Advanced Synthesis of Nitrogen-Containing Aromatic Heterocyclic Diamines for High-Performance Polymers
The landscape of high-performance polymer manufacturing is undergoing a significant transformation driven by innovations in intermediate synthesis, specifically highlighted by the technological breakthroughs detailed in Chinese Patent CN112898198B. This patent introduces a robust and versatile methodology for the preparation of nitrogen-containing aromatic heterocyclic nitro compounds, which serve as critical precursors for advanced polyimides and polyamides. These materials are indispensable in demanding sectors such as microelectronics, aerospace, and functional protection due to their superior thermal stability and mechanical properties. The core innovation lies in a streamlined synthetic route that bypasses the limitations of traditional nitration methods, offering a pathway to structurally diverse heterocyclic diamines with exceptional purity and yield. For industry leaders seeking a reliable polymer additive supplier, understanding the nuances of this Suzuki-coupling-based approach is essential for securing a competitive edge in the supply of next-generation electronic chemicals.
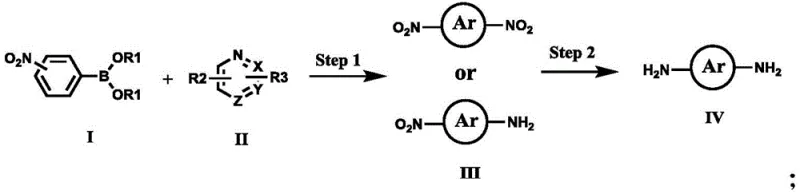
The strategic value of this patent extends beyond mere chemical novelty; it addresses fundamental bottlenecks in the supply chain of high-value monomers. By enabling the synthesis of complex nitrogen-containing heterocycles through a modular coupling strategy, the technology significantly enhances the feasibility of producing custom-tailored polymers. The ability to access a wide array of substitution patterns on the heterocyclic core allows material scientists to fine-tune the glass transition temperature, solubility, and dielectric constants of the final polymer. Consequently, this method represents a pivotal advancement for companies focused on cost reduction in high polymer materials manufacturing, as it simplifies the overall process flow and reduces the reliance on hazardous reagents typically associated with direct nitration processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing aromatic heterocyclic nitro compounds has been plagued by significant technical hurdles that hinder efficient large-scale production. Traditional routes often involve direct nitration of the heterocyclic ring, a process that is notoriously difficult to control due to the electron-deficient nature of many nitrogen heterocycles. This frequently leads to poor regioselectivity, resulting in complex mixtures of isomers that are costly and time-consuming to separate. Furthermore, the harsh acidic conditions required for direct nitration can cause decomposition of sensitive heterocyclic frameworks, leading to low overall yields and the generation of substantial hazardous waste. These inefficiencies not only drive up the cost of goods but also create supply chain vulnerabilities, making it challenging for manufacturers to guarantee the consistent quality and availability of high-purity nitrogen-containing aromatic heterocyclic diamine precursors required for high-end applications.
The Novel Approach
In stark contrast, the methodology disclosed in CN112898198B employs a sophisticated Suzuki-Miyaura cross-coupling reaction to construct the carbon-carbon bonds between the nitro-substituted phenyl ring and the heterocyclic core. This approach fundamentally shifts the paradigm from destructive functionalization to constructive assembly. By utilizing nitro-substituted phenylboronic acids or their esters as coupling partners, the process achieves high regioselectivity and tolerance for various functional groups. The reaction conditions are remarkably mild, typically operating between 50°C and 150°C in biphasic solvent systems such as toluene/water or dioxane/water. This gentler chemistry preserves the integrity of the heterocyclic ring while ensuring high conversion rates. The subsequent reduction of the nitro group to an amine is equally optimized, utilizing catalytic hydrogenation or hydrazine reduction to deliver the final diamine monomers with exceptional purity, thereby facilitating the commercial scale-up of complex polyimide monomers.
Mechanistic Insights into Ni/Pd-Catalyzed Suzuki Coupling
The heart of this innovative synthesis lies in the transition metal-catalyzed cross-coupling mechanism, which elegantly joins the aromatic building blocks. The reaction initiates with the oxidative addition of the metal catalyst, preferably a nickel or palladium complex such as NiCl2(dppp) or Pd(PPh3)4, into the carbon-halogen bond of the heterocyclic substrate. This step generates a reactive organometallic intermediate that is poised for transmetallation. The nitro-substituted phenylboronic acid, activated by the base (e.g., K2CO3 or Cs2CO3), transfers its aryl group to the metal center. This transmetallation step is critical and is facilitated by the specific choice of ligand and solvent system, which stabilizes the catalytic cycle and prevents catalyst deactivation. Finally, reductive elimination occurs to forge the new carbon-carbon bond, releasing the coupled nitro-heterocycle product and regenerating the active catalyst species. This mechanistic precision ensures that even sterically hindered or electronically deactivated substrates can be coupled efficiently.
