolation of 2,2-dichloroacetophenone, the mechanism shifts to nucleophilic substitution during the hydrolysis phase. Under alkaline conditions provided by sodium hydroxide at a controlled temperature of 50°C, the chloride atoms on the alpha-carbon are displaced by hydroxide ions. This substitution proceeds through a gem-diol intermediate which is unstable and spontaneously dehydrates to form the alpha-hydroxy ketone structure, ultimately tautomerizing or rearranging to the stable mandelic acid form upon acidification. The use of activated carbon during the final crystallization step plays a vital role in impurity control, adsorbing colored byproducts and trace organic residues that could compromise the optical properties of the final crystal. This rigorous purification protocol ensures that the final mandelic acid meets the stringent purity specifications required for sensitive applications in ophthalmic drugs and chiral synthesis, demonstrating the robustness of the chemical design.
How to Synthesize Mandelic Acid Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to ensure optimal yield and safety. The process begins with the preparation of the solvent system, where precise ratios of water and organic co-solvents are mixed to create a homogeneous reaction medium. Styrene is added first, followed by the controlled addition of trichloroisocyanuric acid to manage the exothermic nature of the oxidation. Maintaining the temperature within the 50°C to 90°C window is crucial; too low a temperature may result in incomplete conversion, while excessive heat could promote degradation or side reactions. Once the oxidation-chlorination is complete, the reaction mixture is cooled to facilitate the precipitation of cyanuric acid, which is filtered off for recycling. The filtrate is then extracted with dichloromethane to isolate the 2,2-dichloroacetophenone intermediate, which serves as the precursor for the final hydrolysis step.
- React styrene with trichloroisocyanuric acid in a mixed solvent system (e.g., ethylene glycol dimethyl ether and water) at 50-90°C to form 2,2-dichloroacetophenone.
- Filter the reaction mixture to recover cyanuric acid byproduct and extract the organic layer containing the intermediate.
- Hydrolyze the 2,2-dichloroacetophenone in sodium hydroxide solution at 50°C, then acidify with hydrochloric acid to precipitate pure mandelic acid crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this styrene-based synthesis method offers compelling strategic advantages that extend beyond simple chemical efficiency. The primary driver for cost optimization is the substitution of high-cost, high-risk raw materials like benzaldehyde and cyanide with styrene, a bulk petrochemical derivative that is abundantly available and priced significantly lower. This shift in feedstock fundamentally alters the cost structure of mandelic acid production, enabling manufacturers to offer more competitive pricing without sacrificing margin. Furthermore, the ability to recycle the cyanuric acid byproduct back into trichloroisocyanuric acid creates a self-sustaining reagent loop, effectively decoupling production costs from the fluctuating market prices of chlorinating agents. This internal circular economy significantly dampens the impact of external supply shocks, providing a stable cost baseline for long-term contracts.
- Cost Reduction in Manufacturing: The elimination of toxic cyanide reagents removes the need for specialized containment equipment and expensive hazardous waste disposal services, which are major cost centers in traditional facilities. Additionally, the shortened process flow reduces utility consumption (steam, electricity) and labor hours per batch. By avoiding the multiple ether extractions required in phase transfer methods, solvent recovery costs are also minimized. These cumulative efficiencies result in substantial cost savings that can be passed down the supply chain, making the final product more attractive to price-sensitive buyers in the generic drug and agrochemical sectors.
- Enhanced Supply Chain Reliability: Relying on styrene as a feedstock enhances supply security because it is produced on a massive global scale for the plastics industry, ensuring consistent availability even during regional disruptions. The robustness of the reaction conditions (mild temperatures and ambient pressure) reduces the risk of unplanned shutdowns due to equipment failure or safety incidents. Moreover, the simplified workflow allows for faster batch turnover times, enabling manufacturers to respond more agilely to sudden spikes in demand. This reliability is critical for partners seeking to reduce lead time for high-purity pharmaceutical intermediates and maintain just-in-time inventory levels.
- Scalability and Environmental Compliance: The green nature of this process aligns perfectly with increasingly strict global environmental regulations. The reduction in wastewater volume and the absence of heavy metal or cyanide contamination simplify the permitting process for plant expansion. The solid byproduct (cyanuric acid) is easy to handle and store, unlike liquid hazardous wastes. This environmental compatibility facilitates smoother scaling from pilot plants to multi-ton commercial production, ensuring that supply can grow in tandem with market demand without encountering regulatory bottlenecks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel mandelic acid synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in patent CN112321410A, providing a clear picture of what partners can expect when integrating this route into their supply chains. Understanding these details is crucial for making informed decisions about vendor selection and process validation.
Q: What are the primary advantages of using styrene over benzaldehyde for mandelic acid synthesis?
A: Styrene is significantly cheaper, less toxic, and non-irritating compared to benzaldehyde. Furthermore, the traditional benzaldehyde cyanohydrin route involves highly toxic cyanide, whereas the styrene route described in patent CN112321410A utilizes trichloroisocyanuric acid, offering a much safer and environmentally friendly profile.
Q: How does this process address waste management and environmental concerns?
A: The process generates cyanuric acid as a solid byproduct, which can be filtered and recycled back into trichloroisocyanuric acid through chlorination. This circular chemistry drastically reduces waste discharge compared to phase transfer methods that generate large volumes of saline wastewater.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the method features mild reaction conditions (50-90°C) and uses common solvents like methanol, ethanol, or ethylene glycol dimethyl醚 mixed with water. The short process flow and the ability to recover key reagents make it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mandelic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthesis routes is not just a technical upgrade but a strategic necessity for modern pharmaceutical and chemical enterprises. Our team of expert chemists has thoroughly analyzed the potential of the styrene-based pathway described in CN112321410A and is fully prepared to translate this laboratory-scale innovation into industrial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this process are realized in tangible output. Our state-of-the-art facilities are equipped with rigorous QC labs capable of monitoring every step of the oxidation and hydrolysis phases, guaranteeing that every batch of mandelic acid delivered meets the stringent purity specifications demanded by top-tier global clients.
We invite you to collaborate with us to leverage this advanced technology for your specific application needs. Whether you require mandelic acid for API synthesis, cosmetic formulations, or agrochemical intermediates, our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us partner with you to build a more sustainable, cost-effective, and reliable supply chain for your critical chemical ingredients.
