Scalable Electrochemical Production of High-Purity 4-Iodo-5-Aminopyrazole Intermediates
The pharmaceutical and agrochemical industries rely heavily on functionalized heterocycles, particularly 4-iodo-5-aminopyrazoles, which serve as pivotal building blocks for constructing complex biologically active molecules through cross-coupling reactions. A significant technological breakthrough in this domain is detailed in patent CN115537855A, which discloses a novel electrochemical synthesis method that fundamentally alters the production landscape for these critical intermediates. Unlike traditional approaches that depend on costly and wasteful stoichiometric oxidants, this invention utilizes simple potassium iodide and electricity to drive the iodination process with remarkable efficiency. The core innovation lies in the ability to generate reactive iodine species in situ at the anode, facilitating a highly selective electrophilic substitution on the pyrazole ring without the need for external chemical oxidants. This shift towards electro-organic synthesis not only aligns with the principles of green chemistry by minimizing waste but also offers a robust pathway for producing high-purity intermediates essential for downstream drug development. 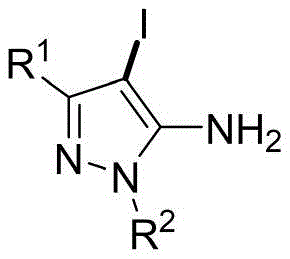
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-iodo-5-aminopyrazoles has predominantly relied on the use of hypervalent iodine reagents, such as N-Iodosuccinimide (NIS), to introduce the iodine atom onto the heterocyclic core. While chemically effective, this conventional methodology suffers from severe drawbacks regarding atom economy and environmental impact, as it generates stoichiometric amounts of succinimide by-products that complicate purification and increase waste disposal costs. Furthermore, the high cost of hypervalent iodine reagents significantly inflates the raw material expenses, making large-scale manufacturing economically challenging for cost-sensitive applications. The reaction systems often require careful control to prevent over-iodination or decomposition, and the handling of these oxidizing agents necessitates strict safety protocols that add operational complexity to the manufacturing process. Consequently, there is a pressing industrial need for a more sustainable and economically viable alternative that can deliver the same high-quality intermediates without the associated baggage of traditional chemical oxidants.
The Novel Approach
The electrochemical method described in the patent data presents a transformative solution by replacing chemical oxidants with electrons, utilizing inexpensive potassium iodide as the iodine source in a one-pot reaction setup. By employing a platinum electrode system in a mixed solvent of acetonitrile and water, the process generates the necessary electrophilic iodine species directly at the anode surface through controlled oxidation. This approach drastically simplifies the reaction workup, as the only by-product is hydrogen gas at the cathode, thereby eliminating the formation of organic waste salts typical of NIS reactions. The method demonstrates exceptional versatility, accommodating a wide range of substrates including those with methyl, tert-butyl, and phenyl substituents, achieving isolated yields as high as 95% under mild room temperature conditions. 
Mechanistic Insights into Electrochemical Anodic Iodination
The underlying mechanism of this transformation involves the anodic oxidation of iodide ions (I-) to generate reactive electrophilic iodine species, likely molecular iodine (I2) or higher oxidation state cationic intermediates, directly within the reaction medium. These electro-generated species then undergo electrophilic aromatic substitution with the electron-rich 5-aminopyrazole substrate, preferentially attacking the 4-position due to the activating effect of the amino group and the electronic properties of the pyrazole ring. The use of a platinum electrode is critical, as it provides a stable surface for electron transfer without participating in side reactions, ensuring that the current efficiency is directed towards the desired iodination pathway. The presence of water in the acetonitrile solvent system aids in the conductivity of the solution and may play a role in stabilizing the transition states or facilitating proton transfer steps during the regeneration of the aromatic system. This precise control over the oxidation potential allows for high regioselectivity, minimizing the formation of di-iodinated by-products that often plague traditional chemical iodination methods.
From an impurity control perspective, the mild nature of the electrochemical conditions prevents the degradation of sensitive functional groups that might occur under harsher chemical oxidation environments. The absence of strong chemical oxidants reduces the risk of over-oxidation of the amino group or the cleavage of protecting groups, such as the Boc group seen in specific embodiments of the patent. Furthermore, the continuous removal of protons at the cathode helps maintain a neutral pH environment, which is beneficial for the stability of the aminopyrazole scaffold. The simplicity of the reaction mixture, consisting mainly of the substrate, KI, and solvent, means that the final product profile is cleaner, requiring less aggressive purification steps to meet stringent pharmaceutical specifications. This inherent cleanliness of the electrochemical route translates directly into higher overall process efficiency and reduced loss of material during isolation.
How to Synthesize 4-Iodo-5-Aminopyrazoles Efficiently
The synthesis protocol outlined in the patent provides a straightforward and reproducible method for accessing these valuable intermediates using standard laboratory electrochemical equipment. The process begins by dissolving the 5-aminopyrazole starting material and potassium iodide in a mixture of acetonitrile and water, followed by the immersion of platinum electrodes connected to a constant current power supply. Reaction progress is conveniently monitored via TLC, allowing operators to determine the exact endpoint to maximize yield while minimizing energy consumption. Following the electrolysis, the workup involves simple solvent removal and purification via silica gel column chromatography, yielding the pure 4-iodo product ready for subsequent coupling reactions.
