Advanced Biocatalytic Production of (R)-Phenylethylene Glycol via Engineered Carbonyl Reductase
Advanced Biocatalytic Production of (R)-Phenylethylene Glycol via Engineered Carbonyl Reductase
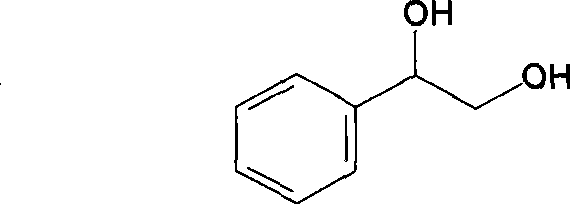
The landscape of chiral intermediate manufacturing is undergoing a profound transformation, driven by the urgent need for sustainable, high-precision synthetic routes that can meet the rigorous demands of the pharmaceutical and advanced materials sectors. Patent CN101407780B introduces a groundbreaking methodology for the preparation of (R)-phenylethylene glycol, a critical chiral building block, through the strategic application of site-directed mutagenesis on carbonyl reductases. This innovation addresses long-standing bottlenecks in stereoselective synthesis by fundamentally altering the enzyme's active site to invert both coenzyme specificity and product stereochemistry. By transitioning from natural (S)-selectivity to engineered (R)-selectivity, this technology offers a robust platform for generating optically pure diols without the heavy reliance on precious metal catalysts or inefficient resolution processes. The implications for supply chain stability and cost efficiency in the production of high-value fine chemicals are substantial, marking a significant leap forward in industrial biotechnology.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of optically pure (R)-phenylethylene glycol has been fraught with significant technical and economic challenges that hinder large-scale adoption. Conventional chemical approaches often rely on the asymmetric reduction of alpha-hydroxy ketones using transition metal catalysts, which necessitate stringent exclusion of oxygen and moisture, alongside the use of expensive chiral ligands that are difficult to recover and recycle. Alternatively, biological methods utilizing wild-type microorganisms, such as Baker's yeast, frequently suffer from poor stereoselectivity, yielding mixtures of enantiomers that require energy-intensive downstream purification to achieve the necessary optical purity for pharmaceutical applications. Furthermore, many native enzymatic systems exhibit a strict dependence on NADPH as a cofactor, which is considerably more expensive and less stable than NADH, thereby inflating the operational costs of the biotransformation process and limiting its commercial viability for bulk chemical production.
The Novel Approach
The methodology disclosed in the patent data represents a paradigm shift by leveraging protein engineering to reconstruct the catalytic machinery at the molecular level. Instead of relying on the inherent limitations of wild-type enzymes, this approach utilizes a recombinant E. coli strain, specifically BL21/pETSCR6768, which harbors a mutated carbonyl reductase gene. Through the precise substitution of amino acids at positions 67 and 68, the enzyme's binding pocket is reshaped to favor the formation of the (R)-enantiomer with exceptional fidelity. Crucially, this genetic modification simultaneously switches the cofactor requirement from NADPH to NADH, unlocking access to cheaper and more abundant reducing equivalents. This dual improvement in stereoselectivity and cofactor economy creates a highly efficient biocatalytic system that operates under mild aqueous conditions, drastically simplifying the reaction setup and reducing the environmental footprint associated with traditional organic synthesis.
Mechanistic Insights into Site-Directed Mutagenesis of Carbonyl Reductase
The core of this technological breakthrough lies in the rational design of the enzyme's active site, where specific amino acid residues dictate the spatial orientation of the substrate during the hydride transfer event. In the native (S)-specific carbonyl reductase, the presence of Serine at position 67 and Histidine at position 68 creates a steric and electronic environment that favors the attack of the hydride ion on one face of the ketone substrate. By mutating these residues to Aspartic Acid (S67D/H68D), the local electrostatic potential and hydrogen bonding network are fundamentally altered. This structural perturbation forces the substrate, 2-hydroxyacetophenone, to bind in an inverted orientation relative to the nicotinamide ring of the cofactor. Consequently, the pro-R face of the carbonyl group is presented to the catalytic center, ensuring that the reduction proceeds exclusively to form the (R)-configured alcohol. This level of mechanistic control demonstrates the power of modern protein engineering to tailor biocatalysts for specific industrial outcomes.
Beyond stereocontrol, the mutation strategy effectively resolves the issue of impurity generation that plagues less specific catalytic systems. In conventional biocatalysis, side reactions such as over-reduction or the formation of regioisomers can compromise the quality of the final API intermediate, necessitating complex chromatographic separations. The engineered enzyme described in the patent exhibits a lock-and-key fit for the target substrate, minimizing off-target interactions and ensuring a clean reaction profile. The high specificity means that the crude reaction mixture contains predominantly the desired (R)-phenylethylene glycol, with minimal byproduct formation. This intrinsic purity reduces the burden on downstream processing units, allowing for simpler extraction and crystallization protocols. For R&D directors, this translates to a more predictable and robust process window, where variations in feedstock quality have a diminished impact on the final product specifications, thereby enhancing overall process reliability.
