Scalable Synthesis of Perampanel Intermediate via Safe Palladium-Catalyzed Cyanation for Commercial Production
The pharmaceutical landscape for central nervous system disorders continues to evolve, with Perampanel standing out as a critical therapeutic agent for epilepsy management. As detailed in patent CN114605286A, a significant technological breakthrough has been achieved in the synthesis of its key precursor, 2-cyanophenylacetanilide. This patent outlines a robust, two-step synthetic strategy that fundamentally shifts the manufacturing paradigm from hazardous, high-cost legacy methods to a safer, more economically viable process suitable for global supply chains. By leveraging palladium-catalyzed cyanation with potassium ferrocyanide, this innovation addresses the longstanding bottlenecks of toxicity and raw material scarcity that have plagued the production of this vital neurological drug intermediate.
For R&D directors and process chemists, the implications of this methodology extend beyond mere academic interest; it represents a tangible pathway to securing high-purity intermediates with a superior impurity profile. The transition from corrosive thionyl chloride protocols and pyrophoric organometallic reagents to a stable, aqueous-compatible cyanation source marks a maturity in process chemistry that aligns perfectly with modern green chemistry principles. This report delves deep into the mechanistic advantages and commercial scalability of this novel route, providing a comprehensive analysis for stakeholders aiming to optimize their API manufacturing strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
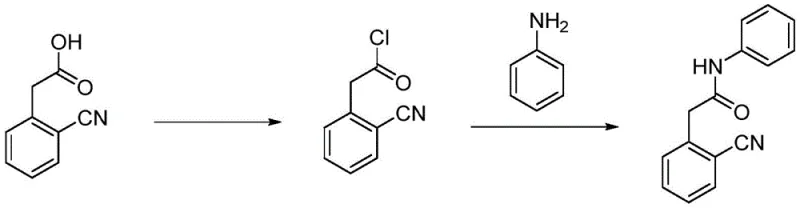
Historically, the synthesis of 2-cyanophenylacetanilide has been fraught with significant operational hazards and economic inefficiencies, primarily stemming from two dominant prior art routes. The first conventional method, illustrated below, relies on the activation of 2-cyanophenylacetic acid using thionyl chloride to form an acid chloride, which is subsequently amidated with aniline. This approach is severely limited by the high cost and market scarcity of the starting material, 2-cyanophenylacetic acid, which is not readily available in bulk quantities. Furthermore, the use of thionyl chloride introduces severe corrosion risks to reactor equipment and generates hazardous acidic waste streams that require complex neutralization and disposal protocols, rendering the process environmentally unsustainable for large-scale operations.
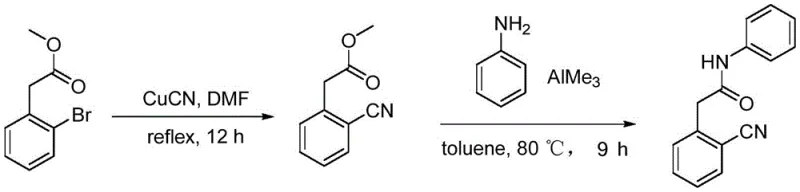
The second legacy route attempts to bypass the expensive acid starting material by starting with methyl 2-bromobenzoate, followed by a Rosenmund-von Braun type cyanation using cuprous cyanide (CuCN). As depicted in the reaction scheme below, this method suffers from even more critical safety defects. Cuprous cyanide is a highly toxic substance that poses acute health risks to operators and creates heavy metal contamination issues in the final product. Moreover, the subsequent amidation step utilizes trimethyl aluminum (AlMe3) as a catalyst, a reagent known for its pyrophoric nature and violent reactivity with moisture. These factors combine to create a process with unacceptable safety profiles for modern GMP facilities, necessitating expensive specialized containment systems and driving up the overall cost of goods significantly.