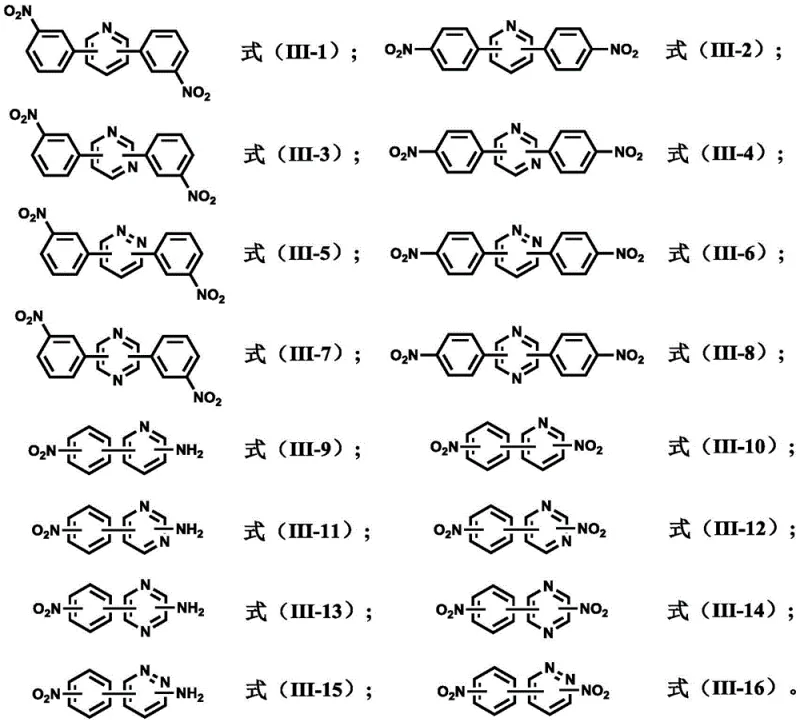
Beyond the coupling mechanism, the control of impurities is paramount for producing monomers suitable for electronic-grade polymers. The patent details a rigorous purification protocol that leverages the differential solubility of the product in aqueous and organic phases. Post-reaction, the mixture is cooled to precipitate the crude solid, which is then washed sequentially with water, toluene, and methanol. This multi-solvent washing strategy effectively removes residual metal catalysts, inorganic salts, and unreacted starting materials. The use of specific bases like potassium carbonate not only drives the coupling reaction but also aids in neutralizing acidic byproducts. Furthermore, the subsequent reduction step is carefully monitored, with options for catalytic hydrogenation using Pd/C or Raney Ni, or chemical reduction using stannous chloride. Each reduction pathway is optimized to minimize the formation of azo or hydroxylamine byproducts, ensuring that the final diamine possesses the stringent purity specifications required for high-performance polyimide synthesis.
How to Synthesize Nitrogen-Containing Aromatic Heterocyclic Diamines Efficiently
The practical implementation of this synthesis route requires careful attention to reaction parameters to maximize yield and reproducibility. The process begins with the selection of appropriate starting materials, specifically matching the halogenated heterocycle with the corresponding nitro-phenylboronic acid derivative. The reaction is conducted under an inert atmosphere, typically nitrogen or argon, to prevent oxidation of the sensitive metal catalyst. Solvent selection is crucial, with a preference for mixtures like toluene and water which allow for easy phase separation during workup. The molar ratios of reactants are optimized, often using a slight excess of the boronic acid to drive the reaction to completion. Following the coupling, the isolation of the nitro-intermediate is straightforward, often involving simple filtration. The final reduction step can be tailored based on the specific substrate; for instance, hydrazine hydrate is excellent for laboratory scale, while catalytic hydrogenation is preferred for larger batches due to cleaner byproduct profiles. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results.
- Perform a metal-catalyzed cross-coupling reaction between a nitro-substituted phenylboronic acid (ester) and a halogenated nitrogen-containing aromatic heterocycle using a Nickel, Palladium, or Copper catalyst in a biphasic solvent system.
