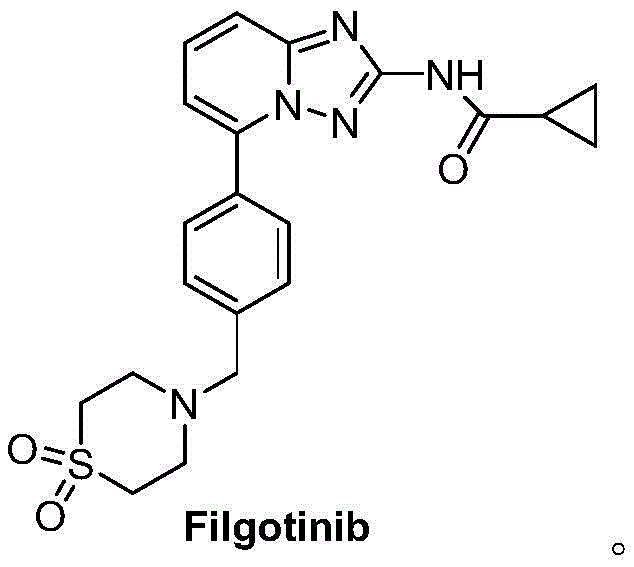
Advanced Synthesis of Filgotinib Intermediate Compound N for Commercial Scale-Up
The pharmaceutical landscape for JAK1 inhibitors continues to evolve rapidly, with Filgotinib standing out as a pivotal therapy for rheumatoid arthritis and ulcerative colitis. As demand for this active pharmaceutical ingredient surges, the efficiency of its supply chain becomes paramount for global manufacturers. Patent CN115368389A introduces a transformative preparation method for Compound N, a critical advanced intermediate in the Filgotinib synthesis pathway. This intellectual property disclosure details a robust, two-step synthetic strategy that bypasses the economic and technical bottlenecks of legacy methods. By shifting the starting point to readily available commodity chemicals and eliminating reliance on precious metal catalysts, this technology offers a compelling value proposition for reliable pharmaceutical intermediate suppliers aiming to optimize their production portfolios. The following analysis dissects the technical merits and commercial implications of this innovation for industry stakeholders.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
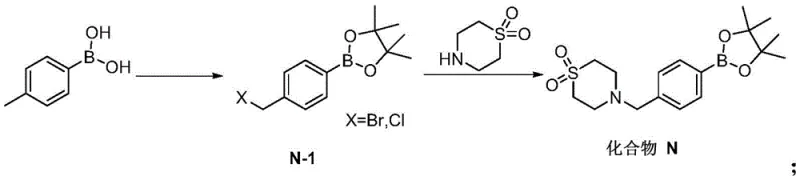
Historically, the synthesis of Compound N has been plagued by significant economic and operational inefficiencies that hinder cost reduction in API manufacturing. Traditional routes, such as those disclosed in earlier patents like WO2010010190, typically commence with 4-bromomethylphenylboronic acid pinacol ester as the foundational building block. This specific starting material is not only prohibitively expensive due to its specialized nature but also presents severe purification challenges. The conventional preparation of this bromo-ester often involves a separate bromination step using N-bromosuccinimide (NBS) on a protected phenylboronic acid, which frequently generates stubborn dibrominated by-products. These impurities are structurally similar to the desired product, making their removal via standard crystallization difficult and often necessitating resource-intensive column chromatography. Furthermore, alternative literature routes, such as those found in WO2008148867, rely on palladium-catalyzed cross-coupling reactions. While chemically effective, the use of palladium introduces substantial raw material costs and creates downstream burdens related to heavy metal residue removal, which is strictly regulated in pharmaceutical production.
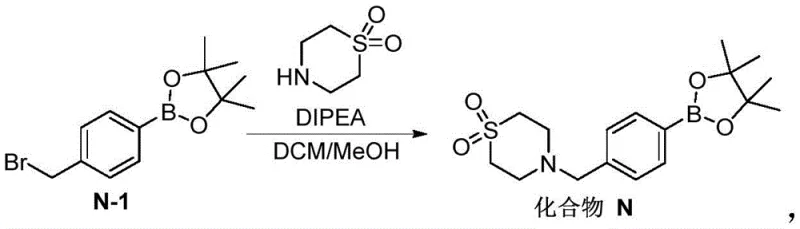
The Novel Approach
In stark contrast to these legacy constraints, the methodology outlined in CN115368389A represents a paradigm shift towards leaner, more sustainable chemistry. The core innovation lies in the direct functionalization of 4-methylphenylboronic acid, a commodity chemical that is vastly more accessible and affordable than its brominated counterpart. The process integrates the protection of the boronic acid with pinacol and the benzylic bromination into a streamlined sequence, effectively generating the key intermediate Compound N-1 in situ. This approach cleverly circumvents the isolation issues associated with the bromo-ester, as the subsequent reaction with thiomorpholine-1,1-dioxide can proceed directly or after simple aqueous workup. By removing the requirement for column chromatography and precious metal catalysts, the new route drastically simplifies the operational workflow. This simplification is critical for achieving commercial scale-up of complex intermediates, as it reduces solvent consumption, waste generation, and processing time, thereby enhancing the overall throughput and economic viability of the manufacturing campaign.