The Novel Approach
In stark contrast to these hazardous legacy methods, the novel approach disclosed in CN114605286A introduces a strategic reversal of synthetic logic that prioritizes safety and availability. Instead of installing the cyano group early on dangerous substrates, this method first constructs the amide backbone using readily available 2-bromophenylacetic acid and aniline. This condensation is achieved under mild conditions using standard carbodiimide coupling agents like EDCI, avoiding the need for corrosive chlorinating agents entirely. The resulting 2-bromobenzene acetanilide is a stable, crystalline solid that can be easily purified before undergoing the critical functional group transformation. This stepwise isolation allows for rigorous quality control of the intermediate, ensuring that impurities do not carry over into the final cyanation step, thereby enhancing the overall purity of the API precursor.
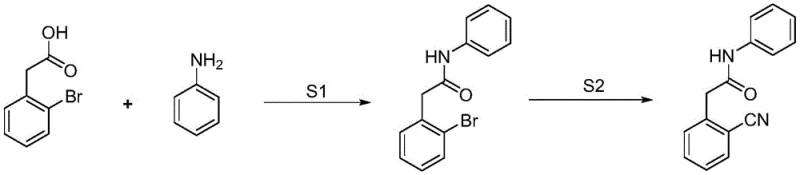
The core innovation lies in the second step, where the bromo-substituent is converted to a cyano group using potassium ferrocyanide trihydrate. This reagent serves as a non-toxic, stable source of cyanide ions, eliminating the handling risks associated with alkali metal cyanides or cuprous cyanide. Catalyzed by a palladium system with specific phosphine ligands, this transformation proceeds efficiently in polar aprotic solvents like DMAc. The result is a process that not only mitigates the safety risks of pyrophoric reagents but also leverages commodity chemicals that are accessible from multiple global suppliers. This shift effectively decouples the production of Perampanel intermediates from niche, high-cost supply chains, offering a robust solution for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Palladium-Catalyzed Cyanation
The success of this novel synthetic route hinges on the efficiency of the palladium-catalyzed cyanation step, which operates through a well-defined catalytic cycle involving oxidative addition, ligand exchange, and reductive elimination. In this specific embodiment, palladium acetate serves as the precatalyst, which is activated in situ by the phosphine ligand, preferably tri-o-tolylphosphine (P(o-tol)3) or DPPF. The steric bulk and electronic properties of these ligands are crucial; they facilitate the oxidative addition of the aryl bromide bond to the palladium center while stabilizing the active catalytic species against aggregation into inactive palladium black. The use of potassium ferrocyanide as the cyanide source is particularly elegant, as it releases cyanide ions slowly under the reaction conditions, maintaining a low concentration of free cyanide that minimizes side reactions such as hydrolysis of the nitrile group to the corresponding amide or acid.
Furthermore, the reaction conditions specified in the patent, particularly the temperature range of 120-130°C in DMAc, are optimized to drive the equilibrium towards the desired nitrile product while maintaining catalyst longevity. The presence of a base, such as anhydrous sodium carbonate, is essential to neutralize the hydrogen bromide generated during the cycle, preventing the protonation of the amine ligands or the substrate which could inhibit the reaction. From an impurity control perspective, this mechanism is superior because the mild nature of the ferrocyanide source prevents the formation of bis-cyanated byproducts or homocoupling impurities often seen with more aggressive cyanating agents. This high selectivity translates directly to a cleaner crude product, reducing the burden on downstream purification processes and improving the overall yield of the commercial process.
How to Synthesize 2-cyanophenylacetanilide Efficiently
The practical implementation of this synthesis involves a straightforward two-step protocol that is highly amenable to standard multipurpose reactor setups found in most CDMO facilities. The initial amidation step is performed at ambient or slightly elevated temperatures (20-60°C) in solvents like acetone or acetonitrile, utilizing EDCI as a coupling agent to activate the carboxylic acid. Following the isolation of the bromo-intermediate, the cyanation is conducted in a sealed vessel capable of withstanding moderate pressure and temperatures up to 130°C. The detailed standardized operating procedures, including precise molar ratios, addition rates, and workup protocols necessary to replicate the high yields reported in the patent examples, are outlined in the technical guide below.
- Condense 2-bromobenzene acetic acid with aniline using EDCI in acetone or acetonitrile to form 2-bromobenzene acetanilide.
