Advanced Tetradentate Phosphine Ligands for Efficient Linear Dialdehyde Manufacturing
Advanced Tetradentate Phosphine Ligands for Efficient Linear Dialdehyde Manufacturing
The chemical industry's relentless pursuit of efficient polyester monomer synthesis has found a significant breakthrough with the disclosure of patent CN115124572A. This pivotal intellectual property introduces a novel class of tetradentate phosphine ligands designed specifically to overcome the longstanding limitations in diolefin hydroformylation reactions. Linear dialdehydes, such as 1,6-hexanedial, serve as critical precursors for high-value downstream products like 1,6-adipic acid and 1,6-hexanediol, which are indispensable in the production of advanced polyester materials. Traditional methods for generating these monomers often suffer from environmental hazards or suboptimal selectivity, creating a pressing demand for greener and more precise catalytic solutions. The technology described in this patent offers a robust alternative, leveraging a sophisticated molecular architecture that ensures exceptional stability and reactivity. By addressing the core issues of catalyst deactivation and metal leaching, this innovation paves the way for more sustainable and cost-effective manufacturing pathways in the fine chemicals sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of linear dialdehydes has been plagued by significant technical hurdles that impact both economic viability and environmental compliance. Existing processes, such as the cyclohexene oxide method, generate substantial quantities of harmful byproducts, necessitating complex waste treatment protocols that drive up operational costs. Furthermore, earlier catalytic systems utilizing cobalt complexes or simple phosphine ligands have demonstrated inadequate performance metrics. For instance, prior art methods often struggle to achieve high selectivity, with some processes yielding linear dialdehydes at rates as low as 40%, leading to extensive purification burdens and material loss. Additionally, the instability of conventional phosphine ligands under harsh reaction conditions frequently results in the dissociation of the metal center. This phenomenon not only shortens the catalyst's lifespan but also leads to contamination of the final product with heavy metals, a critical quality control issue for pharmaceutical and polymer grade intermediates. The inability to maintain high conversion rates over extended periods forces manufacturers to frequently replenish expensive noble metal catalysts, further eroding profit margins.
The Novel Approach
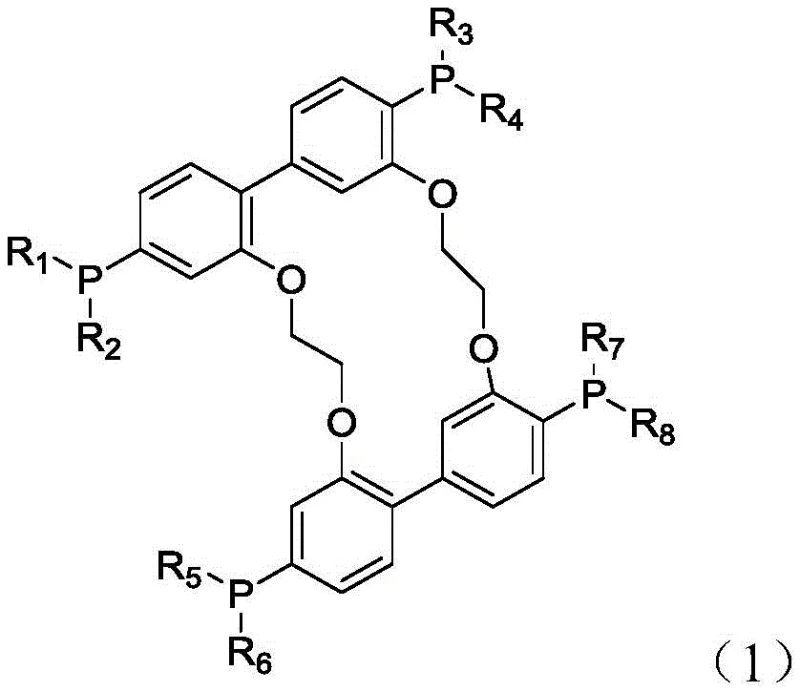
In stark contrast to these legacy technologies, the tetradentate phosphine ligand disclosed in patent CN115124572A represents a paradigm shift in catalyst design. This innovative ligand features a unique macrocyclic structure that provides four coordination sites, creating a remarkably stable complex with transition metals like rhodium and cobalt. This multi-dentate binding mode effectively locks the metal active component in place, virtually eliminating the problem of metal leaching during the reaction cycle. The result is a catalyst system that maintains high activity over prolonged periods, with experimental data indicating successful recycling for up to 20 cycles without significant loss in performance. Moreover, the specific electronic and steric properties of this ligand promote the formation of linear products with extraordinary selectivity, consistently exceeding 97%. This high degree of control minimizes the formation of branched isomers and other side products, simplifying downstream separation processes and enhancing overall yield. The robustness of this new approach allows for operation under milder conditions compared to older methods, reducing energy consumption and improving process safety.
