Industrial Scale-Up of Stable Thiophene Intermediates for Next-Generation Pharmaceutical Synthesis
Industrial Scale-Up of Stable Thiophene Intermediates for Next-Generation Pharmaceutical Synthesis
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more stable, cost-effective, and scalable synthetic routes. A pivotal advancement in this domain is detailed in Chinese Patent CN1671686A, which discloses a novel method for producing N-monoalkyl-3-hydroxy-3-(2-thienyl)propylamine derivatives. These compounds serve as critical building blocks for a wide array of bioactive molecules, yet their historical synthesis has been plagued by the use of unstable precursors and harsh reaction conditions. The patented methodology introduces a robust two-step sequence that begins with the formation of a stable (Z)-enamine intermediate, fundamentally altering the economic and technical feasibility of producing these high-value scaffolds. By shifting away from volatile halogenated ketones to stable enamines, this technology offers a pathway to significantly higher purity and yield consistency.
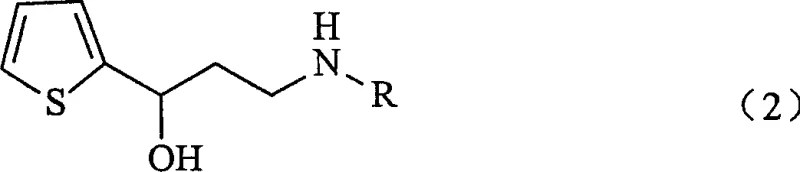
For R&D directors and process chemists, the structural integrity of the intermediate is paramount. The patent highlights the isolation of (Z)-N-monoalkyl-3-oxo-3-(2-thienyl)propenylamine as a key innovation. Unlike previous methods that attempted to generate the carbon-nitrogen bond on an unstable chloroketone backbone, this approach constructs the enamine framework first. This strategic inversion of the synthetic logic ensures that the reactive functionalities are protected within a conjugated system that resists decomposition. The ability to isolate and characterize this intermediate before the final reduction step provides a crucial quality control checkpoint, allowing manufacturers to verify purity before committing to the final reduction, thereby minimizing the risk of processing off-spec materials through expensive downstream purification stages.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-monoalkyl-3-hydroxy-3-(2-thienyl)propylamine relied heavily on the reduction of 1-(2-thienyl)-3-chloropropane-1-one. This conventional route presents severe industrial drawbacks, primarily stemming from the inherent instability of the starting chloroketone. This precursor is prone to rapid degradation and polymerization, making long-term storage impractical and necessitating immediate use or specialized cold-chain logistics that drive up operational costs. Furthermore, the subsequent steps often involve halogen exchange reactions using sodium iodide in acetone, which generates stoichiometric amounts of salt waste and requires careful handling of iodine-containing byproducts. The cumulative effect of these inefficiencies is a process with low atom economy, unpredictable yields due to precursor degradation, and a complex impurity profile that complicates regulatory filing for the final drug substance.
The Novel Approach
The methodology outlined in CN1671686A circumvents these issues by utilizing a condensation reaction between an alkali metal salt of beta-oxo-beta-(2-thienyl)propanal and a monoalkylamine. This reaction cleanly produces the (Z)-N-monoalkyl-3-oxo-3-(2-thienyl)propenylamine intermediate in high yield. As illustrated in the reaction scheme below, this pathway avoids the use of unstable halogenated starting materials entirely.
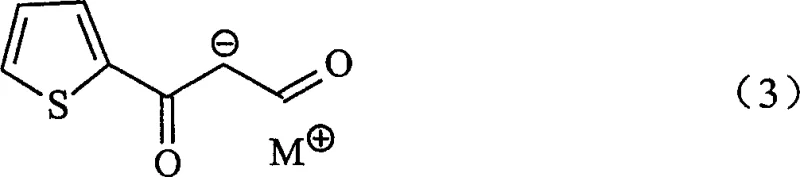
This novel approach not only stabilizes the synthetic route but also simplifies the workup procedure. The intermediate can be isolated via simple phase separation and crystallization from common solvents like methyl tert-butyl ether or toluene. By establishing a stable solid intermediate, the process decouples the synthesis into two distinct, controllable stages. This modularity allows for better inventory management and quality assurance, as the intermediate can be stockpiled without significant degradation. The subsequent reduction step is equally streamlined, utilizing sodium borohydride in the presence of a proton donor, which is a far safer and more economically viable reagent system compared to the complex hydrides or hazardous halogenation agents required by legacy methods.
