Advanced Synthesis of Stable Lipoic Acid Derivatives for Commercial Pharmaceutical Applications
Introduction to Patent CN114105945A: Overcoming Stability Barriers
The pharmaceutical and nutraceutical industries have long recognized the potent antioxidant capabilities of lipoic acid, yet its widespread application has been severely hindered by intrinsic physicochemical limitations. As detailed in the recent patent CN114105945A, natural lipoic acid exhibits poor temperature stability and significant hygroscopicity, leading to rapid degradation when exposed to temperatures exceeding 40°C. This thermal instability creates substantial challenges for formulation scientists who require robust active ingredients capable of withstanding standard processing and storage conditions without losing efficacy. The patent introduces a groundbreaking synthetic methodology that transforms unstable lipoic acid into highly stable derivatives through a sophisticated two-step activation and coupling process. By converting the carboxylic acid moiety into an active ester intermediate using N-hydroxysuccinimide, the process enables mild subsequent reactions that preserve the sensitive dithiolane ring structure. This technological leap not only solves the degradation issue but also opens new avenues for cost reduction in pharmaceutical intermediate manufacturing by utilizing widely available reagents and standard equipment.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to modifying lipoic acid often rely on direct esterification or amidation techniques that necessitate harsh reaction conditions, such as high temperatures and strong acidic catalysts, which are detrimental to the molecule's integrity. The dithiolane ring, which is responsible for the compound's redox activity, is particularly susceptible to thermal decomposition and ring-opening polymerization under these aggressive conditions. Furthermore, direct coupling reactions frequently suffer from low conversion rates and generate complex impurity profiles that are difficult and expensive to remove during downstream purification. The hygroscopic nature of the native acid also complicates handling and weighing operations in large-scale manufacturing environments, leading to batch-to-batch variability. These cumulative factors result in a supply chain that is prone to disruptions, with final products often failing to meet the stringent purity specifications required for clinical applications. Consequently, manufacturers face elevated production costs and extended lead times as they struggle to mitigate these inherent chemical instabilities through elaborate protective group strategies or cryogenic processing.
The Novel Approach
The innovative strategy outlined in the patent circumvents these historical bottlenecks by employing a mild, two-stage synthetic route that prioritizes molecular stability and operational simplicity. In the first stage, lipoic acid is activated at room temperature using N-hydroxysuccinimide and N,N'-dicyclohexylcarbodiimide (DCC) in anhydrous dioxane, forming a reactive succinimidyl ester while precipitating dicyclohexylurea as a removable solid byproduct. This activation step is crucial as it lowers the energy barrier for the subsequent nucleophilic attack, allowing the second stage to proceed efficiently at moderate temperatures between 60°C and 80°C. The process then branches into two distinct pathways: aminolysis with 1,3-propylene diamine yields the amide-linked Derivative A, while transesterification with 1,3-propylene glycol produces the ester-linked Derivative B. 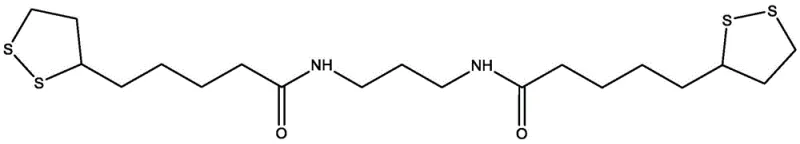
Mechanistic Insights into NHS-Mediated Activation and Coupling
The core of this synthesis lies in the precise control of nucleophilic substitution mechanisms facilitated by the succinimidyl leaving group. When the activated ester intermediate encounters the nucleophile, whether it be the amine groups of propylene diamine or the hydroxyl groups of propylene glycol, the reaction proceeds through a tetrahedral intermediate that collapses to release N-hydroxysuccinimide. This mechanism is significantly more favorable than direct acid-alcohol condensation because the succinimidyl group is a superior leaving group, driving the equilibrium towards product formation without the need for water removal techniques like Dean-Stark traps. The patent highlights the critical role of solvent engineering, specifically the addition of pyridine to the dioxane reaction medium. Pyridine serves a dual function: it acts as a base to scavenge protons generated during the nucleophilic attack, preventing acid-catalyzed degradation of the product, and it enhances the solubility of polar intermediates, ensuring a homogeneous reaction phase. 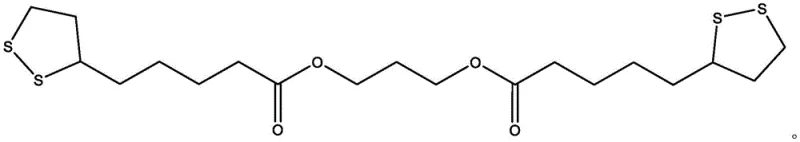
