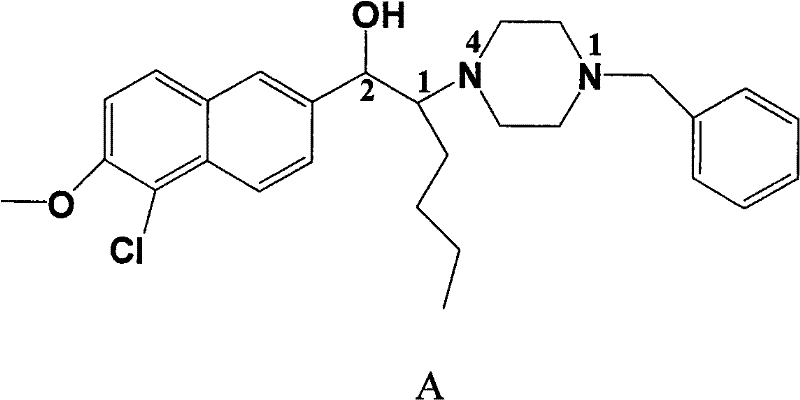
Scalable Chiral Resolution of Piperazine Derivatives for High-Purity Antidepressant Intermediates
The pharmaceutical industry continuously seeks robust methodologies for producing single-enantiomer active pharmaceutical ingredients (APIs) to ensure safety and efficacy. Patent CN102050800A introduces a transformative approach for preparing the optical isomers of 1-butyl-2-hydroxyarylalkanol piperazine derivatives, specifically targeting the potent triple reuptake inhibitor known as Compound A. This compound, characterized by its specific naphthyl-piperazine scaffold, exhibits significant antidepressant activity through the selective inhibition of 5-HT, NA, and DA reuptake. The patent discloses a novel chemical resolution technique that bypasses the limitations of previous asymmetric synthesis routes, offering a pathway to high-purity (1S,2S) and (1R,2R) isomers essential for clinical development.
Historically, the production of such complex chiral molecules relied on constructing the stereocenter from the beginning using chiral pool materials. However, the prior art described in CN200810043821.8 utilized a prochiral synthesis starting from chiral nor-leucine. While theoretically sound, this legacy approach suffers from severe practical drawbacks that hinder commercial viability. The synthetic route is excessively long, requiring protection and deprotection steps that accumulate waste and reduce throughput. Furthermore, the process demands stringent anhydrous conditions and employs intermediates with significant cytotoxicity, posing safety risks for operators and requiring specialized containment infrastructure.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The conventional asymmetric synthesis pathway involves a cumbersome seven-step sequence initiating from chiral nor-leucine, where the amino group is protected via phthaloyl groups and the carboxyl group is activated using oxalyl chloride. This multi-step progression not only extends the lead time for batch production but also results in a cumulative yield that is critically low, often reported at no more than 1%. Such inefficiency translates directly into exorbitant production costs, with historical data indicating prices reaching approximately 150,000 RMB per kilogram, rendering the material inaccessible for large-scale pharmaceutical applications. Additionally, the reliance on column chromatography for final purification creates a bottleneck that is difficult to translate from laboratory glassware to multi-ton industrial reactors.