- Prepare the electrolytic cell by adding potassium iodide, 5-aminopyrazole substrate, acetonitrile, and water, then install platinum electrodes.
- Apply a constant current (e.g., 5mA) at room temperature with continuous stirring to drive the anodic oxidation and electrophilic substitution.
- Upon completion, concentrate the reaction mixture under reduced pressure and purify the crude residue via column chromatography using petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this electrochemical technology offers substantial strategic benefits that extend beyond mere technical feasibility into the realm of cost optimization and supply security. The replacement of expensive hypervalent iodine reagents with commodity-grade potassium iodide represents a direct and significant reduction in raw material costs, which is a critical factor in maintaining competitive pricing for API intermediates. Additionally, the elimination of succinimide waste streams simplifies environmental compliance and reduces the financial burden associated with hazardous waste disposal, contributing to a lower total cost of ownership for the manufacturing process. The scalability of electrochemical reactions is well-established, and the ability to run this synthesis at room temperature reduces energy consumption related to heating or cooling, further enhancing the economic profile of the production line.
- Cost Reduction in Manufacturing: The substitution of high-cost stoichiometric oxidants with inexpensive potassium iodide and electricity fundamentally alters the cost structure of producing 4-iodo-5-aminopyrazoles. By removing the need for NIS, manufacturers avoid the volatility of specialty reagent pricing and the hidden costs associated with the disposal of nitrogen-containing organic waste. The high atom economy of the electrochemical process ensures that a greater proportion of the input mass is converted into the desired product, reducing the effective cost per kilogram of the final intermediate. Furthermore, the simplified purification process resulting from a cleaner reaction profile lowers the consumption of chromatography media and solvents, driving down operational expenditures significantly.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like potassium iodide and standard solvents mitigates the risk of supply chain disruptions that often affect specialized reagents like NIS. The robustness of the electrochemical method, which tolerates various substrates and operates under mild conditions, ensures consistent production output even with slight variations in raw material quality. This reliability is crucial for maintaining continuous supply to downstream customers who depend on these intermediates for their own synthesis timelines. The ability to scale the process from gram to kilogram quantities using modular electrochemical reactors provides flexibility to respond quickly to fluctuating market demands without lengthy process re-validation.
- Scalability and Environmental Compliance: The green chemistry credentials of this electrochemical route align perfectly with increasingly stringent global environmental regulations, future-proofing the manufacturing process against tighter emission standards. The absence of heavy metal catalysts and toxic oxidants simplifies the wastewater treatment process, making it easier to obtain and maintain environmental permits for large-scale production facilities. The inherent safety of operating at room temperature and atmospheric pressure reduces the risk of thermal runaway incidents, lowering insurance premiums and safety management costs. This sustainable approach not only meets regulatory requirements but also enhances the brand value of the supplier as a responsible partner in the pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical synthesis of 4-iodo-5-aminopyrazoles, based on the detailed experimental data and claims found in the patent literature. Understanding these aspects is vital for R&D teams evaluating the feasibility of integrating this technology into their existing workflows. The answers provided reflect the specific conditions and outcomes reported in the patent, ensuring accuracy and relevance for technical decision-making.
Q: What are the primary advantages of this electrochemical method over traditional NIS iodination?
A: The electrochemical method eliminates the need for expensive stoichiometric hypervalent iodine reagents like N-Iodosuccinimide (NIS), significantly improving atom economy and reducing hazardous waste generation while maintaining high regioselectivity at the 4-position.
Q: What represents the optimal reaction conditions for maximizing yield in this process?
A: Optimal results are achieved using a platinum electrode system at room temperature with a constant current of 5mA in an acetonitrile-water solvent mixture, typically reaching maximum conversion within approximately 5 hours of electrolysis.
Q: Is this synthesis method compatible with diverse substituent groups on the pyrazole ring?
A: Yes, the protocol demonstrates excellent functional group tolerance, successfully accommodating various alkyl and aryl substituents at the 3-position and different N-substituents including phenyl and Boc-protected groups with yields ranging from 60% to 95%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Iodo-5-Aminopyrazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in delivering high-value pharmaceutical intermediates with superior efficiency and sustainability. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN115537855A are seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required by global regulatory bodies. We are committed to leveraging this green technology to provide our clients with a reliable supply of high-purity 4-iodo-5-aminopyrazoles that accelerate their drug development timelines.
We invite you to collaborate with us to explore how this electrochemical route can optimize your specific project requirements and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating the tangible economic benefits of switching to this greener synthesis method. Please contact us today to request specific COA data for our electrochemically synthesized intermediates and to discuss route feasibility assessments for your target molecules. Let us be your partner in navigating the complexities of modern chemical manufacturing with innovation and precision.