- React the bromo-intermediate with potassium ferrocyanide trihydrate in DMAc using palladium acetate and P(o-tol)3 ligand at 120°C.
- Purify the final product via filtration, aqueous workup, and vacuum drying to obtain high-purity 2-cyanophenylacetanilide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers transformative benefits that extend far beyond the laboratory bench. The primary advantage lies in the radical simplification of the raw material supply chain; by replacing scarce, expensive specialty chemicals like 2-cyanophenylacetic acid with bulk commodity chemicals like 2-bromophenylacetic acid, manufacturers can secure a more stable and cost-effective supply base. This shift reduces dependency on single-source suppliers and mitigates the risk of price volatility associated with niche intermediates. Additionally, the elimination of hazardous reagents such as thionyl chloride and trimethyl aluminum drastically lowers the operational costs related to safety compliance, waste disposal, and equipment maintenance, contributing to substantial cost savings in API manufacturing.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the replacement of high-cost starting materials with widely available commodity chemicals. By utilizing 2-bromophenylacetic acid, which is produced on a multi-ton scale globally, the raw material cost basis is significantly lowered compared to the proprietary or scarce precursors required by legacy routes. Furthermore, the avoidance of expensive and hazardous catalysts like trimethyl aluminum removes the need for specialized storage and handling infrastructure, leading to reduced capital expenditure and lower overheads. The high atom economy of the palladium-catalyzed step also ensures that reagent consumption is minimized, further optimizing the cost structure for large-scale production runs.
- Enhanced Supply Chain Reliability: Supply chain resilience is markedly improved by the use of robust, non-hazardous reagents that are not subject to strict transportation regulations or seasonal availability fluctuations. Potassium ferrocyanide is a stable solid that can be shipped and stored without the stringent controls required for liquid cyanides or pyrophoric organometallics. This ease of logistics ensures continuous production capability even during periods of global supply disruption. Moreover, the simplicity of the reaction workup, which involves standard filtration and extraction techniques rather than complex distillations or chromatographic separations, shortens the manufacturing cycle time, allowing for faster turnaround and more responsive inventory management to meet market demand.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this process aligns perfectly with increasingly stringent global regulatory standards. The absence of heavy metal contaminants like copper and the elimination of corrosive gas generation simplify the wastewater treatment process, reducing the environmental footprint of the manufacturing site. The process is inherently safer, removing the risk of runaway exotherms associated with pyrophoric reagents, which facilitates easier scale-up from pilot plant to commercial tonnage. This safety profile not only protects personnel but also ensures uninterrupted production schedules by minimizing the risk of safety-related shutdowns or regulatory inspections halting operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on the feasibility and advantages of adopting this method for commercial production. Understanding these details is crucial for technical teams evaluating the integration of this process into existing manufacturing lines.
Q: Why is potassium ferrocyanide preferred over cuprous cyanide for this synthesis?
A: Potassium ferrocyanide is a non-toxic, stable solid, whereas cuprous cyanide is highly toxic and poses significant environmental and safety hazards during industrial handling and waste disposal.
Q: What are the safety advantages of avoiding trimethyl aluminum in this route?
A: Trimethyl aluminum is pyrophoric and explodes upon contact with water, requiring specialized inert atmosphere equipment. The new route eliminates this risk entirely, allowing for standard reactor usage.
Q: Is the starting material 2-bromophenylacetic acid commercially available?
A: Yes, unlike the expensive and scarce 2-cyanophenylacetic acid used in prior art, 2-bromophenylacetic acid is a bulk commodity chemical with a stable global supply chain.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-cyanophenylacetanilide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable supply of high-quality intermediates for the global epilepsy treatment market. Our team of expert process chemists has thoroughly analyzed the technology disclosed in CN114605286A and is fully prepared to execute this advanced palladium-catalyzed route on a commercial scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 2-cyanophenylacetanilide meets the highest industry standards for downstream API synthesis.
We invite you to collaborate with us to leverage this innovative technology for your supply chain. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage potential partners to contact us directly to request specific COA data and route feasibility assessments, allowing us to demonstrate how our mastery of this safe, efficient synthesis can drive value and reliability in your Perampanel production strategy.
