Advanced Copper-Catalyzed Synthesis of 2,3-Diaminopyridine for Commercial API Manufacturing
Advanced Copper-Catalyzed Synthesis of 2,3-Diaminopyridine for Commercial API Manufacturing
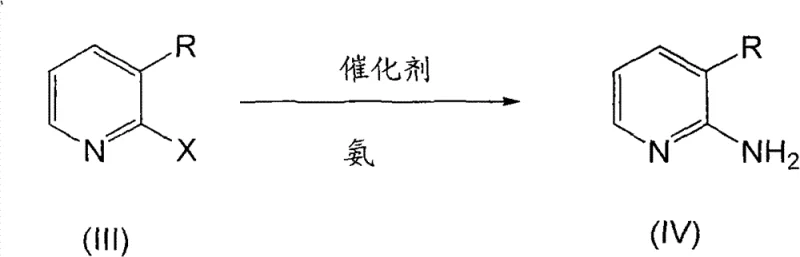
The pharmaceutical industry continuously demands robust and scalable synthetic routes for critical heterocyclic intermediates, particularly those serving as building blocks for complex active pharmaceutical ingredients. Patent CN101723885B introduces a transformative methodology for the preparation of 2,3-diaminopyridine and its derivatives, addressing long-standing challenges in yield optimization and impurity control. This technology leverages a copper-catalyzed amination strategy that replaces traditional noble metal systems, offering a pathway to high-purity products exceeding 99.5% HPLC purity. For R&D directors and procurement specialists, this represents a significant opportunity to streamline supply chains for ulcer medications and inotropic agents. The process utilizes readily available starting materials and operates under conditions that are compatible with large-scale industrial autoclaves, ensuring that the transition from laboratory bench to commercial production is seamless and economically viable.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of diaminopyridine derivatives has relied heavily on hydrogenation protocols utilizing palladium or Raney nickel catalysts, which introduce substantial operational complexities and cost burdens. These conventional methods often require multiple reaction steps, including protection and deprotection sequences, to achieve acceptable selectivity, thereby increasing the overall processing time and solvent consumption. Furthermore, the use of hazardous solvents and the necessity for rigorous removal of trace heavy metals like palladium create significant bottlenecks in regulatory compliance and quality control workflows. The reliance on expensive noble metals not only inflates the raw material costs but also poses supply chain risks due to the geopolitical volatility associated with platinum group metals. Additionally, traditional extraction processes frequently involve toxic organic solvents that complicate waste management and environmental safety protocols, making them less attractive for modern green chemistry initiatives.
The Novel Approach
The innovative process disclosed in the patent data circumvents these issues by employing a direct amination of 3-amino-2-halopyridines using aqueous ammonia in the presence of a cost-effective copper catalyst. This approach simplifies the reaction pathway to a single pot operation, eliminating the need for complex multi-step sequences and reducing the overall footprint of the manufacturing facility. By operating at moderate temperatures between 100°C and 150°C and utilizing self-generated pressure, the method ensures high conversion rates while maintaining safety standards suitable for large-scale production. The substitution of noble metals with copper salts, such as cuprous sulfate, drastically reduces the catalyst cost and simplifies the downstream purification process, as copper residues are easier to manage and remove compared to palladium. This novel route also integrates a recyclable ammonia system, which enhances the sustainability profile of the manufacturing process and aligns with global trends towards greener chemical production.
Mechanistic Insights into Copper-Catalyzed Amination
The core of this technological advancement lies in the nucleophilic aromatic substitution mechanism facilitated by the copper catalyst, which activates the halopyridine substrate towards attack by ammonia. The copper species coordinates with the nitrogen atoms of the pyridine ring and the incoming ammonia, lowering the activation energy required for the displacement of the halogen atom. This catalytic cycle is highly efficient, allowing the reaction to proceed to completion within 7 to 10 hours under controlled pressure conditions of 18 to 25 kg/cm2. The use of aqueous ammonia serves a dual purpose, acting both as the nitrogen source and as a medium that facilitates the solubility of the inorganic copper salts, ensuring homogeneous catalysis throughout the reaction mixture. The specific selection of copper salts, particularly cuprous sulfate, optimizes the electronic environment around the reaction center, minimizing side reactions such as dehalogenation or polymerization that often plague high-temperature amination processes.
Impurity control is meticulously managed through the thermodynamic parameters of the reaction and the subsequent workup procedure, ensuring that the final product meets stringent pharmaceutical specifications. The reaction conditions are tuned to suppress the formation of regio-isomers and over-aminated byproducts, which are common challenges in pyridine chemistry. Following the reaction, the crude mixture undergoes a single-step extraction using Class III solvents like ethyl acetate, which are recognized for their low toxicity and favorable environmental profile. This extraction strategy effectively separates the organic product from inorganic salts and catalyst residues, yielding a crude material that is already of high purity. The ability to achieve greater than 99.5% purity without the need for column chromatography or recrystallization from hazardous solvents is a testament to the selectivity of the copper-catalyzed system. This level of purity is critical for downstream applications where trace impurities could affect the efficacy or safety of the final drug substance.
