Advanced Synthesis of 17-Carboxylic Acid Steroids for High-Purity Pharmaceutical Intermediates
Advanced Synthesis of 17-Carboxylic Acid Steroids for High-Purity Pharmaceutical Intermediates
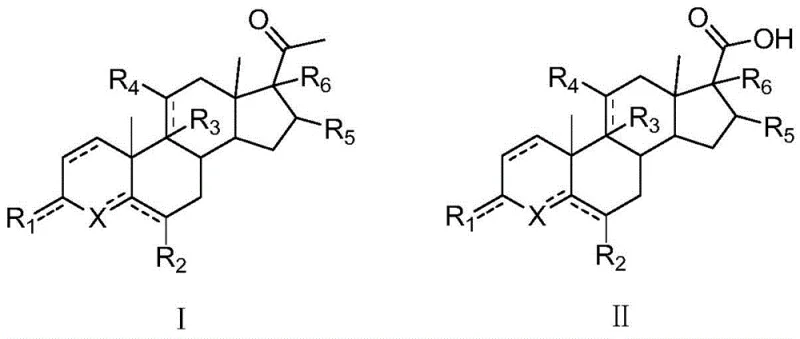
The pharmaceutical industry constantly seeks more efficient and safer pathways for synthesizing critical steroid intermediates, particularly those serving as precursors for hormones and anti-androgen drugs like finasteride. A significant breakthrough in this domain is detailed in patent CN109369762B, which discloses a novel preparation method for 17-formic acid steroid compounds. This technology fundamentally shifts the paradigm from hazardous traditional haloform reactions to a safer, solid-reagent-based trihalogenation process. By utilizing 17-ethanone steroid compounds (Formula I) as starting materials, the method achieves a direct conversion to 17-carboxylic acid steroids (Formula II) under mild alkaline conditions. This innovation not only addresses the severe safety concerns associated with volatile liquid halogens but also delivers exceptional chemical performance, with yields reportedly reaching 95.3% and crude purity levels of 96.5%. For R&D directors and procurement specialists, this represents a pivotal opportunity to optimize the supply chain for high-value steroid intermediates while drastically reducing operational risks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 17-carboxylic acid moiety on the steroid backbone has relied heavily on the classical haloform reaction using elemental halogens such as liquid bromine or chlorine gas. While chemically effective, these reagents pose monumental challenges for modern chemical manufacturing and supply chain management. Liquid bromine is highly corrosive, toxic, and volatile, requiring specialized storage facilities, rigorous containment protocols, and expensive scrubbing systems to manage emissions. Furthermore, the transportation of such hazardous materials is heavily regulated, leading to increased logistical costs and potential supply disruptions. From an environmental perspective, the use of these reagents generates significant waste streams that are difficult to treat, conflicting with the increasingly stringent green chemistry mandates imposed by global regulatory bodies. These factors collectively inflate the cost of goods sold (COGS) and introduce unacceptable safety liabilities for large-scale production facilities.
The Novel Approach
The patented process introduces a transformative alternative by replacing dangerous liquid halogens with stable, solid halogenating agents such as N-bromosuccinimide (NBS), N-chlorosuccinimide (NCS), or dichlorohydantoin. This substitution fundamentally alters the safety profile of the reaction, eliminating the risks of vapor inhalation and corrosive spills. The reaction proceeds through a controlled trihalogenation at the 21-position of the steroid side chain, followed by in-situ hydrolysis to form the carboxylic acid. This approach allows for precise stoichiometric control, minimizing excess reagent usage and simplifying downstream purification. The mild reaction conditions, typically ranging from 0°C to 60°C, ensure that sensitive functional groups elsewhere on the complex steroid nucleus remain intact, thereby preserving the structural integrity required for downstream API synthesis. This method exemplifies how strategic reagent selection can drive both safety and efficiency in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Alkaline Trihalogenation and Hydrolysis
The core of this synthetic strategy lies in the enhanced acidity of the alpha-hydrogens adjacent to the carbonyl group upon initial halogenation. When the first halogen atom is introduced to the methyl group at the C21 position, its strong electron-withdrawing inductive effect significantly increases the acidity of the remaining alpha-hydrogens. This activation facilitates the rapid and sequential substitution of the second and third hydrogen atoms by halogen species under alkaline catalysis. The resulting trihalomethyl ketone intermediate is inherently unstable in the basic medium. The hydroxide ion attacks the carbonyl carbon, leading to the cleavage of the carbon-carbon bond between the steroid nucleus and the trihalomethyl group. This cleavage releases a trihalomethane byproduct (such as bromoform) and generates the corresponding carboxylate salt of the steroid, which is subsequently protonated to yield the target 17-carboxylic acid.
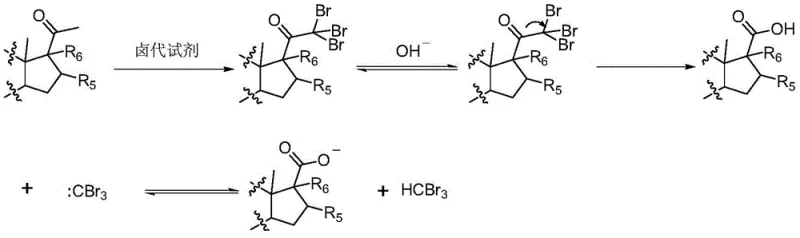
From an impurity control perspective, this mechanism offers distinct advantages over traditional methods. The use of solid reagents like NBS or NCS minimizes the formation of radical species that often lead to unpredictable side reactions on the steroid ring system, such as unwanted allylic halogenation. The patent data indicates that specific structural features, such as conjugated double bonds or amide functionalities in the A-ring, further suppress side reactions by lowering the activity of other alpha-hydrogens. For instance, when R1 is a carbonyl and X is NH (as in azasteroids), the amide character reduces enolization potential at other sites, ensuring high regioselectivity for the C21 position. This inherent selectivity translates to a cleaner reaction profile, reducing the burden on purification teams and ensuring that the final high-purity pharmaceutical intermediate meets strict specifications without extensive chromatographic separation.