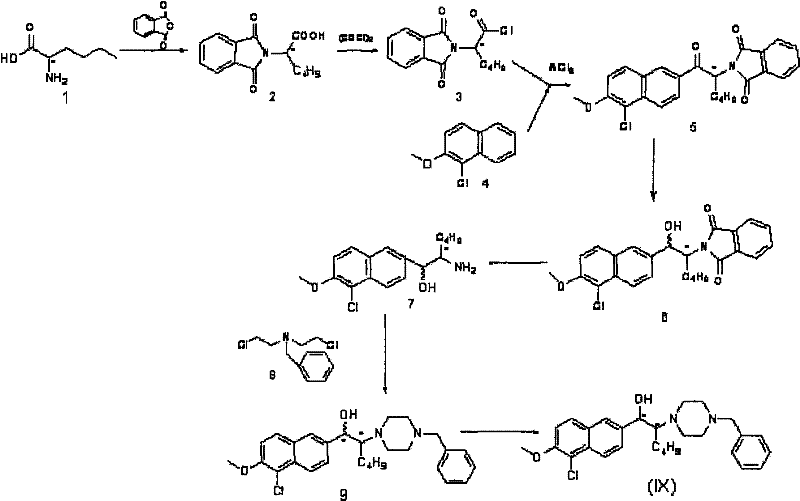
The Novel Approach
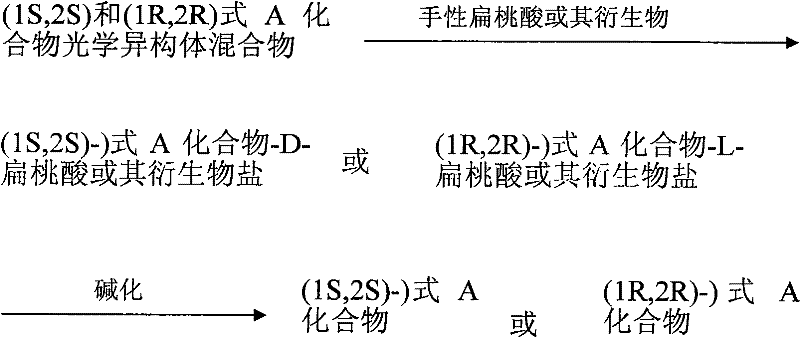
In stark contrast, the methodology presented in CN102050800A adopts a classical yet highly effective chemical resolution strategy. Instead of building chirality atom-by-atom, this process starts with the racemic mixture of the target piperazine derivative and separates the enantiomers using chiral mandelic acid or its derivatives. This shift fundamentally changes the economic equation by allowing the use of cheaper racemic starting materials and simplifying the purification to a crystallization and filtration operation. The reaction scheme involves dissolving the racemate and the resolving agent in a solvent, allowing the less soluble diastereomeric salt to precipitate, and subsequently liberating the free base through alkalization. This approach dramatically shortens the critical path for manufacturing and improves the overall process mass intensity.
Mechanistic Insights into Diastereomeric Salt Resolution
The core of this technological advancement lies in the thermodynamic stability differences between the diastereomeric salts formed during the resolution process. When the racemic piperazine base interacts with a single enantiomer of a chiral acid, such as R-(-)-mandelic acid, two distinct salts are generated: one comprising the (1S,2S)-base and the R-acid, and the other comprising the (1R,2R)-base and the R-acid. These salts possess different physical properties, most notably solubility in specific organic solvents like ethyl acetate or ethanol. By carefully controlling parameters such as temperature, solvent composition, and molar ratios, the process selectively precipitates the desired salt while keeping the unwanted isomer in solution.
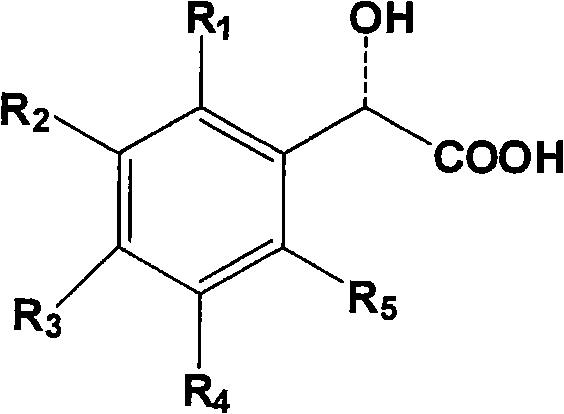
The versatility of this method is further enhanced by the tunability of the resolving agent. The patent outlines a broad scope of mandelic acid derivatives that can be employed, defined by a general formula where substituents on the phenyl ring modulate the acidity and steric bulk of the acid. Derivatives such as o-chloromandelic acid, p-chloromandelic acid, and various halogenated analogues provide a toolkit for optimizing the resolution efficiency. For instance, the electron-withdrawing effects of chlorine or fluorine atoms can alter the pKa of the acid, thereby strengthening the ionic interaction with the basic piperazine nitrogen and potentially improving the crystallinity of the resulting salt. This mechanistic flexibility allows process chemists to tailor the resolution conditions to achieve optical purities exceeding 99% as confirmed by chiral HPLC analysis.
