Advanced Chiral Bisphosphorodiamidate Catalysts for Scalable Asymmetric Pharmaceutical Manufacturing
Advanced Chiral Bisphosphorodiamidate Catalysts for Scalable Asymmetric Pharmaceutical Manufacturing
The landscape of asymmetric synthesis is continuously evolving, driven by the relentless demand for high-purity chiral intermediates in the pharmaceutical and agrochemical sectors. A pivotal advancement in this domain is documented in patent CN101885742B, which introduces a novel class of chiral diphosphorodiamidate compounds. These molecules, specifically bis-N-2-[(4S)-4,5-dihydro-4-R-2-oxazolinyl]diphenyl-phenylphosphonamides, represent a significant leap forward in catalyst design, offering superior enantioselectivity and operational stability. Unlike traditional systems that often struggle with reproducibility or metal contamination, this technology leverages a robust phosphorus-nitrogen framework that can be finely tuned through the selection of specific chiral amino alcohol precursors. For R&D directors and process chemists, understanding the structural nuances and synthetic accessibility of these compounds is critical for integrating them into next-generation manufacturing workflows.
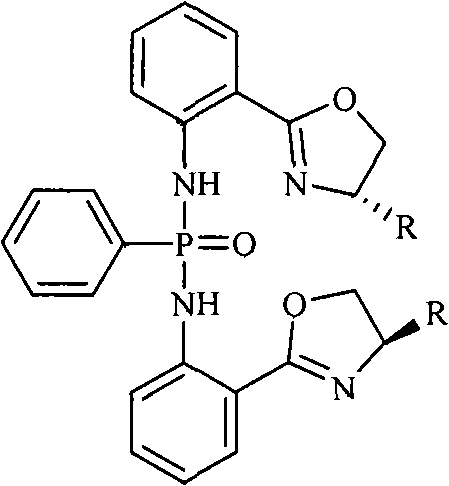
The core innovation lies in the versatility of the R group attached to the oxazoline ring, which can be systematically varied to include isobutyl, isopropyl, phenyl, or benzyl functionalities. This modularity allows process engineers to tailor the steric and electronic environment of the catalyst active site, optimizing performance for a wide array of substrate classes. By establishing a reliable supply chain for these specialized chiral catalysts, manufacturers can significantly mitigate the risks associated with sourcing obscure reagents, thereby ensuring continuity in the production of high-value active pharmaceutical ingredients (APIs). The strategic adoption of such well-defined catalytic systems is essential for maintaining competitive advantage in the global fine chemical market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, asymmetric cyanosilylation and related carbon-carbon bond-forming reactions have relied heavily on transition metal complexes involving titanium, copper, or aluminum. While effective in laboratory settings, these conventional methods present substantial challenges when translated to large-scale industrial operations. The primary concern is the stringent requirement for metal removal, as residual heavy metals can contaminate the final drug substance, necessitating costly and time-consuming purification steps such as scavenging or recrystallization. Furthermore, many traditional chiral ligands are sensitive to moisture and air, requiring rigorous exclusion of atmospheric conditions that drive up operational expenditures and complicate reactor engineering. The instability of these complexes often leads to batch-to-batch variability, undermining the consistency required for regulatory compliance in pharmaceutical manufacturing.
The Novel Approach
The technology disclosed in CN101885742B circumvents these hurdles by introducing a stable, phosphorus-based chiral framework that exhibits remarkable tolerance to reaction conditions. This novel approach utilizes a diphosphorodiamidate backbone that serves as a highly efficient organocatalyst or ligand precursor, delivering exceptional enantiomeric excess values without the immediate need for stoichiometric metal additives in the catalytic cycle. The synthetic route to access these catalysts is streamlined, relying on commodity chemicals like 2-cyanoaniline and readily available L-amino alcohols, which drastically reduces the raw material cost profile compared to exotic chiral pool resources. By shifting towards this more robust chemical architecture, procurement teams can achieve significant cost reduction in asymmetric synthesis manufacturing while simultaneously enhancing the safety and environmental profile of the production process.