Mechanistic Insights into Enamine Formation and Selective Reduction
The core of this technological breakthrough lies in the precise control of stereochemistry and reactivity during the enamine formation. The reaction between the beta-keto aldehyde salt and the monoalkylamine proceeds through a condensation mechanism that favors the formation of the (Z)-isomer. This stereoselectivity is crucial because the spatial arrangement of the enamine double bond influences the subsequent reduction efficiency and the purity of the final amine. The conjugated system formed between the thiophene ring, the carbonyl group, and the nitrogen atom creates a stabilized electronic environment that prevents unwanted side reactions such as dimerization, which was a common failure mode in earlier attempts to synthesize similar structures using dimethylamine hydrochloride. The patent data indicates that maintaining the reaction temperature between 0°C and 100°C, preferably 10°C to 80°C, optimizes this equilibrium, ensuring that the thermodynamic product is the desired Z-enamine rather than unstable byproducts.
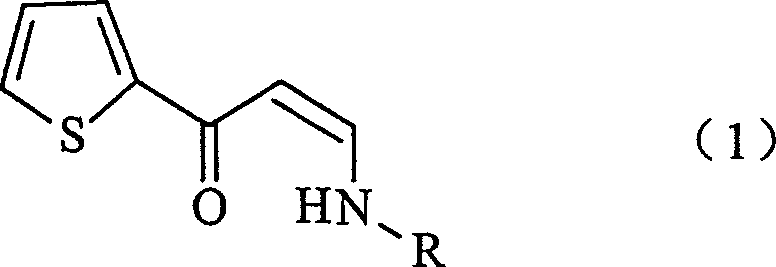
Following the isolation of the intermediate, the reduction mechanism employs sodium borohydride (NaBH4) activated by a proton donor such as acetic acid. This combination generates a reactive borohydride species capable of selectively reducing the carbon-carbon double bond and the ketone functionality without affecting the thiophene ring or the amine group. The presence of the proton donor is mechanistically significant; it facilitates the delivery of hydride ions to the electrophilic centers while preventing the accumulation of basic byproducts that could trigger retro-aldol type decompositions. The patent specifies that the amount of proton donor should be carefully controlled, typically between 0.1 to 10 moles per mole of substrate, to balance reaction rate against the risk of over-acidification which could lead to salt formation or hydrolysis. This fine-tuned reactivity ensures that the final N-monoalkyl-3-hydroxy-3-(2-thienyl)propylamine is obtained with high chemical purity, minimizing the need for extensive chromatographic purification.
How to Synthesize (Z)-N-Monoalkyl-3-Oxo-3-(2-Thienyl)Propenylamine Efficiently
The practical execution of this synthesis requires strict adherence to the optimized parameters regarding solvent choice and stoichiometry to maximize yield and safety. The process begins with the preparation of the beta-keto aldehyde salt, followed by the controlled addition of the monoalkylamine in methanol. Detailed operational guidelines regarding temperature ramps, addition rates, and crystallization protocols are essential for reproducibility at scale. For process engineers looking to implement this route, understanding the solubility profiles of the intermediate in various solvent systems is key to designing an efficient isolation strategy. The following guide outlines the standardized procedural steps derived from the patent examples to ensure successful replication of this high-yielding transformation.
- React the alkali metal salt of beta-oxo-beta-(2-thienyl)propanal with a monoalkylamine compound in an alcoholic solvent to form the Z-enamine intermediate.
- Isolate the stable (Z)-N-monoalkyl-3-oxo-3-(2-thienyl)propenylamine intermediate via phase separation and crystallization.
