Advanced Manufacturing of (4E)-5-Chloro-2-isopropyl-4-pentenoic Acid Esters for Global Pharma
Introduction to Patent CN101035753A
The chemical landscape for producing vital agrochemical and pharmaceutical intermediates is constantly evolving, driven by the need for greener processes and higher economic efficiency. Patent CN101035753A introduces a groundbreaking methodology for the synthesis of (4E)-5-chloro-2-isopropyl-4-pentenoic acid esters, a critical building block for various bioactive compounds including insecticides and antihypertensive agents. This technology addresses long-standing inefficiencies in multi-step organic synthesis by integrating alkylation and decarboxylation into a seamless, continuous workflow. By eliminating the need for aqueous workups between critical reaction stages, the process not only enhances the overall yield but also drastically reduces the environmental footprint associated with solvent waste. For industry leaders seeking a reliable agrochemical intermediate supplier, understanding the nuances of this patent provides a strategic advantage in securing high-quality raw materials while optimizing supply chain resilience.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex pentenoic acid derivatives involved discrete reaction steps separated by rigorous purification protocols that often compromised efficiency. Traditional approaches typically required quenching reaction mixtures with water to isolate intermediates, a practice that poses significant challenges when using high-boiling polar aprotic solvents like sulfolane. In these conventional workflows, the polar solvent tends to partition into the aqueous waste stream, leading to substantial material loss and creating a heavy burden on wastewater treatment facilities. Furthermore, the repeated isolation and drying of intermediates between alkylation and decarboxylation steps introduce opportunities for product degradation and yield erosion. The accumulation of these inefficiencies results in a manufacturing process that is not only cost-prohibitive due to raw material wastage but also environmentally unsustainable due to the generation of large volumes of saline wastewater containing dissolved organic solvents.
The Novel Approach
The innovative strategy outlined in the patent data revolutionizes this synthesis by employing a telescoped, one-pot reaction sequence that bypasses aqueous quenching entirely. Instead of isolating the dialkylated malonate intermediate through water extraction, the process utilizes distillation to remove volatile byproducts and excess reagents directly from the reaction vessel. This allows the subsequent decarboxylation to proceed in the same reactor, utilizing the alkali metal chloride salts generated in situ during the initial alkylation phases as catalysts. This integration means that the solvent system, often a optimized mixture of toluene and sulfolane, can be recovered and reused with minimal degradation. 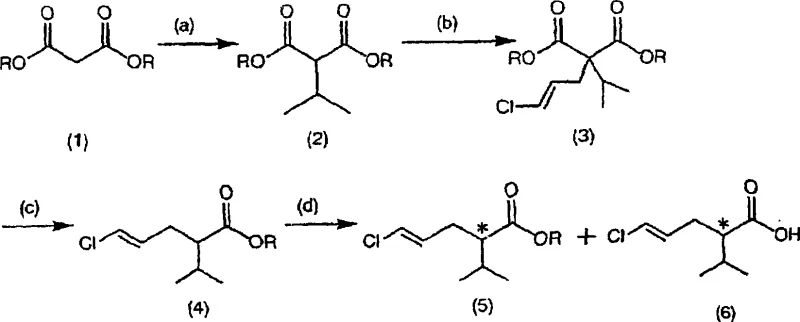 This continuity ensures that cost reduction in pharmaceutical manufacturing is achieved not just through higher chemical yields, but through the drastic simplification of unit operations and the elimination of solvent recovery bottlenecks that plague older technologies.
This continuity ensures that cost reduction in pharmaceutical manufacturing is achieved not just through higher chemical yields, but through the drastic simplification of unit operations and the elimination of solvent recovery bottlenecks that plague older technologies.
Mechanistic Insights into Tandem Alkylation and Decarboxylation
The core chemical innovation lies in the precise management of reaction conditions to facilitate a tandem alkylation followed by a salt-promoted decarboxylation. The process begins with the deprotonation of dimethyl malonate using a strong base such as sodium methoxide in an aprotic solvent, generating a nucleophilic enolate. This species undergoes SN2 substitution with isopropyl halide to form the mono-alkylated intermediate. Crucially, the protocol mandates the removal of the co-generated alcohol (e.g., methanol) via distillation before the second alkylation with (1E)-1,3-dichloro-1-propene. This step is vital because residual protic solvents can inhibit the formation of the second enolate or lead to solvolysis of the allylic chloride electrophile. The resulting dialkylated malonate derivative retains the alkali metal chloride byproduct within the reaction matrix, which serves a dual purpose: it acts as a phase transfer catalyst equivalent and lowers the activation energy for the subsequent thermal decarboxylation.
During the final transformation, the addition of a controlled amount of water to the heated reaction mixture triggers the hydrolysis and decarboxylation of one ester group. The presence of the in-situ generated salt accelerates this step, allowing it to proceed at lower temperatures and shorter reaction times compared to uncatalyzed variants. This mechanistic elegance ensures that the geometric integrity of the (4E)-double bond is preserved, preventing isomerization which is a common impurity profile issue in allylic systems. 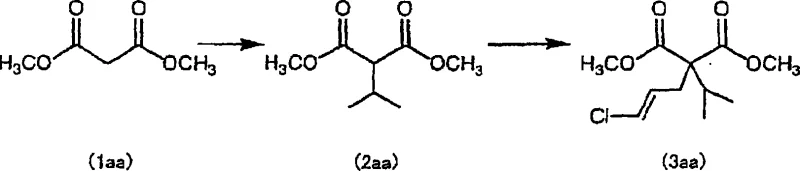 Furthermore, the ability to perform optical resolution on the resulting racemic ester using lipases or esterases adds another layer of sophistication, enabling the production of single-enantiomer intermediates essential for modern chiral drug synthesis. The enzyme selectively hydrolyzes one enantiomer, allowing for the separation of the desired optically active acid from the unreacted ester with high enantiomeric excess.
