Efficient Racemization Process for Lipid Disorder Pharmaceutical Intermediates at Commercial Scale
Introduction to Advanced Intermediate Manufacturing
The pharmaceutical industry's relentless pursuit of effective treatments for metabolic syndromes, particularly dyslipidemia associated with insulin resistance, has driven significant innovation in synthetic chemistry. Patent CN1835918A introduces a pivotal methodology for the preparation of substantially racemic 2-{[2-(4-hydroxyphenyl)ethyl]thio}-3-[4-(2-{4-[(methylsulfonyl)oxy]phenoxy}ethyl)phenyl]propionic acid, a critical intermediate in the synthesis of selective PPARα modulators. These compounds function by regulating lipid metabolism, offering therapeutic potential for complex clinical conditions. However, the presence of chiral centers in such molecules traditionally poses a significant manufacturing bottleneck, where standard resolution techniques discard half of the synthesized material. This patent addresses that inefficiency by providing a robust chemical pathway to recycle the less active or unwanted enantiomer back into the racemic pool, thereby optimizing the overall atomic economy of the production line.
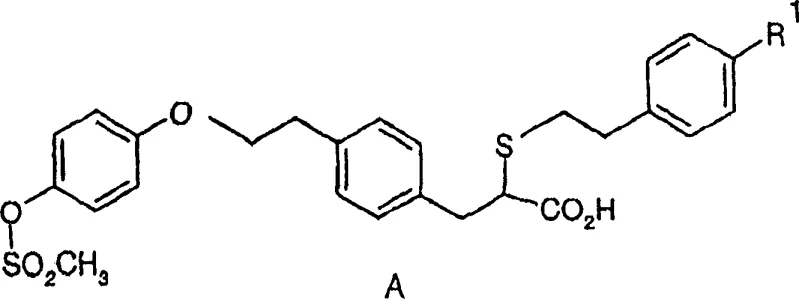
For global procurement teams and R&D directors, understanding this technology is crucial for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials without the prohibitive costs associated with wasteful chiral separation. The ability to interconvert enantiomers ensures a continuous supply loop, mitigating the risks of raw material shortages and price volatility inherent in sourcing enantiopure starting materials. By implementing this racemization strategy, manufacturers can transform a linear, wasteful process into a circular, efficient system, aligning with modern green chemistry principles while maintaining the stringent quality standards required for API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of chiral pharmaceutical intermediates like the propionic acid derivatives described in the background art has relied heavily on classical resolution methods or chiral chromatography. While effective at isolating the biologically active enantiomer, these techniques suffer from a fundamental theoretical yield limit of 50%. In a conventional resolution scenario, the desired enantiomer is separated from the racemic mixture, leaving the opposite enantiomer as a waste stream or a low-value byproduct. For high-volume commercial scale-up of complex pharmaceutical intermediates, this 50% loss translates into doubled raw material consumption, increased solvent usage for purification, and significantly higher waste disposal costs. Furthermore, chiral chromatography, while precise, is often difficult to scale economically due to the high cost of chiral stationary phases and the low throughput associated with column loading capacities.
The Novel Approach
The novel approach detailed in the patent data circumvents these limitations by introducing a chemical racemization step that targets the unwanted enantiomer directly. Instead of discarding the "wrong" isomer, the process subjects the enantiomerically enriched acid to specific reaction conditions that erase its stereochemical information, returning it to a racemic state. This racemic mixture can then be subjected to resolution again, theoretically allowing for multiple passes until nearly all starting material is converted into the desired active form. This strategy effectively decouples the yield of the final product from the 50% ceiling imposed by traditional resolution. By integrating this recycling loop, the manufacturing process achieves a level of efficiency that drastically simplifies the supply chain logistics and reduces the environmental footprint associated with producing high-purity OLED material or pharmaceutical precursors.
Mechanistic Insights into Base-Catalyzed Silylation and Racemization
The core of this technological breakthrough lies in the specific chemical mechanism employed to facilitate racemization under mild conditions. The process involves reacting the enantiomerically enriched acid with a halosilane, such as trimethylchlorosilane, in the presence of a strong nitrogenous base like 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU). This reaction sequence initially converts the carboxylic acid into a silyl ester intermediate. The formation of this silyl ester is critical because it activates the alpha-proton adjacent to the carbonyl group, making it significantly more acidic and susceptible to abstraction by the base. Once the alpha-proton is removed, a planar enolate or enol-like species is formed, which destroys the chirality at that carbon center. Upon reprotonation, the molecule can reform either the R or S configuration with equal probability, thus achieving racemization.