Mechanistic Insights into Tetradentate Phosphine Ligand Catalysis
The superior performance of this tetradentate phosphine ligand can be attributed to its intricate molecular engineering, which optimizes both electronic donation and steric hindrance around the metal center. The ligand's structure, characterized by a central macrocyclic framework linked by ether bridges and substituted phenyl rings, creates a rigid yet adaptable pocket for the metal ion. This geometry facilitates a strong chelating effect, where the four phosphorus atoms coordinate simultaneously with the metal, forming a thermodynamically stable complex. This strong binding affinity is crucial for preventing the decomposition of the catalyst under the high pressures and temperatures typical of hydroformylation reactions. Furthermore, the presence of aromatic rings within the ligand backbone enables pi-pi conjugation interactions with the diolefin substrate. This interaction acts as a pre-organization step, effectively capturing the olefin reactant and positioning it ideally for the insertion of the carbonyl group. Such mechanistic features accelerate the reaction kinetics, allowing for shorter reaction times and higher throughput in industrial reactors.
From an impurity control perspective, the stability of the metal-ligand complex is paramount. In conventional systems, the dissociation of phosphine ligands often leads to the precipitation of metal black or the formation of inactive metal clusters, which act as impurities and reduce catalytic efficiency. The tetradentate nature of this new ligand mitigates this risk by ensuring that the metal remains solubilized and active throughout the process. Analytical data from the patent indicates that the residual metal content in the organic phase after reaction is exceptionally low, often below 2 ppm. This level of purity is critical for applications in sensitive industries such as electronics or high-performance polymers, where trace metal contaminants can degrade material properties. The ligand's resistance to hydrolysis and oxidation further ensures that the catalyst system remains intact even in the presence of trace moisture or oxygen, providing a wider operating window and greater tolerance to feedstock variations.
How to Synthesize Tetradentate Phosphine Ligand Efficiently
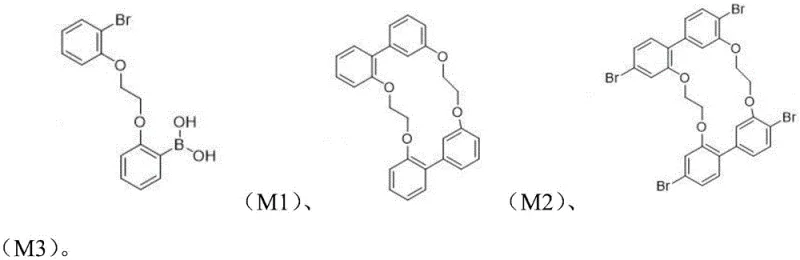
The preparation of this high-performance ligand follows a logical and scalable four-step synthetic route that utilizes readily available starting materials. The process begins with the formation of a key ether-linked intermediate, followed by a palladium-catalyzed coupling to establish the macrocyclic core. Subsequent bromination activates the aromatic rings for the final phosphination step, where the phosphine groups are introduced using substituted phosphine chlorides. This modular approach allows for the customization of the ligand's electronic properties by varying the substituents on the phosphorus atoms, such as phenyl, thienyl, or isopropyl groups. The detailed standard operating procedures for each reaction stage, including precise temperature controls and stoichiometric ratios, are essential for achieving the high purity required for catalytic applications. Manufacturers looking to implement this technology should adhere strictly to the specified reaction conditions to maximize yield and minimize byproduct formation.
- React 1-bromo-2-(chloromethoxy)benzene with (2-hydroxyphenyl)boronic acid using an alkali metal salt catalyst to form intermediate M1.
- Perform a palladium-catalyzed coupling reaction on intermediate M1 to generate the macrocyclic precursor intermediate M2.
- Brominate intermediate M2 using N-bromosuccinimide (NBS) at low temperatures to obtain the brominated intermediate M3.