How to Synthesize N1-benzyl-N4-[1-butyl-2-(5'-chloro-6'-methoxy-2'-naphthyl) hydroxyethyl] piperazine Efficiently
The synthesis of these high-value optical isomers begins with the preparation of the racemic precursor, which can be sourced from established methods, followed by the critical resolution step. The process is designed to be operationally simple, requiring standard reactor equipment capable of heating, cooling, and solid-liquid separation. The key to success lies in the selection of the appropriate solvent system and the precise control of the crystallization kinetics to maximize the yield of the target diastereomer. Detailed standardized synthesis steps see the guide below.
- Dissolve the racemic mixture of the piperazine derivative and a chiral mandelic acid resolving agent in a suitable solvent such as ethyl acetate or ethanol under heated conditions.
- Allow the solution to cool and stand to facilitate the precipitation of the specific diastereomeric salt, then separate the crystals via filtration or centrifugation.
- Treat the isolated salt with an alkali solution in an organic solvent to liberate the free base, followed by separation and solvent removal to obtain the pure optical isomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from asymmetric synthesis to chemical resolution represents a significant opportunity for cost optimization and risk mitigation. The elimination of expensive chiral starting materials, such as protected amino acids, removes a major cost driver from the bill of materials. Furthermore, the simplification of the downstream processing removes the need for preparative chromatography, which is often a resource-intensive and time-consuming operation in fine chemical manufacturing. This streamlining of the workflow directly contributes to substantial cost savings and enhances the overall economic feasibility of the project.
- Cost Reduction in Manufacturing: The new resolution method eliminates the need for costly chiral auxiliaries and complex protection group chemistry, leading to a drastic reduction in raw material expenses. By replacing a low-yielding seven-step sequence with a high-efficiency crystallization process, the overall consumption of solvents and reagents is significantly minimized. This efficiency gain allows for a much lower cost of goods sold, making the final API more competitive in the global market without compromising on quality standards.
- Enhanced Supply Chain Reliability: Sourcing complex chiral building blocks often involves long lead times and reliance on specialized suppliers, creating potential bottlenecks. The resolution approach utilizes commodity chemicals like racemic intermediates and widely available mandelic acid derivatives, which are readily accessible from multiple vendors. This diversification of the supply base ensures continuity of supply and reduces the risk of production delays caused by raw material shortages, thereby stabilizing the manufacturing schedule.
- Scalability and Environmental Compliance: The process avoids the use of highly toxic intermediates associated with the previous synthetic route, improving the safety profile of the manufacturing facility. The reliance on crystallization and filtration rather than chromatography makes the process inherently easier to scale from pilot plant to commercial production volumes. Additionally, the reduced solvent usage and simpler waste streams facilitate better environmental compliance and lower waste disposal costs, aligning with modern green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of these piperazine derivatives. The answers are derived directly from the experimental data and process descriptions found in the patent literature, providing clarity on the feasibility and advantages of the resolution method.
Q: Why is the chiral resolution method preferred over asymmetric synthesis for this compound?
A: The conventional asymmetric synthesis involves a lengthy seven-step route with hazardous reagents and extremely low overall yields below 1%, whereas the resolution method utilizes readily available racemates and achieves high optical purity greater than 99% with significantly simplified processing.
Q: What resolving agents are effective for separating the (1S,2S) and (1R,2R) isomers?
A: Chiral mandelic acid and its derivatives, such as o-chloromandelic acid, p-chloromandelic acid, and various fluoro- or bromo-substituted mandelic acids, have been proven effective in forming separable diastereomeric salts with high enantiomeric excess.
Q: How does this process impact the cost of goods for pharmaceutical manufacturing?
A: By eliminating the need for expensive chiral starting materials like protected nor-leucine and avoiding complex column chromatography purification, the resolution process drastically reduces raw material costs and operational complexity, making industrial scale-up economically viable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N1-benzyl-N4-[1-butyl-2-(5'-chloro-6'-methoxy-2'-naphthyl) hydroxyethyl] piperazine Supplier
At NINGBO INNO PHARMCHEM, we understand the critical importance of securing a stable supply of high-quality chiral intermediates for your drug development pipeline. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in pre-clinical trials or full-scale manufacturing. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of optical isomer meets the highest regulatory standards for pharmaceutical use.
We invite you to collaborate with us to leverage this advanced resolution technology for your specific projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your production needs. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your timeline to market while optimizing your manufacturing costs.