Mechanistic Insights into Chiral Phosphorodiamidate Catalysis
The efficacy of these chiral diphosphorodiamidate compounds stems from their ability to activate nucleophiles or electrophiles through precise Lewis acid-base interactions within a rigid chiral pocket. In the context of nitrile silicification reactions, the catalyst likely coordinates with the silicon center of trimethylsilyl cyanide (TMSCN), increasing its nucleophilicity towards the carbonyl substrate. The chiral information encoded in the oxazoline rings creates a differentiated energy landscape for the approaching reagent, favoring the formation of one enantiomer over the other with high fidelity. This mechanism ensures that the resulting cyanohydrin derivatives possess the stereochemical integrity necessary for downstream elaboration into complex therapeutic agents. Understanding this mechanistic pathway is vital for chemists aiming to expand the substrate scope beyond simple aromatic aldehydes to more challenging ketones or aliphatic systems.
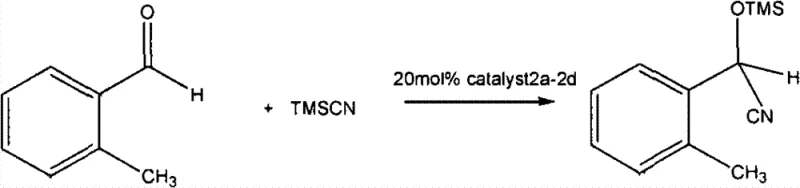
Furthermore, the impurity profile generated during catalysis is inherently cleaner due to the absence of metal-mediated side reactions such as oxidation or radical polymerization that often plague transition metal systems. The stability of the P-N bond under the reaction conditions described (20-30°C) ensures that the catalyst remains intact throughout the transformation, minimizing the generation of degradation byproducts that could co-elute with the desired product. This high level of chemoselectivity translates directly into simplified isolation protocols, where standard aqueous workups and chromatography suffice to obtain analytical grade materials. For quality control laboratories, this means reduced analytical burden and faster release times for intermediate batches, facilitating a more agile response to market demands for chiral building blocks.
How to Synthesize Chiral Bisphosphorodiamidate Efficiently
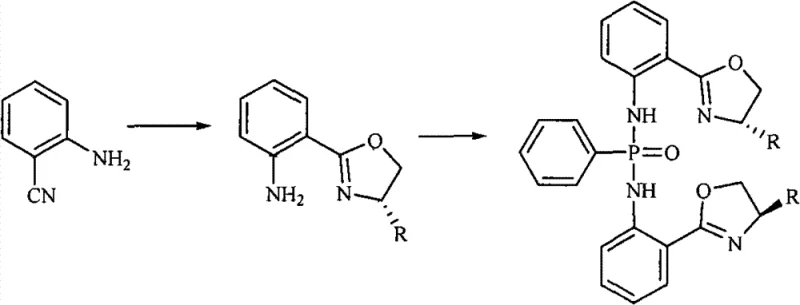
The preparation of these high-performance catalysts follows a logical two-step sequence that balances yield with optical purity, making it amenable to both laboratory optimization and plant-scale execution. The initial phase involves the condensation of 2-cyanoaniline with a chosen L-amino alcohol in the presence of a zinc chloride promoter, establishing the chiral oxazoline core with retention of configuration. Following isolation and purification of this key intermediate, the second step entails phosphorylation using phenylphosphonic dichloride under basic conditions to install the central phosphorus bridge. This modular synthesis allows for the rapid generation of a library of catalyst variants by simply swapping the amino alcohol starting material, providing R&D teams with a versatile toolkit for reaction screening. Detailed standardized operating procedures for each unit operation are outlined below to ensure reproducibility.
- React 2-cyanoaniline with L-amino alcohol (e.g., L-leucinol) using anhydrous ZnCl2 catalyst in chlorobenzene under reflux for 24 hours to form the oxazoline intermediate.
- Purify the intermediate via aqueous workup, chloroform extraction, and column chromatography to ensure high optical purity.
