Industrial Scale Production of Pyrethroid Intermediates via Palladium Catalysis
The global demand for high-efficiency pyrethroid insecticides continues to drive innovation in the synthesis of their core structural motifs, specifically the (1-alkenyl)cyclopropane scaffold. Patent CN1934063A introduces a transformative industrial method for producing these critical agrochemical intermediates, addressing long-standing inefficiencies in traditional manufacturing routes. By leveraging a palladium-catalyzed decarboxylation strategy, this technology enables the conversion of readily available formylcyclopropane precursors into valuable (1-alkenyl)cyclopropane derivatives with exceptional purity and stereocontrol. For R&D directors and procurement specialists seeking a reliable agrochemical intermediate supplier, this process represents a significant leap forward in sustainable chemical manufacturing, offering a robust alternative to legacy methods that are increasingly scrutinized for their environmental footprint.
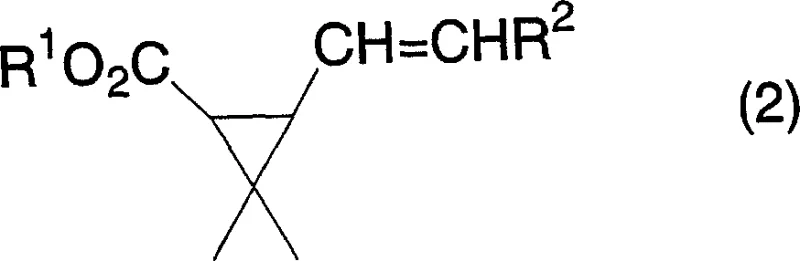
The versatility of this synthetic approach is underscored by its compatibility with a wide range of substituents, allowing for the customization of the final product to meet specific biological activity profiles required by different pest control formulations. The patent details the successful application of this method across various alkyl, alkenyl, and aryl substitutions, demonstrating its broad utility in the fine chemical sector. As the industry moves towards greener chemistry standards, adopting such catalytic processes becomes not just a technical advantage but a strategic necessity for maintaining competitiveness in the global supply chain of crop protection agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of (1-alkenyl)cyclopropane compounds has relied heavily on the Wittig reaction, a classic organic transformation that, while effective, suffers from severe drawbacks when applied to large-scale industrial production. The primary limitation lies in the stoichiometric generation of triphenylphosphine oxide as a by-product, which creates a massive waste stream that is difficult and costly to dispose of in an environmentally compliant manner. Furthermore, the Wittig reagents themselves are often expensive and sensitive to moisture and air, requiring stringent handling conditions that increase operational complexity and safety risks in a manufacturing plant. These factors collectively contribute to a higher cost of goods and a larger carbon footprint, making the conventional route less attractive for modern, sustainability-focused chemical enterprises aiming for cost reduction in pyrethroid manufacturing.
The Novel Approach
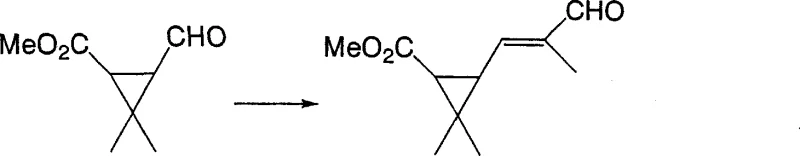
In stark contrast, the method disclosed in CN1934063A employs a two-step sequence that begins with an aldol-type condensation to form a (2-formyl-1-enyl)cyclopropane intermediate, followed by a palladium-catalyzed decarboxylation to yield the final product. This novel pathway elegantly bypasses the need for phosphorus-based reagents, thereby eliminating the associated waste disposal issues entirely. The initial condensation step, as illustrated in the reaction scheme below, utilizes common aldehydes and formylcyclopropanes under mild basic conditions, ensuring high atom economy and ease of operation.
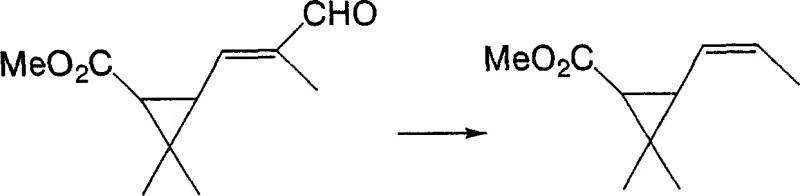
Following the formation of the intermediate, the subsequent decarboxylation step utilizes a palladium catalyst to efficiently remove the carboxyl group, driving the reaction to completion with high conversion rates. This second transformation, depicted in the following scheme, proceeds smoothly in high-boiling solvents like xylene, facilitating easy product isolation and catalyst recovery. By shifting from a stoichiometric waste-generating process to a catalytic cycle, this approach significantly simplifies the downstream processing workflow, reduces solvent consumption, and enhances the overall safety profile of the manufacturing facility, making it an ideal candidate for commercial scale-up of complex agrochemical intermediates.
Mechanistic Insights into Palladium-Catalyzed Decarboxylation
The core of this technological advancement lies in the mechanistic efficiency of the palladium-catalyzed decarboxylation step, which converts the (2-formyl-1-enyl)cyclopropane precursor into the desired (1-alkenyl)cyclopropane. The reaction mechanism involves the oxidative addition of the palladium catalyst to the substrate, followed by the extrusion of carbon dioxide and subsequent reductive elimination to form the new carbon-carbon double bond. Crucially, the choice of ligand and support material for the palladium catalyst plays a pivotal role in determining the reaction kinetics and the stereochemical outcome. For instance, the use of heterogeneous catalysts like palladium on carbon or palladium on alumina allows for facile separation of the metal from the reaction mixture, preventing metal contamination in the final API intermediate and reducing the need for expensive scavenging resins.
