Advanced Chiral Nickel Complex Catalysis: Bridging Lab Innovation to Commercial Scale Manufacturing
The landscape of asymmetric catalysis is constantly evolving, driven by the relentless demand for high-purity chiral intermediates in the pharmaceutical and fine chemical sectors. A pivotal development in this domain is documented in patent CN103570562A, which details the synthesis and application of a novel chiral nickel complex. This specific coordination compound, identified as tris[(S)-phenylglycinol] nickel complex, represents a significant advancement in non-precious metal catalysis. Unlike traditional systems that rely heavily on expensive rhodium or palladium frameworks, this innovation leverages the abundant availability of nickel to create a robust chiral environment. The patent outlines a straightforward yet highly effective methodology where L-phenylglycinol acts as a chiral auxiliary, coordinating with nickel acetate to form a stable, octahedral geometry. For R&D directors and process chemists, this offers a compelling alternative for constructing carbon-carbon bonds, specifically in Henry reactions, where controlling stereoselectivity is paramount. The ability to achieve a conversion rate of 54.3% in benchmark reactions underscores the potential of this complex to serve as a reliable workhorse in the synthesis of complex API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral catalysts for asymmetric induction has been plagued by prohibitive costs and operational complexities. Traditional methods often necessitate the use of noble metals such as platinum, palladium, or rhodium, which not only inflate the raw material budget but also introduce stringent regulatory hurdles regarding heavy metal residues in final drug products. Furthermore, the ligands required for these noble metal systems are frequently multi-step synthetic targets themselves, requiring harsh conditions, protecting group strategies, and extensive purification protocols. This cumulative complexity results in long lead times and reduced overall atom economy. From a supply chain perspective, reliance on scarce precious metals introduces volatility; price fluctuations in the global commodities market can destabilize production budgets overnight. Additionally, the removal of trace noble metals from the final active pharmaceutical ingredient often requires specialized scavenging resins or additional chromatographic steps, further eroding profit margins and extending the manufacturing timeline. These factors collectively create a bottleneck for the commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology presented in CN103570562A offers a paradigm shift towards sustainability and economic efficiency. The novel approach utilizes nickel, a base metal that is orders of magnitude cheaper and more abundant than its noble counterparts. The synthesis is elegantly simple, described as a one-step procedure involving the direct reflux of L-phenylglycinol and nickel acetate tetrahydrate in methanol. This eliminates the need for intricate ligand synthesis or anhydrous, oxygen-free environments typically associated with sensitive organometallic chemistry. The resulting complex precipitates as distinct blue crystals upon natural volatilization, indicating a high degree of crystallinity and purity without the need for column chromatography. For procurement managers, this translates to a drastic simplification of the supply chain, as the starting materials are commodity chemicals available from multiple global vendors. The operational simplicity also means that the barrier to entry for manufacturing this catalyst is low, allowing for rapid deployment in pilot plants and facilitating a smoother transition from laboratory discovery to industrial production.
Mechanistic Insights into Ni-Catalyzed Asymmetric Henry Reaction
To fully appreciate the utility of this chiral Ni complex, one must delve into the coordination chemistry that governs its reactivity. The complex features a central nickel(II) ion coordinated by three bidentate L-phenylglycinol ligands. Each ligand binds through the nitrogen atom of the amine group and the oxygen atom of the hydroxyl group, creating a distorted octahedral geometry around the metal center. This specific arrangement creates a rigid chiral pocket that is essential for discriminating between enantiotopic faces of the substrate during the catalytic cycle. In the context of the Henry reaction (nitroaldol reaction), the nickel center acts as a Lewis acid, activating the nitroalkane by coordinating to the nitro group, thereby increasing the acidity of the alpha-protons. Simultaneously, the chiral ligands direct the approach of the aldehyde electrophile, ensuring that the new carbon-carbon bond is formed with high stereocontrol. The phenyl rings of the ligands provide steric bulk that shields specific quadrants of the metal center, effectively filtering the trajectory of the incoming reactants.
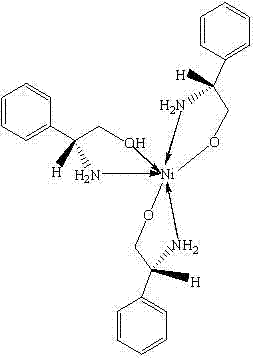
Beyond the primary catalytic cycle, the stability of this complex is a critical factor for its commercial viability. The patent data indicates strong bonding interactions, with Ni-N bond lengths ranging approximately from 2.054 Å to 2.091 Å and Ni-O bond lengths between 2.090 Å and 2.127 Å. These bond metrics suggest a thermodynamically stable complex that can withstand the thermal conditions of reflux without decomposing. For quality control teams, this stability is advantageous as it minimizes the formation of nickel black or other decomposition byproducts that could contaminate the reaction mixture. The mechanism also implies that the catalyst operates homogeneously in the initial stages, allowing for intimate contact with substrates, yet the product isolation strategy suggests that the catalyst or its byproducts can be managed effectively. Understanding these mechanistic nuances allows process engineers to optimize reaction parameters such as temperature, solvent polarity, and concentration to maximize the 54.3% conversion rate reported, potentially pushing these figures even higher through iterative process intensification.
