Scalable Manufacturing of Phenoxybenzamine Hydrochloride via Novel Six-Step Synthetic Route
Introduction to Advanced Phenoxybenzamine Hydrochloride Manufacturing
The pharmaceutical industry continuously demands more efficient and safer pathways for producing critical active pharmaceutical ingredients and their intermediates. Patent CN102887830B discloses a robust preparation method for Phenoxybenzamine Hydrochloride, a vital alpha-adrenergic blocking agent used in treating peripheral vasospasm and benign prostatic hyperplasia. This novel synthetic strategy addresses historical limitations by utilizing a six-step sequence that prioritizes atom economy and operational safety. The process begins with the etherification of phenol and proceeds through a sophisticated Mitsunobu coupling to establish the core carbon-nitrogen bond with high regioselectivity. By leveraging mild reaction conditions and avoiding toxic heavy metal catalysts, this methodology represents a significant leap forward in green chemistry for cardiovascular drug intermediates.
Furthermore, the technical specifications outlined in the patent demonstrate exceptional control over impurity profiles, with intermediate purities consistently exceeding 98% HPLC analysis. This level of quality is paramount for downstream processing and final drug substance approval. The route effectively mitigates the risks associated with traditional chlorination methods by deferring the introduction of chlorine until the final step, thereby reducing equipment corrosion and hazardous waste generation. For procurement and supply chain stakeholders, understanding this technological shift is crucial for securing a reliable pharmaceutical intermediates supplier capable of meeting stringent regulatory standards while maintaining cost efficiency.
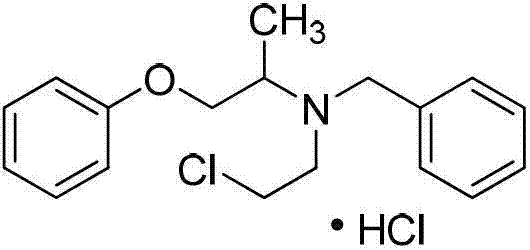
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Phenoxybenzamine Hydrochloride has been plagued by significant safety hazards and inefficiencies that hinder large-scale application. Prior art methods, such as those reported by D.G. Ahern in 1986, relied on the reaction of 1-phenoxy-2-chloropropane with 2-aminoethanol. This approach necessitates the handling of reactive chlorohydrins which are not only difficult to source commercially but also pose substantial toxicity risks to operators. Additionally, alternative routes described in patents like US2599000 require the use of thionyl chloride at multiple stages of the synthesis. The repeated exposure to thionyl chloride drastically increases the potential for safety incidents and generates excessive acidic waste streams that complicate environmental compliance.
Moreover, these conventional pathways often suffer from poor overall yields due to the harsh conditions required for nucleophilic substitutions on secondary carbons. The lack of selectivity in early chlorination steps can lead to complex impurity spectra that are difficult to purge in later stages. Consequently, manufacturers face elevated production costs driven by low throughput and expensive waste treatment protocols. These factors collectively create a bottleneck for the cost reduction in pharmaceutical intermediates manufacturing, making it challenging to secure a stable supply of high-quality material for global markets without incurring prohibitive expenses.
The Novel Approach
In stark contrast, the method disclosed in CN102887830B introduces a paradigm shift by employing a protective group strategy coupled with a Mitsunobu reaction to construct the molecular skeleton. Instead of starting with hazardous chlorinated building blocks, the synthesis initiates with the benign reaction of phenol and methyl oxirane in an aqueous alkaline medium at 45°C. This initial step achieves yields as high as 95% with minimal byproduct formation. The subsequent protection of benzylamine with 2-nitrobenzenesulfonyl chloride allows for precise control over the nitrogen center, preventing unwanted poly-alkylation during the critical coupling phase. This strategic modification ensures that the reaction trajectory remains clean and predictable throughout the sequence.
