Scalable Synthesis of Axially Chiral Biaryl Lignans for Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing axially chiral biaryl scaffolds, which serve as critical motifs in bioactive natural products and advanced materials. Patent CN108250206B introduces a groundbreaking synthetic strategy for producing biaryl lignan intermediates, specifically targeting high-value targets like (+)-Isoschizandrin and (+)-Steganone. This technology leverages a palladium-catalyzed asymmetric alkynylation protocol that operates under remarkably mild conditions, utilizing amino acids as chiral inducers to achieve exceptional stereocontrol. For R&D directors and procurement specialists, this represents a significant shift away from traditional, labor-intensive total synthesis routes towards a more efficient, catalytic approach that promises better impurity profiles and reduced manufacturing costs. The ability to maintain high enantiomeric purity throughout the transformation sequence addresses a major pain point in the supply of complex chiral intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the total synthesis of biarylcyclopentadienyl lignans has been plagued by significant challenges that hinder their widespread commercial availability and application in drug discovery. Traditional routes often rely on lengthy multi-step sequences involving harsh reaction conditions, such as extreme temperatures or stoichiometric amounts of toxic reagents, which complicate waste management and increase operational risks. Furthermore, achieving high axial chirality in these congested biaryl systems typically requires expensive, specialized chiral catalysts or resolution steps that inherently limit the maximum theoretical yield to 50%. These inefficiencies result in high production costs and extended lead times, making it difficult for supply chain managers to secure reliable quantities of high-purity intermediates. The lack of stereoselectivity in early stages often propagates through the synthesis, necessitating difficult purification processes that further erode overall process efficiency and economic viability.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a palladium-catalyzed asymmetric alkynylation that streamlines the construction of the chiral axis with remarkable precision. By employing chiral amino acids, such as L-tert-leucine, as transient directing groups, the process eliminates the need for complex, custom-synthesized chiral ligands while maintaining enantiomeric excess values as high as 98%. The reaction proceeds at a moderate temperature of 55°C in acetic acid, demonstrating excellent functional group tolerance and operational simplicity. This approach not only shortens the synthetic route significantly but also ensures that the stereochemical integrity established in the initial alkynylation step is preserved throughout subsequent functionalizations. For manufacturers, this translates to a more predictable and scalable process capable of delivering consistent quality.
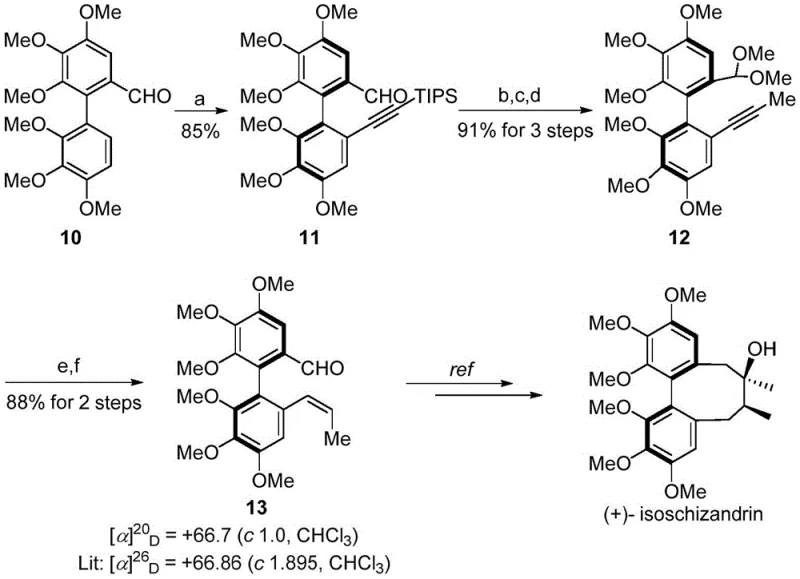
The synthetic route for (+)-isoschizandrin exemplifies this efficiency, starting from a biaryl aldehyde and proceeding through a highly selective alkynylation to form intermediate 11 with 85% yield and 98% ee. Subsequent steps involving protection, desilylation, and methylation are executed with high fidelity, culminating in the formation of the cis-alkene precursor 13 with retained stereochemistry. This logical progression minimizes side reactions and maximizes atom economy, providing a clear advantage over older methods that struggled with low yields and poor stereocontrol. The versatility of this platform is further demonstrated by its applicability to different lignan subclasses, offering a unified strategy for accessing diverse biaryl architectures.