Mechanistic Insights into One-Pot Radical Bromination and Substitution
The chemical elegance of this new process is rooted in a carefully orchestrated radical bromination mechanism followed by a nucleophilic substitution. In the first stage, 4-methylphenylboronic acid reacts with pinacol to form the boronic ester, while simultaneously undergoing benzylic bromination initiated by a radical source such as AIBN (azobisisobutyronitrile) in the presence of a halogenating agent like NBS or liquid bromine. The reaction temperature is maintained between 40°C and 80°C, a range that provides sufficient energy to propagate the radical chain reaction without promoting excessive thermal decomposition or uncontrolled poly-bromination. The stoichiometry of the halogenating agent is tightly controlled, typically kept between 1.0 and 3.0 equivalents relative to the boronic acid, to favor mono-bromination at the benzylic position. This precision is vital for minimizing the formation of the dibrominated impurity, which is the primary quality concern in traditional syntheses. The resulting Compound N-1 is generated in a solvent system compatible with the subsequent step, such as ethyl acetate or heptane, allowing for a telescoped process flow.
Following the formation of the bromo-intermediate, the second stage involves a classic SN2 nucleophilic substitution where the nitrogen atom of thiomorpholine-1,1-dioxide attacks the benzylic carbon, displacing the bromide leaving group. This reaction is facilitated by the presence of a base, such as diisopropylethylamine (DIPEA) or inorganic carbonates, which serves to neutralize the hydrobromic acid by-product and drive the equilibrium forward. The reaction proceeds smoothly at mild temperatures ranging from 0°C to 100°C, demonstrating remarkable robustness across different solvent systems including esters, ethers, and nitriles. From an impurity control perspective, the avoidance of isolating the reactive bromo-intermediate N-1 is a strategic masterstroke. By keeping the intermediate in solution or performing a simple wash, the process prevents the degradation or side-reactions that often occur during the drying and storage of reactive alkyl halides. This "telescoped" nature ensures that the final product, Compound N, is obtained with high purity, often exceeding 98% HPLC purity after a single recrystallization, meeting the stringent specifications required for high-purity pharmaceutical intermediates.
How to Synthesize Filgotinib Intermediate Efficiently
The practical implementation of this synthesis route offers a clear pathway for process chemists to establish a robust manufacturing protocol. The procedure begins with the charging of 4-methylphenylboronic acid, pinacol, and a radical initiator into a reactor containing a non-polar or moderately polar solvent like n-heptane or ethyl acetate. Upon heating to reflux or near-reflux temperatures, the halogenating agent is introduced to trigger the bromination. Once the conversion is complete, monitored by HPLC, the reaction mixture is cooled. Instead of isolating the solid bromo-intermediate, the mixture is either filtered to remove insolubles or washed with aqueous base to remove acidic by-products. The resulting solution containing Compound N-1 is then treated directly with thiomorpholine-1,1-dioxide and a base. After the substitution is complete, the product is isolated via concentration and recrystallization using a solvent pair like ethyl acetate and heptane. For detailed operational parameters, safety guidelines, and specific stoichiometric ratios validated in the patent examples, please refer to the standardized synthesis guide below.
- React 4-methylphenylboronic acid with pinacol and a halogenating agent (e.g., NBS) in a suitable solvent at 40-80°C to generate Compound N-1 without column chromatography.
- Treat the crude Compound N-1 solution with thiomorpholine-1,1-dioxide and a base in solvent B at 0-100°C to effect nucleophilic substitution.