How to Synthesize Chiral Ni Complex Efficiently
The preparation of this high-value catalyst is designed for reproducibility and ease of execution, making it an ideal candidate for technology transfer. The protocol avoids the use of exotic reagents or specialized equipment, relying instead on standard glassware and common solvents found in any synthetic laboratory. The key to success lies in the precise stoichiometric ratio of the ligand to the metal salt (3:1) and the maintenance of reflux conditions to drive the coordination equilibrium to completion. Following the reaction, the workup procedure is remarkably benign, utilizing filtration and crystallization rather than extractive workups that generate large volumes of organic waste. For detailed operational parameters, safety data, and specific troubleshooting tips regarding crystal growth, please refer to the standardized synthesis guide below.
- Dissolve L-phenylglycinol (0.03 mol) in absolute methanol (50 ml) within a 100 ml round-bottom flask under stirring conditions.
- Add nickel acetate tetrahydrate (0.01 mol) to the solution and heat the mixture to reflux for 48 hours to ensure complete coordination.
- Filter the hot reaction solution, concentrate the filtrate, dissolve the residue in absolute ethanol to form a saturated solution, and allow natural volatilization to isolate blue crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply chain resilience, the adoption of this chiral nickel technology offers profound strategic benefits. The shift from precious metal catalysis to base metal catalysis is not merely a technical adjustment but a fundamental economic optimization. By eliminating the dependency on volatile noble metal markets, manufacturers can secure more predictable costing models for their intermediate production. The simplicity of the synthesis route means that the catalyst itself can be produced in-house or sourced at a fraction of the cost of commercial chiral ligands, directly impacting the cost of goods sold (COGS). Furthermore, the robust nature of the nickel complex reduces the risk of batch failures due to catalyst degradation, enhancing overall manufacturing reliability.
- Cost Reduction in Manufacturing: The most immediate impact is seen in raw material expenditure. Nickel acetate and phenylglycinol are commodity chemicals with established, high-volume supply chains, ensuring consistent pricing and availability. Unlike proprietary chiral ligands that carry premium price tags due to intellectual property restrictions and complex synthesis, the components of this complex are generic and accessible. Additionally, the one-step synthesis eliminates the labor and utility costs associated with multi-step ligand preparation. The absence of expensive metal scavengers in the downstream processing further contributes to substantial cost savings, as the purification train is significantly shortened. This holistic reduction in operational overhead allows for a more competitive pricing structure for the final pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the geopolitical concentration of precious metal mining and refining. By pivoting to a nickel-based system, manufacturers diversify their supply risk, as nickel is produced globally in much larger quantities. The starting materials for this complex are not subject to the same export controls or scarcity issues as rhodium or iridium. Moreover, the simplified synthesis protocol reduces the dependency on highly specialized contract manufacturing organizations (CMOs) capable of handling sensitive organometallics. This democratization of the manufacturing process means that a broader network of suppliers can potentially produce the catalyst, creating a more resilient and flexible supply web that can absorb shocks from regional disruptions.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this technology aligns well with green chemistry principles. The use of methanol and ethanol as solvents is preferable to chlorinated solvents often used in traditional organometallic chemistry. The high yield of the catalyst synthesis (reported at 78%) and the crystalline nature of the product minimize waste generation. Scaling this process from grams to tons does not require exponential increases in safety infrastructure, as the reagents are not pyrophoric or excessively toxic. The reduced heavy metal load also simplifies wastewater treatment and compliance with strict environmental regulations regarding metal discharge. This ease of scale-up ensures that the transition from pilot plant to commercial production is seamless, reducing the time-to-market for new drug candidates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral nickel technology. These insights are derived directly from the experimental data and structural analysis provided in the patent literature, aiming to clarify the practical implications for industrial adoption. Understanding these details is crucial for making informed decisions about integrating this catalyst into existing production workflows.
Q: What is the catalytic efficiency of this chiral Ni complex in Henry reactions?
A: According to patent CN103570562A, the complex demonstrates favorable catalytic performance in the Henry reaction of benzaldehyde, achieving a conversion rate of 54.3%.
Q: Why is this nickel-based catalyst preferred over traditional noble metal catalysts?
A: This catalyst utilizes abundant and inexpensive nickel salts instead of costly precious metals like palladium or rhodium. Furthermore, the synthesis is a simple one-step process, significantly reducing production complexity and raw material costs.
Q: How is the stereochemical purity of the complex ensured?
A: The complex is synthesized using chiral L-phenylglycinol as the ligand source. Its structure and absolute configuration are rigorously confirmed via single-crystal X-ray diffraction analysis, ensuring high stereochemical integrity for asymmetric synthesis applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Ni Complex Supplier
While the patent CN103570562A provides the foundational intellectual property for this innovative catalyst, translating laboratory success into commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. NINGBO INNO PHARMCHEM stands at the forefront of this translation, offering comprehensive CDMO services tailored to the unique demands of chiral catalysis. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising conversion rates observed in the lab are maintained at an industrial level. We understand that consistency is key; therefore, our rigorous QC labs enforce stringent purity specifications for every batch of catalyst and intermediate we produce. Whether you require custom synthesis of the nickel complex itself or its application in the production of downstream pharmaceutical intermediates, our infrastructure is designed to support your growth.
We invite you to explore the potential of this cost-effective catalytic system for your next project. By leveraging our technical proficiency, you can accelerate your development timelines while optimizing your production costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis specific to your target molecule. We are ready to provide specific COA data and route feasibility assessments to demonstrate how this chiral nickel technology can become a cornerstone of your sustainable manufacturing strategy.