Mechanistic Insights into Pd-Catalyzed Asymmetric Alkynylation
The core innovation of this technology lies in the mechanistic interplay between the palladium catalyst and the chiral amino acid additive, which creates a transient chiral environment around the metal center. During the catalytic cycle, the amino acid likely coordinates to the palladium species, inducing a specific conformation that favors the approach of the alkynyl bromide to one face of the biaryl aldehyde substrate. This dynamic kinetic resolution or asymmetric induction mechanism allows for the differentiation of prochiral faces in the transition state, leading to the preferential formation of one atropisomer over the other. The use of silver salts, such as silver trifluoroacetate, acts as a halide scavenger to facilitate the oxidative addition or transmetallation steps, ensuring the catalytic turnover remains high. Understanding this mechanism is crucial for R&D teams aiming to optimize the process further or adapt it to novel substrates, as it highlights the importance of the amino acid structure and the silver additive in dictating selectivity.
Impurity control is inherently built into this mechanistic design, as the high stereoselectivity reduces the formation of diastereomeric byproducts that are notoriously difficult to separate in biaryl systems. The mild acidic conditions provided by the acetic acid solvent help suppress unwanted side reactions such as polymerization or decomposition of sensitive functional groups often found in lignan precursors. Moreover, the stability of the intermediates, such as the TIPS-protected alkynes, allows for robust handling and purification via standard column chromatography without significant loss of optical purity. This level of control over the impurity profile is essential for meeting the stringent regulatory requirements of pharmaceutical intermediates, where even trace amounts of the wrong enantiomer can be detrimental. The process effectively decouples the complexity of the molecule from the difficulty of its synthesis, enabling the production of high-purity materials suitable for biological testing.
How to Synthesize Biaryl Lignan Intermediates Efficiently
The practical implementation of this synthesis involves a straightforward sequence of reactions that can be readily adapted for pilot or commercial scale production. The initial alkynylation step is the critical determinant of success, requiring precise control over the molar ratios of the palladium catalyst, amino acid, and silver salt to maximize yield and enantioselectivity. Following the formation of the chiral alkyne intermediate, standard organic transformations such as acetal protection, Grignard additions, and reductions are employed to build the remaining carbon framework. Detailed standardized synthetic steps see the guide below.
- React biaryl aldehyde with TIPS-protected alkynyl bromide using Pd(OAc)2, L-tert-leucine, and AgTFA in acetic acid at 55°C.
- Perform functional group transformations including aldehyde protection, desilylation, and methylation to establish the carbon framework.
- Execute final cyclization or reduction steps followed by deprotection to yield target molecules like (+)-Isoschizandrin or (+)-Steganone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers tangible benefits that directly impact the bottom line and operational resilience. The shift from stoichiometric chiral auxiliaries to catalytic systems driven by inexpensive amino acids represents a fundamental cost reduction in pharmaceutical intermediates manufacturing. By simplifying the synthetic pathway and reducing the number of unit operations, the process lowers energy consumption and solvent usage, contributing to a more sustainable and cost-effective production model. Additionally, the robustness of the reaction conditions minimizes the risk of batch failures, ensuring a steady flow of materials that supports just-in-time manufacturing strategies. This reliability is paramount for maintaining continuity in the supply of critical drug substances and avoiding costly delays in clinical or commercial programs.
- Cost Reduction in Manufacturing: The elimination of expensive, proprietary chiral ligands in favor of commodity amino acids like L-tert-leucine drastically lowers the raw material costs associated with catalyst systems. Furthermore, the high yields observed in key steps, such as the 85% yield in the initial alkynylation and 94% in the Knoevenagel condensation for Steganone, mean that less starting material is wasted, improving the overall mass balance of the process. The reduction in synthetic steps also translates to lower labor costs and reduced equipment occupancy time, allowing facilities to produce more material with existing infrastructure. These cumulative efficiencies result in substantial cost savings that can be passed down the supply chain or reinvested into further R&D initiatives.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials, such as biaryl aldehydes and protected alkynyl bromides, mitigates the risk of supply disruptions often associated with exotic reagents. The mild reaction temperature of 55°C and the use of common solvents like acetic acid and ethyl acetate ensure that the process can be executed in a wide range of manufacturing facilities without requiring specialized cryogenic or high-pressure equipment. This flexibility enhances the agility of the supply chain, allowing for rapid scaling of production volumes in response to market demand. Consequently, partners can rely on a stable source of high-quality intermediates, reducing the need for safety stock and freeing up working capital.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reagents and conditions that are compatible with large-scale reactor systems. The high enantioselectivity reduces the burden on downstream purification processes, minimizing the generation of solvent waste associated with repeated recrystallizations or chromatographic separations. Moreover, the avoidance of heavy metal contaminants, beyond the catalytic amounts of palladium which can be effectively scavenged, simplifies the environmental compliance landscape. This alignment with green chemistry principles not only reduces disposal costs but also aligns with the increasing regulatory pressure for sustainable manufacturing practices in the fine chemical industry.