How to Synthesize (R)-Phenylethylene Glycol Efficiently
The practical implementation of this biocatalytic route involves a streamlined fermentation and biotransformation workflow that is compatible with existing industrial infrastructure. The process begins with the cultivation of the recombinant E. coli host, where expression of the mutant enzyme is induced under controlled temperature conditions to maximize soluble protein yield. Following cell harvest, the whole cells can be employed directly as biocatalysts, eliminating the need for costly enzyme purification steps while retaining high catalytic activity. The reaction is conducted in a buffered aqueous system at ambient pressure, where the substrate is fed gradually to maintain optimal conversion rates.
- Cultivate the recombinant strain E. coli BL21/pETSCR6768 in LB medium with ampicillin induction at 30°C to express the mutated carbonyl reductase.
- Prepare the biotransformation system using acetate buffer (pH 6.0) and add 2-hydroxyacetophenone substrate along with the recombinant whole cells.
- Incubate the reaction mixture at 30°C for 48 hours, followed by extraction and purification to isolate (R)-phenylethylene glycol with >98% e.e.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this engineered biocatalytic route offers compelling economic and logistical benefits that extend beyond simple yield improvements. The shift from NADPH-dependent to NADH-dependent catalysis is particularly significant, as NADH is not only less expensive but also benefits from a wider array of established industrial regeneration systems. This change fundamentally alters the cost structure of the reducing agent, which is often a major variable cost in enzymatic reductions. Furthermore, the use of recombinant E. coli allows for high-density fermentation, meaning that a vast amount of catalytic power can be generated in a relatively small footprint. This scalability ensures that supply can be ramped up quickly to meet market demand without the lead times associated with sourcing rare earth metals or complex chiral ligands. The robustness of the biological system also implies greater resilience against raw material price volatility, providing a more stable cost baseline for long-term contracting.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts and chiral ligands results in a direct reduction in raw material expenditures, while the switch to NADH lowers cofactor costs significantly. The mild reaction conditions reduce energy consumption for heating and cooling, and the high selectivity minimizes waste disposal costs associated with solvent-heavy purification steps. These factors combine to create a leaner manufacturing process with a lower overall cost of goods sold, enhancing margin potential for downstream pharmaceutical products.
- Enhanced Supply Chain Reliability: Reliance on fermentation-based production decouples the supply chain from the geopolitical and mining constraints often associated with transition metal catalysts. The recombinant strain can be stored and propagated indefinitely, ensuring a consistent and renewable source of catalytic activity. This biological continuity reduces the risk of supply disruptions caused by vendor shortages or logistics bottlenecks, providing a secure foundation for just-in-time manufacturing strategies and long-term supply agreements.
- Scalability and Environmental Compliance: The aqueous nature of the biotransformation aligns perfectly with green chemistry principles, significantly reducing the volume of hazardous organic solvents required compared to traditional chemical synthesis. This simplifies regulatory compliance regarding volatile organic compound (VOC) emissions and wastewater treatment. The process is inherently scalable from liter-scale flasks to cubic-meter fermenters without losing efficiency, facilitating a smooth technology transfer from pilot plant to commercial production facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented biocatalytic technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific application needs. Understanding these details is crucial for making informed decisions about process integration and supplier qualification.
Q: How does the site-directed mutagenesis improve coenzyme specificity?
A: The mutation of Serine-67 and Histidine-68 to Aspartic Acid in the carbonyl reductase switches the enzyme's preference from the expensive NADPH to the more industrially viable NADH, significantly lowering cofactor costs.
Q: What is the optical purity achievable with this method?
A: Under optimized conditions using the recombinant strain CCTCC NO: M208079, the process yields (R)-phenylethylene glycol with an optical purity of up to 100% e.e., eliminating the need for complex chiral resolution steps.
Q: Is this process scalable for industrial manufacturing?
A: Yes, the use of recombinant E. coli allows for high-density fermentation and whole-cell biocatalysis, which are proven technologies for scaling from laboratory grams to multi-ton commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-Phenylethylene Glycol Supplier
The technical potential of the site-directed mutagenesis route for (R)-phenylethylene glycol is immense, offering a clear pathway to high-purity chiral intermediates that are essential for next-generation pharmaceuticals and liquid crystal materials. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are successfully translated into robust industrial realities. Our facility is equipped with rigorous QC labs and advanced analytical capabilities to meet stringent purity specifications, guaranteeing that every batch of (R)-phenylethylene glycol delivered meets the exacting standards required by global regulatory bodies. We understand the critical nature of chiral integrity in drug development and are committed to providing a supply partner that prioritizes quality and consistency above all else.
We invite you to engage with our technical procurement team to discuss how this advanced biocatalytic technology can be integrated into your supply chain to drive efficiency and reduce costs. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits specific to your volume requirements. We encourage you to contact us to obtain specific COA data and route feasibility assessments, allowing you to validate the performance of our materials against your internal benchmarks. Let us collaborate to optimize your synthesis of complex pharmaceutical intermediates and secure a competitive advantage in the marketplace.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