Furthermore, the process exhibits remarkable control over the geometric isomerism of the resulting double bond, a critical quality attribute for bioactive molecules. Experimental data from the patent indicates that by fine-tuning reaction parameters such as temperature and catalyst loading, manufacturers can achieve Z/E ratios favoring the biologically active isomer, often exceeding 95/5 selectivity. This level of precision minimizes the formation of inactive or potentially harmful isomers, thereby simplifying the purification burden and ensuring a consistent, high-quality impurity profile. For quality assurance teams, this inherent selectivity translates to more robust batch-to-batch consistency and reduced risk of regulatory rejection due to unexpected impurities.
How to Synthesize (1-Alkenyl)Cyclopropane Efficiently
To implement this synthesis effectively, operators must adhere to precise reaction conditions regarding temperature control and reagent addition rates to maximize yield and selectivity. The process begins with the careful preparation of the condensation mixture, ensuring that the molar ratios of aldehyde to formylcyclopropane are optimized to prevent self-condensation side reactions. Detailed standard operating procedures for the subsequent decarboxylation step involve maintaining specific thermal profiles to activate the palladium catalyst without degrading the sensitive cyclopropane ring. The detailed standardized synthesis steps are outlined in the guide below.
- Perform an aldol-type condensation between a formylcyclopropane compound and an aldehyde in the presence of a base (e.g., pyrrolidine) and acid to generate the (2-formyl-1-enyl)cyclopropane intermediate.
- Isolate the intermediate organic layer through aqueous washing and concentration, optionally purifying via distillation or column chromatography to remove by-products.
- Subject the intermediate to decarboxylation using a palladium catalyst (e.g., Pd/C or Pd(PPh3)4) in a high-boiling solvent at 100-180°C to yield the final (1-alkenyl)cyclopropane product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers tangible strategic benefits that extend beyond mere technical feasibility. The elimination of phosphorus waste streams directly correlates to a substantial reduction in waste treatment costs and regulatory compliance overhead, freeing up capital for other operational investments. Additionally, the reliance on commodity chemicals like simple aldehydes and esters ensures a stable and resilient raw material supply base, mitigating the risks associated with sourcing specialized or volatile reagents that often plague the fine chemical industry.
- Cost Reduction in Manufacturing: The transition to a catalytic process fundamentally alters the cost structure by removing the need for expensive stoichiometric reagents and the associated waste disposal fees. The ability to use supported palladium catalysts further enhances economic viability, as these materials can often be regenerated or recycled, drastically lowering the per-kilogram cost of the catalyst component. Moreover, the simplified workup procedures reduce solvent usage and energy consumption during distillation, contributing to a leaner and more cost-effective production model that improves overall margin potential.
- Enhanced Supply Chain Reliability: By utilizing widely available starting materials and robust reaction conditions, this method significantly reduces lead time for high-purity agrochemical intermediates. The process is less susceptible to supply disruptions caused by the scarcity of niche reagents, ensuring a continuous flow of materials to downstream formulation plants. This reliability is crucial for meeting the seasonal demands of the agricultural sector, where timely delivery of crop protection agents can determine the success of a harvest season.
- Scalability and Environmental Compliance: The reaction conditions described, such as operating temperatures between 100°C and 180°C in common solvents like xylene or toluene, are fully compatible with existing standard stainless-steel reactor infrastructure. This compatibility facilitates rapid commercial scale-up from pilot plant to multi-ton production without requiring specialized equipment modifications. Furthermore, the reduced environmental load aligns with increasingly strict global environmental regulations, future-proofing the manufacturing site against tighter emission standards and enhancing the company's corporate social responsibility profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed synthesis route. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners and stakeholders.
Q: How does this palladium-catalyzed method improve upon traditional Wittig reactions?
A: Traditional Wittig reactions generate stoichiometric amounts of phosphorus oxide waste, creating significant environmental burdens and disposal costs. This patented method utilizes a catalytic decarboxylation pathway that eliminates phosphorus waste entirely, resulting in a cleaner process with reduced downstream purification requirements and lower environmental compliance costs.
Q: What level of stereoselectivity (Z/E ratio) can be achieved with this process?
A: The process demonstrates excellent control over double bond geometry. Experimental data indicates Z/E ratios reaching as high as 99/1 depending on the specific catalyst and reaction conditions employed, such as using palladium on alumina at controlled temperatures. This high stereoselectivity is critical for ensuring the biological efficacy of the final pyrethroid insecticide.
Q: Is the palladium catalyst recoverable for large-scale manufacturing?
A: Yes, the protocol specifically supports the use of supported palladium catalysts, such as palladium on carbon or palladium on silica-alumina. These heterogeneous catalysts can be easily removed via simple filtration after the reaction is complete, allowing for potential catalyst recovery and significantly reducing the cost of goods sold compared to homogeneous systems that require complex extraction.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (1-Alkenyl)Cyclopropane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable synthesis routes in the modern agrochemical landscape. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising laboratory results of patent CN1934063A can be seamlessly translated into industrial reality. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of (1-alkenyl)cyclopropane intermediate performs consistently in your final formulations.
We invite you to collaborate with us to leverage this advanced technology for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can drive value and efficiency for your organization.