Impurity control is meticulously managed through the physical properties of the byproducts and the selection of recrystallization solvents. The primary byproduct, N,N'-dicyclohexylurea, is insoluble in dioxane and is removed via simple filtration before the coupling step, preventing it from contaminating the final API intermediate. In the second step, any unreacted starting materials or side products are effectively separated by exploiting solubility differences in hot ethanol/dichloromethane mixtures. The final purification via silica gel flash chromatography ensures that the resulting lipoic acid derivatives meet high-purity standards essential for pharmaceutical use. This rigorous approach to impurity management minimizes the risk of toxicological issues associated with residual reagents, thereby streamlining the regulatory approval process for downstream drug formulations. The ability to achieve yields ranging from 82% to 90% across different examples demonstrates the robustness of this mechanistic pathway against minor variations in reaction parameters.
How to Synthesize Lipoic Acid Derivatives Efficiently
The synthesis protocol described in the patent offers a clear roadmap for laboratory and pilot-scale production, emphasizing reproducibility and safety. The process begins with the preparation of Solution A containing the activating agents and Solution B containing the lipoic acid, which are combined under controlled stirring to manage the exotherm of the activation step. Following the removal of the urea precipitate, the crude active ester is subjected to a secondary reaction with the chosen linker molecule in the presence of the pyridine-dioxane catalytic system. The reaction progress is monitored to ensure complete consumption of the active ester, typically requiring 4 to 6 hours at elevated temperatures.
- Activate lipoic acid by reacting it with N-hydroxysuccinimide and DCC in anhydrous dioxane to form an active ester intermediate, filtering off the urea byproduct.
- React the active ester intermediate with either 1,3-propylene diamine or 1,3-propylene glycol in a dioxane-pyridine mixture at 60-80°C.
- Purify the resulting crude solid by dissolving in hot ethanol/dichloromethane, filtering impurities, and performing silica gel flash chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers profound advantages by shifting the dependency from scarce biological sources to abundant petrochemical-derived feedstocks. The primary raw materials, including N-hydroxysuccinimide, DCC, and propylene diamine, are commodity chemicals produced on a massive global scale, ensuring a stable and predictable supply chain that is immune to the seasonal fluctuations affecting agricultural extracts. This transition to fully synthetic inputs drastically simplifies inventory management and reduces the risk of supply interruptions caused by crop failures or geopolitical instability in sourcing regions. Furthermore, the elimination of complex extraction and purification steps associated with natural products leads to a significantly streamlined manufacturing workflow. The process operates at atmospheric pressure and moderate temperatures, reducing the energy footprint and capital expenditure required for specialized high-pressure or cryogenic reactors. These factors collectively contribute to substantial cost savings in pharmaceutical intermediate manufacturing, allowing buyers to negotiate more favorable pricing structures without compromising on quality.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the high atom economy of the coupling reaction and the ease of byproduct removal. Since the dicyclohexylurea byproduct precipitates out of the solution spontaneously, there is no need for expensive chromatographic separation at the intermediate stage, which significantly lowers solvent consumption and waste disposal costs. Additionally, the high yields reported in the patent examples indicate efficient utilization of raw materials, minimizing the financial loss associated with unreacted starting materials. The avoidance of precious metal catalysts, which are often required in alternative cross-coupling strategies, further eliminates the cost burden of metal scavenging and residual metal testing. These cumulative efficiencies translate into a lower cost of goods sold (COGS), providing a competitive edge in price-sensitive markets.