- Condense the purified intermediate with phenylphosphonic dichloride in a toluene and triethylamine mixture under reflux for 24 hours to yield the target chiral catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this catalytic technology offers profound benefits for supply chain resilience and overall cost structure optimization. The reliance on bulk commodity chemicals such as anilines and amino alcohols means that raw material availability is not constrained by the geopolitical or agricultural fluctuations that often impact natural product-derived chiral sources. This stability allows procurement managers to negotiate long-term contracts with fixed pricing, shielding the organization from volatile market spikes. Moreover, the synthetic pathway avoids the use of precious metals like palladium or rhodium in the final catalyst structure, eliminating the financial risk associated with fluctuating noble metal prices and the logistical complexity of recycling spent catalysts. These factors collectively contribute to a more predictable and manageable cost of goods sold (COGS) for the final pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive transition metals and the use of standard reflux conditions significantly lower the capital and operational expenditure required for production facilities. By removing the need for specialized metal-scavenging resins and extensive purification trains, manufacturers can realize substantial cost savings in downstream processing. The high turnover frequency of the catalyst further reduces the loading required per batch, amplifying the economic efficiency of the process. Additionally, the robustness of the catalyst minimizes batch failures due to sensitivity, ensuring consistent throughput and maximizing asset utilization rates across the manufacturing plant.
- Enhanced Supply Chain Reliability: Sourcing strategies are simplified as the key precursors are widely produced by multiple global suppliers, reducing the risk of single-source dependency. The synthetic route does not rely on cryogenic reagents or unstable intermediates that require cold-chain logistics, thereby streamlining transportation and storage requirements. This inherent stability ensures that inventory can be held for longer periods without degradation, providing a buffer against supply disruptions. Consequently, supply chain heads can maintain leaner inventory levels while still guaranteeing continuity of supply to downstream API manufacturers, enhancing overall network agility.
- Scalability and Environmental Compliance: The process operates in common organic solvents like toluene and chlorobenzene, which are well-understood in terms of waste management and solvent recovery infrastructure. The absence of heavy metal waste streams simplifies environmental compliance and reduces the burden on wastewater treatment facilities. Scaling from pilot to commercial production is straightforward as the reaction kinetics do not change drastically with volume, allowing for direct technology transfer without extensive re-optimization. This ease of scale-up accelerates time-to-market for new drug candidates relying on this chemistry, providing a competitive edge in the fast-paced pharmaceutical industry.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of integrating this technology into their existing portfolios, we have compiled answers to common inquiries regarding performance and handling. These insights are derived directly from the experimental data and structural analysis provided in the patent literature, ensuring accuracy and relevance. Addressing these questions early in the development cycle helps mitigate technical risks and aligns expectations between R&D and commercial teams. We encourage stakeholders to review these details thoroughly to understand the full potential of this catalytic system.
Q: What represents the primary advantage of this diphosphorodiamidate catalyst over traditional metal complexes?
A: This catalyst system offers exceptional enantioselectivity (up to 99% ee) in cyanosilylation reactions while potentially simplifying downstream purification by reducing reliance on heavy metal residues often associated with traditional transition metal catalysts.
Q: Is the synthesis of this catalyst scalable for industrial production?
A: Yes, the synthetic route utilizes standard reflux conditions in common solvents like chlorobenzene and toluene, avoiding cryogenic temperatures or ultra-high pressure, which facilitates straightforward commercial scale-up from kilogram to multi-ton quantities.
Q: What represent the key structural variables (R groups) available for this catalyst family?
A: The catalyst structure allows for tunability via the R group on the oxazoline ring, which can be selected from isobutyl, isopropyl, phenyl, or benzyl moieties derived from corresponding L-amino alcohols to optimize steric environment for specific substrates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Diphosphorodiamidate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced catalytic technologies play in accelerating drug discovery and development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly transitioned into viable industrial processes. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of chiral catalyst meets the highest international standards. Our commitment to quality and consistency makes us a trusted partner for multinational corporations seeking to secure their supply chains for complex chiral intermediates.
We invite you to engage with our technical procurement team to discuss how this specific catalytic route can be optimized for your unique application needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper visibility into the potential economic benefits of switching to this methodology. We are prepared to provide specific COA data and route feasibility assessments to support your internal validation processes. Let us collaborate to engineer a more efficient and sustainable future for your chemical manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