The core innovation lies in the use of triphenylphosphine and diisopropyl azodicarboxylate (DIAD) to couple the phenoxy alcohol with the protected amine. This Mitsunobu-type transformation proceeds under mild conditions, typically at room temperature, avoiding the thermal degradation often seen in high-temperature substitutions. Following the coupling, the protecting group is removed using thiophenol and potassium carbonate, a process that is both rapid and high-yielding. By reserving the chlorination step for the very end of the synthesis, the process minimizes the time corrosive reagents are in contact with reactor vessels. This holistic redesign results in a streamlined workflow that enhances supply chain reliability and significantly lowers the barrier to entry for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Mitsunobu Coupling and Final Chlorination
The heart of this synthetic route is the third step, where the carbon-nitrogen bond is formed via a Mitsunobu reaction mechanism. In this transformation, the hydroxyl group of 1-phenoxy-2-hydroxypropane is activated by the phosphine-azodicarboxylate system to form a highly reactive oxyphosphonium intermediate. This activation converts the poor leaving group (hydroxyl) into an excellent leaving group, facilitating nucleophilic attack by the sulfonamide nitrogen. The stereochemical outcome and regioselectivity are tightly controlled by the steric environment of the reactants and the specific solvent choice, with tetrahydrofuran proving optimal for solubility and reaction rate. This mechanism bypasses the need for pre-activating the alcohol as a halide, thereby eliminating a whole unit operation and its associated waste.
The final conversion of the hydroxyethyl intermediate to the chloroethyl hydrochloride salt is equally critical for product performance. As illustrated in the reaction scheme below, this step involves the simultaneous introduction of the chloride ion and the formation of the hydrochloride salt using thionyl chloride and hydrogen chloride gas in benzene. The reaction is conducted under reflux conditions for approximately 8 hours to ensure complete conversion of the alcohol functionality. The presence of HCl gas helps to protonate the amine nitrogen in situ, preventing side reactions and driving the equilibrium towards the desired salt form. This one-pot chlorination and salt formation strategy simplifies isolation, as the product often precipitates directly or can be easily crystallized from the reaction mixture.
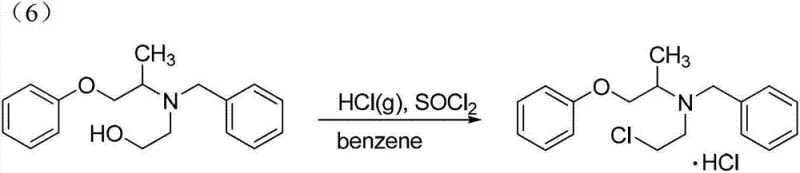
Impurity control is rigorously maintained throughout these mechanistic steps through careful monitoring of reaction parameters. For instance, the deprotection step using thiophenol is monitored by TLC to ensure complete removal of the nitrobenzenesulfonyl group before proceeding. Any residual protecting group could lead to downstream contamination that is difficult to separate. Similarly, the final chlorination is optimized by controlling the addition rate of thionyl chloride to 3ml/min and the HCl gas flow to 1.5L/min. These precise controls prevent local exotherms that could degrade the sensitive amine backbone. The result is a final product with HPLC purity consistently above 98%, meeting the stringent requirements for high-purity pharmaceutical intermediates needed by regulatory bodies worldwide.
How to Synthesize Phenoxybenzamine Hydrochloride Efficiently
Executing this synthesis requires strict adherence to the optimized reaction conditions detailed in the patent to maximize yield and safety. The process is divided into six distinct operational units, each with specific temperature and stoichiometric requirements. For example, the initial etherification must be maintained between 25°C and 65°C, with 45°C being the preferred setpoint to balance reaction rate and selectivity. The subsequent sulfonamide formation is exothermic and benefits from cooling to 0°C during the addition of reagents. Operators should note that the Mitsunobu coupling requires anhydrous conditions, necessitating the use of dried tetrahydrofuran and inert atmosphere protection to prevent hydrolysis of the DIAD reagent.
- React phenol with methyl oxirane using alkali catalyst in water at 45°C to form 1-phenoxy-2-hydroxypropane.