- Reduce the isolated enamine intermediate using sodium borohydride in the presence of a proton donor to yield the final hydroxy-propylamine product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the transition to this novel synthetic route offers profound advantages in terms of cost stability and risk mitigation. The elimination of unstable starting materials removes a major bottleneck in raw material sourcing, as suppliers no longer need to maintain specialized storage conditions for volatile chloroketones. This shift translates directly into lower raw material costs and reduced waste disposal fees associated with degraded batches. Furthermore, the use of commodity chemicals such as sodium borohydride, acetic acid, and toluene ensures that the supply chain is resilient against market fluctuations for exotic reagents. The robustness of the process means that production schedules are less likely to be disrupted by failed batches, providing a reliable flow of intermediates to downstream API manufacturing sites.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the high stability of the intermediate and the use of inexpensive reducing agents. By avoiding the need for cryogenic conditions or expensive transition metal catalysts, the overall utility consumption and capital equipment requirements are significantly lowered. The ability to recycle solvents like toluene and methanol further enhances the cost efficiency, while the high yield reduces the cost per kilogram of the final active intermediate. Additionally, the simplified workup procedure reduces labor hours and processing time, contributing to substantial overall cost savings in the manufacturing budget.
- Enhanced Supply Chain Reliability: Supply chain continuity is greatly improved by the thermal stability of the (Z)-enamine intermediate. Unlike previous precursors that required just-in-time delivery to prevent decomposition, this intermediate can be manufactured in large campaigns and stored for extended periods without quality loss. This capability allows manufacturers to build strategic inventory buffers, protecting against upstream supply disruptions. The reliance on widely available bulk chemicals also diversifies the supplier base, reducing dependency on single-source vendors for critical reagents and ensuring consistent availability for long-term production contracts.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on standard unit operations such as crystallization and liquid-liquid extraction. The absence of heavy metals or hazardous halogenating agents simplifies environmental compliance and waste treatment, lowering the regulatory burden on manufacturing facilities. The atom economy of the condensation step is superior to substitution-based routes, resulting in less chemical waste generation. This alignment with green chemistry principles not only reduces disposal costs but also supports corporate sustainability goals, making the supply chain more attractive to environmentally conscious pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy synthesis routes. Understanding these nuances is critical for technical teams evaluating the feasibility of adopting this process for commercial production of thiophene-based pharmaceutical intermediates.
Q: Why is the new enamine intermediate superior to traditional chloroketone precursors?
A: Traditional precursors like 1-(2-thienyl)-3-chloropropane-1-one are thermally unstable and prone to degradation during storage, leading to inconsistent yields. The novel (Z)-enamine intermediate described in CN1671686A exhibits superior thermal stability and shelf-life, ensuring consistent quality in large-scale manufacturing.
Q: What are the safety advantages of using sodium borohydride in this process?
A: Sodium borohydride is a safer, more manageable reducing agent compared to complex metal hydrides or hazardous halogen exchange reagents used in older methods. It operates effectively under mild conditions (20-100°C) in common solvents like toluene, significantly reducing operational risks and waste treatment costs.
Q: Can this process be scaled for commercial API production?
A: Yes, the process utilizes industrially common solvents (methanol, toluene) and reagents (NaBH4, monoalkylamines) without requiring cryogenic conditions or exotic catalysts. The high stability of the intermediate allows for flexible batch scheduling, making it highly suitable for multi-ton commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Monoalkyl-3-Hydroxy-3-(2-Thienyl)Propylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of life-saving medications. Our technical team has thoroughly analyzed the methodology presented in CN1671686A and possesses the expertise to translate this laboratory-scale innovation into commercial reality. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of N-monoalkyl-3-hydroxy-3-(2-thienyl)propylamine meets the highest international standards for pharmaceutical intermediates.
We invite global partners to collaborate with us to leverage this advanced technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both the quality and cost-efficiency of your pharmaceutical manufacturing operations.