- Isolate and purify the resulting nitrogen-containing aromatic heterocyclic nitro compound intermediate through filtration, washing, and recrystallization.
- Subject the purified nitro intermediate to a reduction reaction using catalytic hydrogenation, hydrazine hydrate, or stannous chloride to yield the final diamine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic methodology offers profound strategic benefits that extend well beyond the laboratory. The shift from multi-step, low-yield traditional syntheses to this streamlined coupling-reduction sequence translates directly into enhanced supply chain reliability and cost efficiency. By utilizing widely available commodity chemicals like nitro-phenylboronic acids and chlorinated heterocycles, manufacturers can mitigate the risks associated with sourcing exotic or proprietary intermediates. This modularity allows for rapid adjustment of production schedules in response to market demand fluctuations, ensuring a steady flow of critical monomers for downstream polymerization processes. Furthermore, the simplified workup procedures reduce the burden on waste treatment facilities, aligning with increasingly strict environmental regulations and sustainability goals.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the elimination of expensive and hazardous nitration reagents and the associated safety infrastructure. The use of earth-abundant nickel catalysts, alongside highly efficient palladium systems, lowers the raw material cost per kilogram of product. Additionally, the high yields reported in the patent examples mean that less raw material is wasted, and the throughput of existing reactor volumes is significantly increased. The simplified purification process reduces solvent consumption and energy usage during distillation and drying, contributing to a substantially lower cost of goods sold without compromising on the quality of the final diamine.
- Enhanced Supply Chain Reliability: One of the most compelling advantages is the robustness of the supply chain enabled by this chemistry. Because the starting materials are common industrial chemicals, the risk of supply disruption due to the scarcity of a single specialized precursor is minimized. The broad substrate scope demonstrated in the patent means that if one specific heterocycle becomes unavailable, alternative structures with similar properties can often be synthesized using the same platform technology. This flexibility ensures reducing lead time for high-purity electronic chemicals and provides a buffer against geopolitical or logistical disruptions that often plague the fine chemical industry.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial production in mind. The reaction conditions avoid extreme pressures or cryogenic temperatures, allowing for the use of standard stainless steel reactors found in most fine chemical plants. The aqueous workup minimizes the volume of organic waste, and the potential for solvent recycling further enhances the environmental profile. This compliance with green chemistry principles not only reduces disposal costs but also future-proofs the manufacturing process against tightening global environmental standards, making it a sustainable choice for long-term production.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this patent, we have compiled a set of frequently asked questions based on the specific embodiments and claims within the document. These answers address common concerns regarding catalyst selection, substrate compatibility, and process safety. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this technology into their existing manufacturing lines. The following insights are derived directly from the experimental data and theoretical framework presented in the patent, ensuring accuracy and relevance for technical decision-makers.
Q: What are the primary advantages of this Suzuki coupling method over traditional nitration routes?
A: This method avoids the harsh conditions and regioselectivity issues associated with direct nitration of heterocycles. It allows for the modular assembly of complex structures using readily available boronic acids and halogenated heterocycles, resulting in higher yields and simpler purification.
Q: Which catalysts are most effective for this specific transformation?
A: The patent highlights the effectiveness of Nickel catalysts such as NiCl2(dppp) and NiCl2(DME), as well as Palladium catalysts like Pd(PPh3)4. These catalysts facilitate the coupling under relatively mild temperatures (50-150°C) with high turnover.
Q: Can this process be scaled for industrial production of polyimide monomers?
A: Yes, the process is explicitly designed for large-scale industrial production. It utilizes common solvents like toluene and water, operates at manageable pressures for the reduction step, and offers a wide substrate scope suitable for producing various high-performance polymer precursors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nitrogen-Containing Aromatic Heterocyclic Nitro Compound Supplier
The technological advancements described in CN112898198B represent a significant leap forward in the synthesis of high-performance polymer precursors, yet translating this potential into commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. NINGBO INNO PHARMCHEM stands at the forefront of this industry, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of Suzuki coupling and catalytic reduction processes, ensuring that every batch meets stringent purity specifications. With our rigorous QC labs and commitment to process optimization, we guarantee the delivery of high-quality intermediates that empower your downstream polymerization efforts.
We invite you to explore how our expertise can enhance your supply chain and reduce your overall manufacturing costs. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments for your target molecules. By partnering with us, you secure not just a supplier, but a strategic ally dedicated to driving innovation and efficiency in your high polymer materials manufacturing.