Furthermore, the ability to perform optical resolution on the resulting racemic ester using lipases or esterases adds another layer of sophistication, enabling the production of single-enantiomer intermediates essential for modern chiral drug synthesis. The enzyme selectively hydrolyzes one enantiomer, allowing for the separation of the desired optically active acid from the unreacted ester with high enantiomeric excess.
How to Synthesize (4E)-5-Chloro-2-isopropyl-4-pentenoic Acid Ester Efficiently
Implementing this synthesis requires strict adherence to the sequential addition of reagents and the precise control of distillation parameters to manage solvent composition. The process is designed to be robust, leveraging commercially available starting materials like dimethyl malonate and isopropyl bromide to ensure supply chain stability. Operators must monitor the removal of volatile alcohols and halides carefully to prevent side reactions in the subsequent steps, ensuring that the reaction environment remains strictly anhydrous until the final decarboxylation stage. The detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these high-efficiency results.
- React dimethyl malonate with a base and isopropyl halide in an aprotic solvent, then distill off the solvent and byproduct alcohol.
- In the same vessel, react the intermediate with base and (1E)-1,3-dichloro-1-propene, followed by distillation to remove unreacted starting materials.
- Heat the resulting mixture with water to effect decarboxylation, utilizing the in-situ generated alkali metal chloride salt as a catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers tangible strategic benefits that extend beyond simple chemical yield. By transitioning from a batch process with aqueous workups to a continuous distillation-based workflow, manufacturers can significantly reduce the total cost of ownership for this critical intermediate. The elimination of wastewater treatment associated with solvent-laden aqueous streams translates directly into lower operational expenditures and reduced regulatory compliance burdens. Moreover, the ability to recover and recycle high-value solvents like sulfolane and toluene creates a closed-loop system that insulates the production process from volatility in raw material pricing. This efficiency makes the technology an ideal candidate for the commercial scale-up of complex polymer additives and fine chemical intermediates where margin pressure is high.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the complete avoidance of aqueous extraction steps, which traditionally result in the loss of expensive polar solvents into the waste stream. By utilizing distillation for purification, the process allows for the quantitative recovery of the solvent system, meaning that the same batch of solvent can be reused multiple times without significant loss of performance. Additionally, the utilization of byproduct salts as catalysts for the decarboxylation step removes the need to purchase and handle external catalytic additives, further streamlining the bill of materials. These cumulative efficiencies result in substantial cost savings that can be passed down the supply chain, enhancing the competitiveness of the final API or agrochemical product.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as dimethyl malonate, isopropyl halides, and standard alkali bases ensures that the raw material supply is robust and less susceptible to geopolitical disruptions. Unlike processes that depend on specialized transition metal catalysts or exotic reagents, this methodology utilizes widely available industrial inputs that can be sourced from multiple global suppliers. This diversification of the supply base reduces the risk of production stoppages due to raw material shortages. Furthermore, the simplified processing equipment requirements—primarily reactors equipped with distillation columns rather than complex extraction centrifuges—mean that production can be easily transferred between different manufacturing sites without extensive requalification, ensuring consistent supply continuity.
- Scalability and Environmental Compliance: From an environmental perspective, the reduction in wastewater volume is a critical advantage, as it minimizes the load on effluent treatment plants and reduces the facility's overall environmental footprint. The process generates significantly less saline waste, which is often difficult and costly to treat in industrial settings. This aligns with increasingly stringent global environmental regulations regarding solvent emissions and liquid waste discharge. The scalability of the process is also enhanced by the simplicity of the workup; distillation is a unit operation that scales linearly and predictably from pilot plant to full commercial production, unlike liquid-liquid extractions which can suffer from emulsion issues and phase separation delays at larger scales.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages for potential partners. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing manufacturing portfolios.
Q: How does this patent improve solvent recovery compared to traditional methods?
A: Traditional methods often require aqueous quenching which dissolves polar solvents like sulfolane, making recovery difficult. This patented process utilizes distillation to remove solvents and byproducts, allowing for near-quantitative recovery and reuse of expensive aprotic solvents.
Q: What is the role of the in-situ generated salt in the decarboxylation step?
A: The alkali metal chloride (e.g., NaCl or KCl) generated during the initial alkylation steps acts as a reaction accelerator for the subsequent decarboxylation. This eliminates the need to add external salts, simplifying the process and reducing raw material costs.
Q: Can this process be scaled for industrial production of chiral intermediates?
A: Yes, the process is designed for scalability. The elimination of aqueous extraction steps reduces wastewater generation significantly, and the final optical resolution can be performed enzymatically to achieve high enantiomeric excess suitable for API manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (4E)-5-Chloro-2-isopropyl-4-pentenoic Acid Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the development of next-generation therapeutics and crop protection solutions. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of (4E)-5-chloro-2-isopropyl-4-pentenoic acid ester meets the exacting standards required for GMP manufacturing. Our commitment to quality ensures that your downstream synthesis proceeds without interruption due to impurity-related failures.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific project needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing process can enhance your supply chain efficiency and reduce your overall production costs.
