Advanced Synthesis of Asymmetric 9,9'-Spirobifluorene Amines for Next-Gen OLEDs
Advanced Synthesis of Asymmetric 9,9'-Spirobifluorene Amines for Next-Gen OLEDs
The rapid evolution of the organic light-emitting diode (OLED) industry demands intermediates that offer superior thermal stability and morphological control. Patent CN102126963A introduces a groundbreaking class of 9,9'-spirobifluorene amino compounds, specifically designed to address the limitations of symmetric derivatives in high-performance display applications. This technology leverages a novel synthetic pathway that replaces costly palladium-catalyzed couplings with robust, cost-effective Friedel-Crafts and Grignard chemistries. The resulting compounds, characterized by the general formula (I), exhibit enhanced amorphous properties and solubility, making them ideal precursors for next-generation optoelectronic materials.
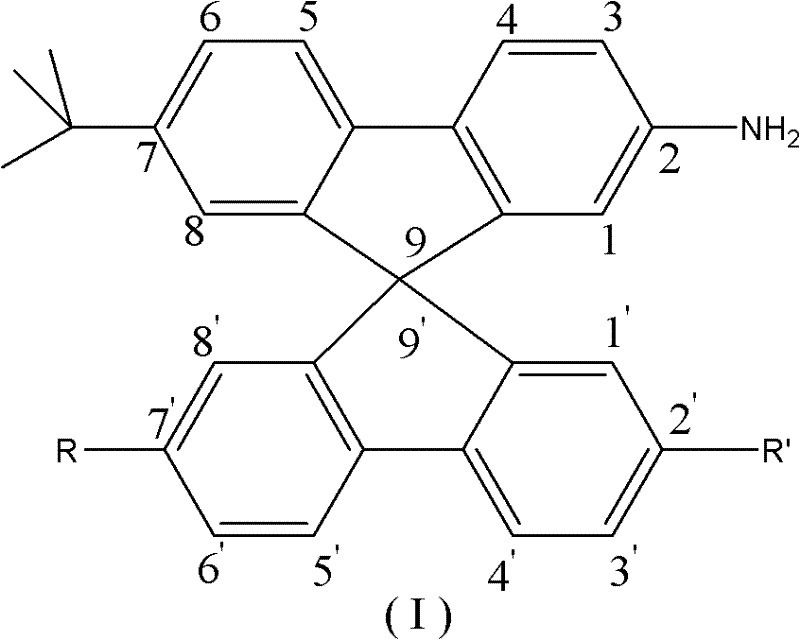
For R&D directors and procurement specialists, this patent represents a significant shift towards sustainable and economical manufacturing of complex electronic chemicals. By utilizing readily available biphenyl derivatives and introducing strategic steric bulk through tert-butyl groups, the process ensures high purity without the burden of removing trace noble metals. This report analyzes the technical merits and commercial viability of this synthesis route, positioning it as a cornerstone for reliable OLED material supplier strategies in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of spirobifluorene derivatives often relies heavily on the use of 2-bromobiphenyl as a key starting material, which is typically derived from o-dibromobenzene via coupling reactions. This conventional approach presents severe economic and operational bottlenecks, primarily due to the exorbitant cost of o-dibromobenzene and the absolute necessity for expensive palladium catalysts to facilitate the bond formation. Furthermore, the reliance on transition metal catalysis introduces significant downstream processing challenges, requiring rigorous purification steps to eliminate trace metal contaminants that can degrade the performance of organic electronic devices. The operational requirements for maintaining inert atmospheres and precise temperature controls for palladium systems also escalate the capital expenditure and complexity of the manufacturing infrastructure.
The Novel Approach
In stark contrast, the methodology disclosed in CN102126963A circumvents these hurdles by initiating the synthesis with inexpensive biphenyl and employing a sequence of alkylation, bromination, and Grignard reactions. This innovative route eliminates the need for precious metal catalysts entirely, substituting them with abundant reagents like aluminum chloride and magnesium. The strategic introduction of tert-butyl groups not only serves as a directing group for subsequent functionalization but also imparts critical asymmetry to the final molecular architecture. This structural modification disrupts molecular packing, thereby preventing crystallization and enhancing the glass transition temperature, which is vital for the longevity and efficiency of OLED layers.
Mechanistic Insights into FeCl3-Catalyzed Cyclization and Functionalization
The core of this synthetic strategy lies in the efficient construction of the spiro-center followed by precise regioselective functionalization. The process begins with a Friedel-Crafts alkylation where biphenyl reacts with tert-butyl chloride in the presence of aluminum chloride to yield 4,4'-di-tert-butyl biphenyl. This intermediate is then subjected to controlled bromination using iodine and bromine, selectively installing a bromine atom at the ortho-position relative to the biphenyl linkage. The resulting bromide is converted into a Grignard reagent, which subsequently attacks a fluorenone derivative. This nucleophilic addition, followed by acid-catalyzed cyclization, constructs the rigid 9,9'-spirobifluorene core with high fidelity.