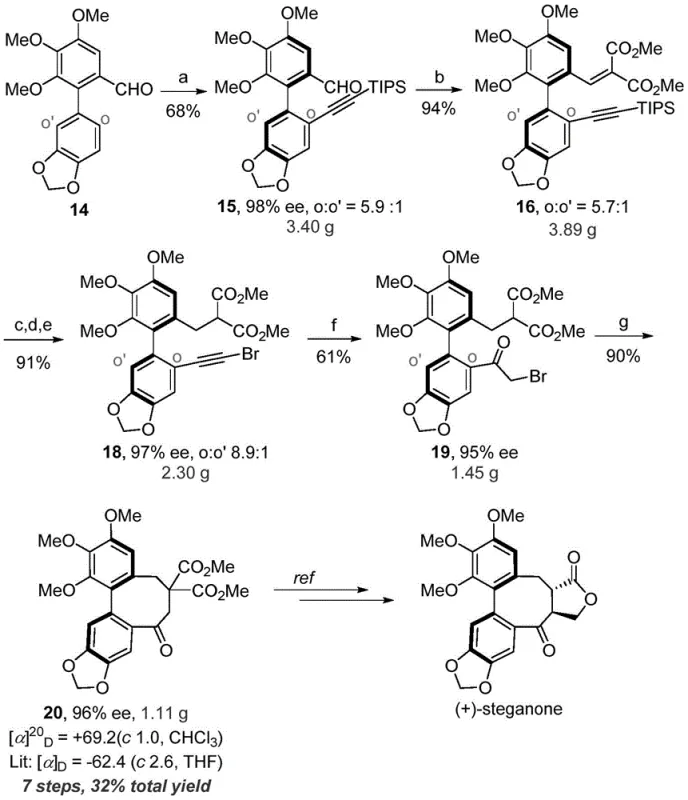
The synthesis of (+)-steganone further validates the broad applicability of this platform, showcasing a 7-step sequence with a total yield of 32% and high enantiomeric purity. The ability to access such complex natural product scaffolds through a convergent and selective route underscores the strategic value of this technology for companies looking to expand their portfolio of bioactive compounds. The consistent performance across different substrates suggests that the underlying chemistry is robust and tolerant of structural variations, making it a versatile tool for medicinal chemists exploring structure-activity relationships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear picture of what partners can expect when adopting this methodology. Understanding these details helps in making informed decisions about process integration and resource allocation.
Q: What is the enantiomeric excess achieved in this synthesis?
A: The process consistently achieves high enantioselectivity, with reported ee values reaching up to 98% for key intermediates like compound 11 and 13.
Q: Does this method require expensive chiral ligands?
A: No, the method utilizes commercially available chiral amino acids, such as L-tert-leucine, as transient directing groups, significantly reducing catalyst costs compared to traditional chiral phosphine ligands.
Q: Can this process be scaled for industrial production?
A: Yes, the reaction conditions are mild (55°C) and operationally simple, avoiding cryogenic temperatures or ultra-high pressures, which facilitates commercial scale-up and supply chain reliability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (+)-Isoschizandrin Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed asymmetric synthesis for the production of high-value biaryl lignans. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to market supply is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the precise measurement of enantiomeric excess required for chiral pharmaceutical intermediates. We are committed to delivering materials that meet the highest standards of quality and consistency, supporting your critical drug development timelines.
We invite you to engage with our technical procurement team to discuss how this innovative route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability as your trusted partner in advancing complex chemical synthesis.