- Condense 2-nitrobenzenesulfonyl chloride with benzylamine in dichloromethane with triethylamine to obtain N-2-nitrobenzenesulfonylbenzylamine.
- Perform Mitsunobu coupling between the phenoxy alcohol and protected amine using PPh3 and DIAD in THF.
- Remove the nitrobenzenesulfonyl protecting group using thiophenol and base in acetonitrile.
- Alkylate the secondary amine with 2-bromoethanol in ethanol at 110°C to introduce the hydroxyethyl chain.
- Convert the terminal hydroxyl group to chloride using thionyl chloride and HCl gas in benzene to yield the final hydrochloride salt.
Following the coupling, the deprotection and alkylation steps utilize common bases like potassium carbonate or sodium carbonate in polar aprotic or protic solvents. The alkylation with 2-bromoethanol is performed at elevated temperatures, specifically 110°C in ethanol, to overcome the activation energy barrier for the nucleophilic substitution. Finally, the chlorination step demands careful handling of corrosive gases and liquids. Detailed standardized synthesis steps see the guide below for specific molar ratios and workup procedures that ensure reproducibility from gram to kilogram scale.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this novel synthetic route offers profound advantages that directly impact the bottom line and supply security. The primary driver for cost reduction in pharmaceutical intermediates manufacturing is the elimination of expensive and hazardous starting materials. By replacing 1-phenoxy-2-chloropropane with commodity chemicals like phenol and propylene oxide, the raw material cost base is significantly lowered. Furthermore, the avoidance of double thionyl chloride usage reduces the consumption of this regulated reagent, lowering both procurement costs and the expenses associated with scrubbing acidic off-gases. These efficiencies translate into a more competitive pricing structure for the final API intermediate without compromising on quality.
Enhanced supply chain reliability is another critical benefit derived from this methodology. The reliance on readily available bulk chemicals ensures that production is not held hostage by the supply constraints of niche specialty reagents. Solvents such as water, ethanol, and dichloromethane are standard in almost every chemical manufacturing facility, reducing the logistical complexity of solvent recovery and recycling. The robustness of the reaction conditions, particularly the tolerance for mild temperatures in the early steps, reduces the risk of batch failures due to thermal runaway. This stability allows for consistent lead times and reduces the need for excessive safety stock, optimizing inventory management for downstream partners.
Scalability and environmental compliance are seamlessly integrated into this process design. The absence of heavy metal catalysts means there is no need for costly metal scavenging steps or rigorous testing for residual metals in the final product. This simplifies the purification train and reduces the volume of solid hazardous waste generated. The use of water as a solvent in the first step is a standout feature of green chemistry, minimizing the organic load in the initial wastewater stream. These factors collectively lower the environmental footprint of the manufacturing process, ensuring alignment with increasingly strict global environmental regulations and facilitating smoother regulatory audits for commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Phenoxybenzamine Hydrochloride using this patented method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation. Understanding these nuances helps stakeholders make informed decisions regarding technology transfer and procurement strategies. The answers reflect the practical realities of scaling this chemistry from the laboratory to industrial reactors.
Q: What are the key advantages of this synthesis method over prior art?
A: This method avoids the use of hazardous 1-phenoxy-2-chloropropane as a starting material and eliminates the need for double thionyl chloride usage found in older routes, significantly improving safety and overall yield.
Q: How is high purity ensured in the final Phenoxybenzamine Hydrochloride product?
A: The process utilizes specific recrystallization steps with petroleum ether and ethyl acetate, along with rigorous column chromatography purification for intermediates, consistently achieving HPLC purity greater than 98% to 99%.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the route employs readily available raw materials like phenol and benzylamine, uses common solvents such as water and ethanol, and operates under mild conditions, making it highly adaptable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenoxybenzamine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving needs of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. Our infrastructure is designed to handle the specific solvent systems and reaction conditions required for this six-step synthesis, guaranteeing a consistent supply of high-quality intermediates.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your projects. Together, we can drive efficiency and reliability in the production of essential cardiovascular medications, ensuring that patients receive the treatments they need without interruption.