- React intermediate M3 with substituted phosphine chloride and an alkyllithium catalyst to finalize the tetradentate phosphine ligand structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this tetradentate phosphine ligand technology offers compelling strategic benefits that extend beyond mere technical performance. The primary value proposition lies in the drastic reduction of total cost of ownership associated with the catalyst system. Because the ligand forms such a stable bond with the metal center, the rate of catalyst deactivation is significantly slowed, meaning that less fresh catalyst needs to be purchased over the lifetime of the plant. This longevity translates directly into substantial cost savings on expensive noble metals like rhodium, which represent a major portion of the variable costs in hydroformylation processes. Additionally, the high selectivity of the reaction reduces the burden on downstream purification units. With fewer byproducts and isomers to separate, facilities can operate their distillation columns more efficiently, consuming less energy and reducing solvent usage. This streamlined workflow enhances the overall throughput of the manufacturing site, allowing for better utilization of existing assets.
- Cost Reduction in Manufacturing: The elimination of frequent catalyst replacement and the reduction in metal loss provide a clear pathway to lowering production expenses. By preventing the leaching of active metal components into the product stream, the process avoids the costly and complex steps often required for metal scavenging and product polishing. This efficiency gain is particularly significant when scaling up to commercial volumes, where even minor improvements in yield or catalyst life can result in massive financial impacts. Furthermore, the ability to recycle the catalyst system multiple times without significant loss of activity means that the effective cost per kilogram of product is drastically reduced compared to traditional single-use or short-life catalyst systems.
- Enhanced Supply Chain Reliability: The synthesis of the ligand relies on common chemical building blocks and standard reaction types, such as Suzuki coupling and nucleophilic substitution, which ensures a robust and reliable supply chain. Unlike exotic catalysts that depend on scarce or geopolitically sensitive raw materials, the precursors for this ligand are widely available from multiple global suppliers. This diversity in sourcing mitigates the risk of supply disruptions and price volatility, providing procurement teams with greater negotiating power and stability. The scalability of the synthesis process also means that production can be ramped up quickly to meet surging demand, ensuring continuity of supply for downstream customers who rely on consistent deliveries of linear dialdehydes for their own manufacturing operations.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this technology aligns perfectly with modern sustainability goals. The high atom economy of the hydroformylation reaction, combined with the low waste generation due to high selectivity, minimizes the environmental footprint of the process. The reduction in hazardous waste streams simplifies compliance with increasingly stringent environmental regulations, reducing the administrative and financial burden on the organization. Moreover, the stability of the catalyst reduces the risk of accidental releases of toxic metals, enhancing workplace safety. The ability to operate under milder conditions also contributes to a lower carbon footprint, as less energy is required for heating and pressurization, supporting corporate initiatives to reduce greenhouse gas emissions and achieve carbon neutrality targets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this tetradentate phosphine ligand technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a clear understanding of its practical implications. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this catalyst into existing production lines and for management assessing the long-term strategic value of the technology.
Q: How does this tetradentate ligand improve catalyst stability compared to traditional phosphines?
A: The ligand features a unique macrocyclic structure with strong chelating ability, firmly binding metal active components and preventing leaching, which significantly extends catalyst service life.
Q: What represent the primary advantages for linear dialdehyde selectivity?
A: The specific spatial arrangement and pi-pi conjugation effects of the ligand facilitate rapid capture of diolefins and ensure high insertion selectivity for terminal CO groups, achieving over 97% linear selectivity.
Q: Can this ligand be used for large-scale industrial hydroformylation processes?
A: Yes, the synthesis method is scalable, and the resulting catalyst system demonstrates excellent recyclability and low metal residue in the organic phase, making it highly suitable for continuous industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetradentate Phosphine Ligand Supplier
As the global demand for high-performance polymer intermediates continues to rise, securing a reliable source of advanced catalytic technologies is more critical than ever. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, offering comprehensive support for the commercialization of complex chemical pathways like the one described in patent CN115124572A. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We understand that the success of your hydroformylation process depends not just on the ligand structure, but on the precision of its synthesis and the consistency of its quality. Therefore, we employ stringent purity specifications and rigorous QC labs to guarantee that every batch of ligand meets the exacting standards required for high-selectivity catalysis.
We invite you to explore how our expertise can optimize your manufacturing operations and drive down your overall production costs. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific process requirements, helping you identify the most efficient route to market. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you unlock the full potential of this innovative tetradentate phosphine ligand technology and secure a competitive advantage in the global fine chemicals market.