How to Synthesize 2,3-Diaminopyridine Efficiently
The implementation of this synthesis route requires precise control over reaction parameters to maximize yield and ensure operator safety during the high-pressure amination step. The process begins with the charging of 3-amino-2-halopyridine, the copper catalyst, and aqueous ammonia into a pressure-rated autoclave, followed by the application of initial ammonia pressure to establish the reaction environment. Detailed standard operating procedures regarding temperature ramping, pressure monitoring, and venting protocols are essential to replicate the success reported in the patent examples consistently. The subsequent workup involves careful phase separation and solvent recovery, which are critical for maintaining the economic advantages of the process. For a comprehensive understanding of the specific operational parameters and safety guidelines, please refer to the standardized synthesis steps provided in the technical guide below.
- Charge 3-amino-2-halopyridine, copper catalyst (e.g., CuSO4), and aqueous ammonia into a high-pressure autoclave.
- Heat the reaction mixture to 100-150°C under controlled ammonia pressure to facilitate nucleophilic substitution.
- Perform a single-step extraction using Class III solvents like ethyl acetate to isolate the product with >99.5% purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the shift from noble metal catalysts to copper-based systems offers substantial cost reduction in pharmaceutical intermediate manufacturing by lowering the bill of materials significantly. The elimination of expensive palladium or platinum catalysts removes a major variable cost driver, allowing for more stable pricing models and improved margin protection for the final API. Furthermore, the use of recyclable ammonia and Class III solvents reduces the consumption of raw materials and minimizes waste disposal costs, contributing to a leaner and more efficient production model. The simplified workup procedure, which avoids complex purification steps, translates directly into reduced labor hours and increased throughput capacity for the manufacturing plant. These factors combined create a robust economic case for adopting this technology, particularly for high-volume production runs where marginal savings per kilogram accumulate into significant financial benefits.
- Cost Reduction in Manufacturing: The replacement of precious metal catalysts with abundant copper salts fundamentally alters the cost structure of the synthesis, removing the dependency on volatile commodity markets for palladium. This transition not only lowers the direct material costs but also reduces the capital expenditure associated with metal recovery systems, which are often required to reclaim expensive catalysts from waste streams. Additionally, the high selectivity of the reaction minimizes the loss of starting materials to byproducts, improving the overall atom economy and further driving down the cost per unit of the active intermediate. The ability to reuse solvents like ethyl acetate through distillation adds another layer of cost efficiency, ensuring that operational expenses remain predictable and controlled over the long term.
- Enhanced Supply Chain Reliability: Utilizing widely available raw materials such as copper sulfate and aqueous ammonia mitigates the risk of supply disruptions that are often associated with specialized reagents or imported catalysts. The robustness of the reaction conditions allows for flexible manufacturing scheduling, as the process is less sensitive to minor variations in reagent quality compared to sensitive hydrogenation reactions. This reliability ensures consistent delivery timelines for downstream API manufacturers, reducing the need for excessive safety stock and enabling a more responsive just-in-time inventory management strategy. The scalability of the process from pilot plant to commercial scale further guarantees that supply can be ramped up quickly to meet surges in market demand without compromising product quality.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing standard high-pressure equipment that is commonly available in fine chemical manufacturing facilities, thereby reducing the need for specialized infrastructure investment. The use of environmentally benign solvents and the recycling of ammonia align with strict environmental regulations, reducing the regulatory burden and potential liabilities associated with hazardous waste generation. This compliance facilitates smoother audits and approvals from regulatory bodies, accelerating the time to market for new drug formulations that rely on this intermediate. The reduced environmental footprint also enhances the corporate social responsibility profile of the supply chain, which is increasingly important for partnerships with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed amination technology in industrial settings. These answers are derived directly from the patent specifications and are intended to provide clarity on the feasibility and benefits of the process for potential partners. Understanding these details is crucial for making informed decisions about process adoption and supply chain integration. For further technical discussions or specific data requests, our team is available to provide detailed route feasibility assessments.
Q: What are the primary advantages of the copper-catalyzed method over palladium-based routes?
A: The copper-catalyzed method eliminates the need for expensive noble metal catalysts like palladium, significantly reducing raw material costs and simplifying the removal of trace metal impurities from the final API intermediate.
Q: How does this process ensure high purity suitable for pharmaceutical applications?
A: By utilizing a single-step extraction with environmentally benign Class III solvents and controlled reaction temperatures, the process achieves HPLC purity greater than 99.5% without complex chromatographic purification.
Q: Is the ammonia used in this synthesis recyclable for industrial scale-up?
A: Yes, the patent explicitly describes a system where excess ammonia gas is vented, scrubbed, and recycled back into the process, enhancing both environmental compliance and operational cost efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Diaminopyridine Supplier
NINGBO INNO PHARMCHEM stands at the forefront of chemical manufacturing innovation, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at translating complex patent methodologies like the copper-catalyzed amination of halopyridines into robust, GMP-compliant manufacturing processes. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of 2,3-diaminopyridine meets the exacting standards required for pharmaceutical applications. Our commitment to quality and consistency makes us an ideal partner for companies seeking to secure a stable supply of critical intermediates for their drug development pipelines.
We invite you to collaborate with us to optimize your supply chain and achieve significant operational efficiencies through the adoption of advanced synthetic routes. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your long-term strategic goals. Let us help you engineer a more resilient and cost-effective supply chain for your most valuable pharmaceutical assets.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