How to Synthesize 17-Carboxylic Acid Steroids Efficiently
Implementing this synthesis route requires careful attention to reagent addition rates and temperature control to manage the exothermic nature of the halogenation. The process typically begins by dissolving the 17-acetyl steroid substrate in a mixed solvent system, such as dioxane and water or tert-butanol and water, to ensure homogeneity. An alkaline reagent, preferably sodium hydroxide or potassium hydroxide, is added to establish the necessary basic environment. The solid halogenating agent is then introduced in batches to maintain thermal stability, preventing local hot spots that could degrade the product. Following the completion of trihalogenation, confirmed by TLC, the mixture is stirred to allow hydrolysis to proceed to completion.
- Perform a 21-site trihalogenation reaction on the 17-acetyl steroid starting material using a solid halogenating reagent (e.g., NBS, NCS) in an organic solvent/water mixture under alkaline conditions.
- Proceed directly to hydrolysis under the same alkaline conditions to cleave the trihalomethyl group, forming the carboxylate salt.
- Acidify the reaction mixture to pH ≤ 7, concentrate the solvent, and induce crystallization by cooling to isolate the high-purity 17-carboxylic acid steroid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers substantial strategic benefits beyond mere chemical yield. The shift from hazardous liquid halogens to solid reagents dramatically simplifies the logistics of raw material acquisition and storage. Solid reagents like NBS and NCS are stable, non-volatile, and easy to handle, which eliminates the need for specialized corrosion-resistant storage tanks and complex ventilation systems. This reduction in infrastructure requirements directly contributes to cost reduction in pharmaceutical intermediate manufacturing by lowering capital expenditure (CAPEX) on safety equipment and reducing ongoing maintenance costs. Furthermore, the reliability of the supply chain is enhanced, as solid reagents are less susceptible to shipping restrictions and delays compared to regulated hazardous liquids.
- Cost Reduction in Manufacturing: The elimination of expensive hazard mitigation measures associated with liquid bromine or chlorine leads to significant operational savings. By avoiding the need for specialized scrubbing systems and personal protective equipment (PPE) required for toxic gases, manufacturers can allocate resources more efficiently. Additionally, the high yield (up to 95.3%) and high crude purity (96.5%) minimize material loss and reduce the consumption of solvents and energy during purification, further driving down the unit cost of production.
- Enhanced Supply Chain Reliability: Utilizing widely available, stable solid reagents ensures a consistent and uninterrupted supply of critical inputs. Unlike liquid halogens, which may face strict transportation quotas or seasonal supply fluctuations, solid halogenating agents can be stocked in bulk without degradation. This stability allows for better production planning and reducing lead time for high-purity pharmaceutical intermediates, enabling manufacturers to respond more agilely to market demands for steroid APIs.
- Scalability and Environmental Compliance: The mild reaction conditions (0-60°C) and the absence of toxic gas evolution make this process highly amenable to commercial scale-up of complex steroid intermediates. The simplified waste profile, devoid of heavy halogen loads, eases the burden on wastewater treatment facilities and ensures compliance with increasingly rigorous environmental regulations. This sustainability advantage not only mitigates regulatory risk but also enhances the corporate social responsibility (CSR) profile of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis technology. These insights are derived directly from the experimental data and claims within the patent literature, providing a factual basis for evaluating the process viability. Understanding these details is crucial for technical teams assessing the feasibility of integrating this route into existing production lines.
Q: Why is this new haloform reaction method safer than traditional methods?
A: Traditional methods utilize liquid halogens like bromine or chlorine, which are highly toxic, volatile, and difficult to transport. This patented process employs solid halogenating agents such as N-bromosuccinimide (NBS) or N-chlorosuccinimide (NCS), which are non-volatile, stable, and significantly reduce environmental and safety hazards during large-scale production.
Q: What yields and purity levels can be expected from this synthesis route?
A: The process demonstrates exceptional efficiency, with reported yields reaching up to 95.3% and crude product purity achieving 96.5%. Specific examples in the patent data show isolated yields of 96% with purities up to 98% after simple crystallization, indicating a robust and high-quality manufacturing pathway.
Q: Is this process suitable for commercial scale-up of complex steroids?
A: Yes, the reaction conditions are mild, typically operating between 0°C and 60°C, which facilitates easy thermal control in large reactors. The use of common solvents like dioxane or tert-butanol and the elimination of hazardous gas handling make this method highly scalable for industrial API intermediate production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 17-Carboxylic Acid Steroid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and safe synthetic routes in the production of life-saving medications. Our technical team has thoroughly analyzed the potential of this alkaline trihalogenation technology and is fully prepared to implement it for our global partners. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 17-carboxylic acid steroid meets the highest international standards for API intermediates.
We invite you to collaborate with us to leverage this advanced chemistry for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your supply chain is built on a foundation of safety, quality, and economic efficiency.