- Enhanced Supply Chain Reliability: The robustness of the chemical process ensures consistent output quality, which is a critical factor for supply chain planners managing just-in-time inventory systems. The use of common organic solvents like dioxane, ethanol, and dichloromethane means that solvent supply risks are minimized, as these are stocked by virtually all chemical distributors worldwide. The scalability of the reaction from gram to kilogram scales has been demonstrated through the patent examples, suggesting that technology transfer to large-scale production facilities can be achieved with minimal re-optimization. This predictability allows procurement managers to secure long-term supply agreements with confidence, knowing that the manufacturer can ramp up production volumes rapidly in response to market demand spikes without encountering technical bottlenecks.
- Scalability and Environmental Compliance: Environmental, health, and safety (EHS) considerations are increasingly becoming a deciding factor in vendor selection, and this process aligns well with modern green chemistry principles. While organic solvents are used, the ability to recycle dioxane and the solid nature of the major byproduct facilitate effective waste management strategies. The mild reaction conditions reduce the risk of thermal runaway incidents, enhancing plant safety and lowering insurance premiums. Moreover, the high stability of the final derivatives reduces the need for energy-intensive cold chain logistics during transportation and storage. This durability ensures that the product arrives at the customer's facility in optimal condition, reducing claims and returns. The overall process design supports sustainable manufacturing goals, making it an attractive option for companies aiming to reduce their carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this lipoic acid derivative synthesis technology. These answers are derived directly from the experimental data and technical disclosures found within the patent documentation, providing a reliable basis for decision-making. Understanding the nuances of solvent ratios, temperature controls, and purification methods is essential for successfully adopting this route in a commercial setting.
Q: Why is the NHS activation step critical for lipoic acid derivative synthesis?
A: Natural lipoic acid suffers from poor thermal stability and degrades above 40°C. The NHS activation step converts the carboxylic acid into a highly reactive succinimidyl ester under mild conditions, allowing subsequent coupling without the harsh heat typically required for direct amidation, thus preserving the integrity of the sensitive dithiolane ring.
Q: What role does pyridine play in the coupling reaction?
A: Pyridine acts as a basic catalyst and a co-solvent. It facilitates the nucleophilic attack by neutralizing protons released during the reaction and ensures homogeneity when mixed with dioxane. The patent specifies a volume ratio of pyridine to dioxane between 0.2:1 and 0.5:1 to optimize yield and reaction kinetics.
Q: How does this method improve supply chain reliability compared to traditional extraction?
A:
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lipoic Acid Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this patented synthesis route for the next generation of antioxidant therapeutics and nutraceuticals. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory bench to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle the specific solvent systems and reaction conditions required for NHS-mediated activations, while our rigorous QC labs enforce stringent purity specifications to guarantee batch-to-batch consistency. We understand that the stability of the dithiolane ring is paramount, and our process engineers are adept at optimizing the pyridine-dioxane ratios to maximize yield and minimize degradation products. By leveraging our deep technical expertise, we help you mitigate the risks associated with process scale-up and accelerate your time to market.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis to your specific volume and quality requirements. Request a Customized Cost-Saving Analysis today to quantify the economic benefits of switching to this stable derivative platform. Our team is ready to provide specific COA data from pilot batches and comprehensive route feasibility assessments to support your regulatory filings. Partner with us to secure a reliable supply of high-performance lipoic acid derivatives that meet the demanding standards of the global pharmaceutical industry.