- Purify the final reaction mixture via aqueous workup and recrystallization to obtain high-purity Compound N suitable for API synthesis.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route translates into tangible strategic benefits that extend beyond simple unit cost metrics. The primary advantage lies in the fundamental restructuring of the bill of materials. By replacing the expensive, specialty 4-bromomethylphenylboronic acid pinacol ester with 4-methylphenylboronic acid, manufacturers can access a raw material market characterized by high competition and stable pricing. This shift significantly mitigates the risk of supply disruptions caused by the limited number of vendors capable of producing complex brominated building blocks. Furthermore, the elimination of palladium catalysts removes a major volatility factor from the cost model, as precious metal prices are subject to significant geopolitical and market fluctuations. The simplified purification process, which eschews column chromatography in favor of crystallization and filtration, also implies a drastic reduction in solvent usage and waste disposal costs, contributing to a leaner and more environmentally compliant operation.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substitution of high-value starting materials with commodity chemicals and the removal of expensive catalytic systems. The avoidance of palladium catalysts not only saves on the initial purchase price of the metal but also eliminates the downstream costs associated with scavenging resins and analytical testing for residual metals, which are mandatory for regulatory compliance. Additionally, the ability to telescope the reaction steps without isolating the intermediate reduces labor hours and equipment occupancy time, leading to substantial operational expenditure savings. The overall yield improvement and high purity achieved through recrystallization further enhance the cost-efficiency by maximizing the output per batch and minimizing the need for re-processing or off-spec material handling.
- Enhanced Supply Chain Reliability: From a logistics perspective, relying on 4-methylphenylboronic acid ensures a more resilient supply chain. This chemical is a bulk commodity produced by numerous global manufacturers, ensuring consistent availability and short lead times even during periods of high market demand. In contrast, the specialized brominated precursors used in older methods often have longer lead times and are susceptible to supply bottlenecks due to their complex synthesis. The robustness of the new reaction conditions, which tolerate a variety of solvents and bases, also provides flexibility in sourcing. If a specific solvent becomes unavailable or expensive due to regional supply issues, the process can be adapted to use alternative green solvents without compromising the reaction outcome, thereby securing continuous production capabilities.
- Scalability and Environmental Compliance: The design of this synthesis is inherently scalable, addressing a critical pain point for reducing lead time for high-purity intermediates during technology transfer. The absence of column chromatography is perhaps the most significant factor here; chromatography is notoriously difficult to scale efficiently and is often a bottleneck in kilogram-to-ton production. By replacing it with crystallization, the process aligns perfectly with standard chemical engineering unit operations found in multipurpose plants. Moreover, the reduced solvent intensity and the potential to use greener solvents like ethyl acetate and heptane lower the environmental footprint of the manufacturing process. This alignment with green chemistry principles facilitates easier regulatory approval and supports corporate sustainability goals, making the supply chain more attractive to environmentally conscious partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Filgotinib intermediate synthesis. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for R&D teams evaluating the feasibility of adopting this route and for procurement teams assessing the long-term viability of the supply source. The clarity provided here aims to bridge the gap between academic patent language and practical industrial application.
Q: What are the primary cost drivers eliminated in this new Filgotinib intermediate process?
A: The novel process eliminates the need for expensive palladium catalysts and avoids the procurement of high-cost 4-bromomethylphenylboronic acid pinacol ester, replacing them with commodity chemicals like 4-methylphenylboronic acid.
Q: How does the new method improve impurity profiles compared to traditional routes?
A: By optimizing the stoichiometry of the halogenating agent and avoiding isolation of the intermediate N-1 via column chromatography, the process minimizes the formation of difficult-to-remove dibrominated by-products.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the method utilizes mild reaction conditions, common solvents like ethyl acetate and heptane, and avoids complex purification steps like column chromatography, making it highly amenable to multi-kilogram manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Filgotinib Intermediate Supplier
As the global demand for JAK inhibitors continues to expand, securing a dependable source for critical intermediates like Compound N is a strategic imperative for pharmaceutical companies. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging deep expertise in process chemistry to deliver solutions that balance quality, cost, and speed. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to maintaining stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify that every batch meets the exacting standards required for API synthesis. Our infrastructure is designed to handle complex organic transformations safely and consistently, providing a secure foundation for your supply chain.
We invite you to collaborate with us to explore how this advanced synthesis route can be integrated into your procurement strategy. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to contact our technical procurement team to request specific COA data, route feasibility assessments, and sample availability. By partnering with NINGBO INNO PHARMCHEM, you gain access to not just a chemical supplier, but a strategic ally dedicated to optimizing your Filgotinib supply chain for long-term success.