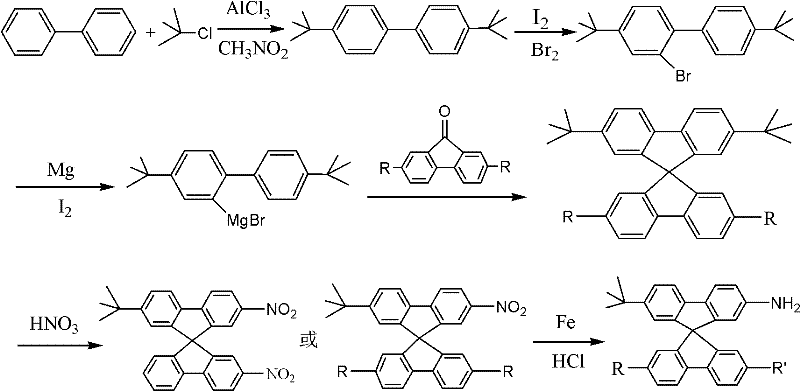
Following the formation of the spiro-skeleton, the patent details a robust nitration and reduction sequence to install the amino functionality. The nitration step utilizes concentrated nitric acid under controlled thermal conditions to introduce nitro groups at the 2 and 2' positions, leveraging the activating effects of the existing substituents to guide regioselectivity. Subsequent reduction using iron powder and hydrochloric acid converts these nitro groups into amines. This metal-acid reduction system is particularly advantageous for industrial scale-up as it avoids the use of high-pressure hydrogenation equipment and expensive heterogeneous catalysts, ensuring a safer and more cost-contained operation while maintaining high chemical purity.
How to Synthesize 9,9'-Spirobifluorene Amines Efficiently
The synthesis of these high-value intermediates requires precise control over reaction stoichiometry and temperature profiles to maximize yield and minimize byproduct formation. The patented procedure outlines a sequential workflow starting from commodity chemicals, emphasizing the importance of anhydrous conditions during the Grignard formation and the careful management of exothermic events during nitration. Operators must adhere to strict protocols for quenching and workup, particularly during the acidification and neutralization steps, to ensure the isolation of the product in its highest possible purity. For a detailed breakdown of the specific molar ratios, solvent choices, and temperature ramps required for each stage, please refer to the standardized synthesis guide below.
- Perform Friedel-Crafts alkylation of biphenyl with tert-butyl chloride using AlCl3 to form 4,4'-di-tert-butyl biphenyl.
- Execute selective bromination using I2/Br2 to generate 4,4'-di-tert-butyl-2-bromo biphenyl, followed by Grignard reagent formation.
- React the Grignard intermediate with fluorenone derivatives to form the spiro-core, followed by nitration and iron-mediated reduction.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this synthetic route offers transformative benefits regarding cost stability and raw material security. By decoupling the production of spirobifluorene intermediates from the volatile market of palladium and specialized halogenated aromatics, manufacturers can achieve a much more predictable cost structure. The reliance on bulk chemicals like biphenyl and tert-butyl chloride, which are produced on a massive scale for various industries, ensures a continuous and resilient supply line that is less susceptible to the geopolitical and logistical disruptions often associated with specialty catalysts. This shift fundamentally alters the cost basis of producing high-purity OLED material, making it accessible for broader commercial applications.
- Cost Reduction in Manufacturing: The elimination of palladium catalysts represents a direct and substantial saving in raw material costs, as noble metals constitute a significant portion of the expense in traditional cross-coupling routes. Additionally, the use of iron powder for reduction instead of catalytic hydrogenation removes the need for high-pressure reactors and expensive supported metal catalysts, further lowering both capital and operational expenditures. The simplified purification process, devoid of heavy metal scavenging steps, reduces solvent consumption and waste treatment costs, contributing to a leaner and more economically efficient manufacturing model.
- Enhanced Supply Chain Reliability: Sourcing strategies are significantly de-risked by the use of commodity-grade starting materials that are available from multiple global suppliers, preventing single-source bottlenecks. The robustness of the chemical transformations, which tolerate standard industrial conditions without requiring ultra-low temperatures or exotic reagents, ensures consistent batch-to-batch quality and reliable delivery schedules. This stability is crucial for downstream electronics manufacturers who require uninterrupted supply chains to meet the demanding production timelines of consumer display markets.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing unit operations such as filtration, distillation, and crystallization that are standard in fine chemical plants, facilitating a smooth transition from pilot to commercial scale. The avoidance of toxic heavy metals aligns with increasingly stringent environmental regulations, reducing the regulatory burden and liability associated with hazardous waste disposal. Furthermore, the high atom economy of the alkylation and cyclization steps minimizes waste generation, supporting sustainability goals and reducing the overall environmental footprint of the production facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these spirobifluorene derivatives. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the feasibility and advantages of this specific manufacturing route. Understanding these details is essential for stakeholders evaluating the integration of this technology into their existing supply chains or R&D pipelines.
Q: What are the primary advantages of this spirobifluorene synthesis route over conventional methods?
A: The primary advantage is the elimination of expensive palladium catalysts and the use of low-cost starting materials like biphenyl instead of o-dibromobenzene. This significantly reduces raw material costs and simplifies the purification process by avoiding heavy metal residues.
Q: How does the structural asymmetry of these compounds benefit OLED performance?
A: The introduction of tert-butyl groups at the 7-position creates strong structural asymmetry. This inhibits molecular crystallization, thereby enhancing the amorphous nature, thermal stability, and solubility of the material, which are critical for uniform film formation in OLED devices.
Q: Is this process scalable for industrial production of electronic chemicals?
A: Yes, the process utilizes standard industrial reagents such as aluminum chloride, magnesium, and iron powder, and avoids sensitive transition metal catalysts. The reaction conditions, such as reflux temperatures and standard atmospheric pressure, are highly conducive to large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 9,9'-Spirobifluorene Amino Compounds Supplier
As the demand for high-performance organic electronic materials continues to surge, partnering with an experienced CDMO is critical for translating laboratory innovations into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from gram-scale optimization to full-scale manufacturing. Our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications, essential for the defect-free performance of OLED displays and lighting applications.
We invite you to collaborate with our technical team to explore how this cost-effective synthesis route can be tailored to your specific product requirements. By engaging with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this palladium-free methodology. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a competitive advantage in the rapidly evolving electronic chemicals market.