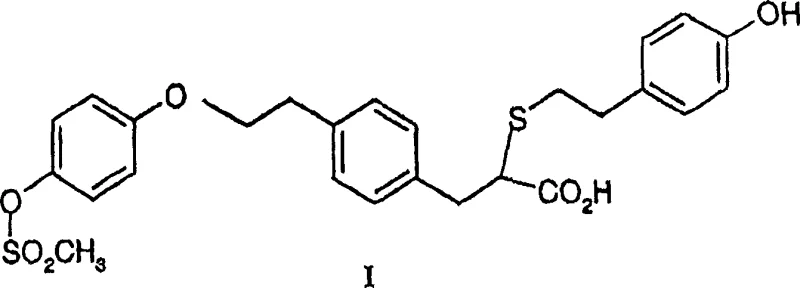
This mechanism is highly advantageous because it avoids the harsh conditions often required for thermal racemization, which can degrade sensitive functional groups present in complex molecules. The use of DBU as a non-nucleophilic base ensures that side reactions, such as nucleophilic attack on the mesylate or thioether linkages, are minimized. The reaction proceeds efficiently in inert solvents like toluene or tetrahydrofuran at temperatures ranging from 0°C to 150°C, with room temperature often being sufficient for completion. Following the racemization of the silyl ester, a simple hydrolysis step—using either aqueous acid or base—cleaves the silyl group to regenerate the free carboxylic acid, now in a substantially racemic form ready for re-processing.
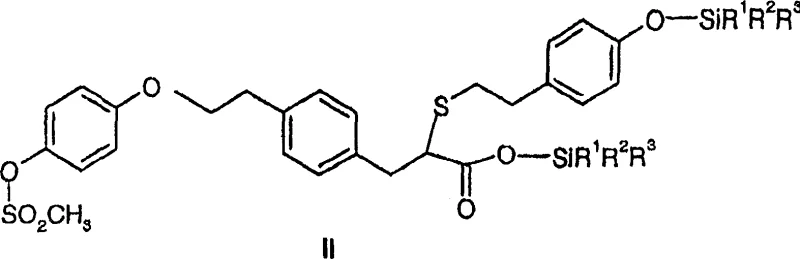
How to Synthesize Racemic Propionic Acid Derivatives Efficiently
Implementing this synthesis route requires precise control over reaction parameters to ensure maximum conversion and minimal impurity formation. The standardized protocol involves dissolving the enriched enantiomer in a dry, inert solvent system, typically a mixture of toluene and THF, under a nitrogen atmosphere to prevent moisture interference. The addition of the base and halosilane must be controlled to manage the exotherm and ensure uniform mixing. Detailed operational procedures, including specific molar ratios and workup techniques to isolate the product from the reaction mixture, are essential for reproducibility. For a comprehensive guide on executing this transformation with optimal yield and purity, please refer to the standardized synthesis steps outlined below.
- React the enantiomerically enriched acid with a halosilane (e.g., trimethylchlorosilane) and a nitrogenous base (e.g., DBU) in an inert solvent.
- Maintain the reaction mixture at a temperature between 0°C and 150°C to facilitate the formation of the silyl ester intermediate and subsequent racemization.
- Hydrolyze the resulting silyl ester intermediate using acid or base to recover the substantially racemic free acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this racemization technology offers profound benefits for cost reduction in pharmaceutical intermediate manufacturing. By enabling the recycling of the unwanted enantiomer, the process effectively doubles the utility of the initial chiral building blocks. This leads to a substantial reduction in the cost of goods sold (COGS), as the expenditure on expensive chiral starting materials is amortized over a much larger output of final product. Furthermore, the reagents utilized, such as DBU and trimethylchlorosilane, are commodity chemicals available in bulk quantities, ensuring that the process does not rely on scarce or proprietary catalysts that could introduce supply chain vulnerabilities.
- Cost Reduction in Manufacturing: The elimination of the 50% waste stream inherent in traditional resolution directly translates to lower raw material costs per kilogram of finished API. Additionally, the simplified downstream processing reduces solvent consumption and energy usage associated with separating and disposing of large volumes of unwanted isomers. This efficiency gain allows for more competitive pricing models without compromising margin, providing a strategic advantage in tender negotiations for large-scale contracts.
- Enhanced Supply Chain Reliability: Relying on a process that maximizes material throughput reduces the dependency on external suppliers for enantiopure precursors, which can often have long lead times. The ability to internally recycle material creates a buffer against supply disruptions, ensuring consistent delivery schedules for downstream API manufacturers. This reliability is critical for maintaining uninterrupted production lines in the fast-paced pharmaceutical sector, where delays can have cascading effects on drug launch timelines.
- Scalability and Environmental Compliance: The reaction conditions are amenable to large-scale batch processing, utilizing standard stainless steel reactors found in most multipurpose chemical plants. The absence of heavy metal catalysts simplifies the purification process and reduces the burden of residual metal testing, a common regulatory hurdle. Moreover, the improved atom economy aligns with increasingly stringent environmental regulations regarding waste generation, facilitating smoother regulatory approvals and enhancing the sustainability profile of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this racemization technology. Understanding these details helps stakeholders evaluate the feasibility of integrating this process into their existing manufacturing workflows. The answers are derived directly from the experimental data and claims presented in the patent literature, ensuring accuracy and relevance for technical decision-makers.
Q: Why is racemization preferred over simple resolution for this intermediate?
A: Traditional resolution discards 50% of the material as the unwanted enantiomer. Racemization allows this unwanted portion to be recycled back into the process, theoretically doubling the yield from the starting material and significantly reducing raw material costs.
Q: What are the key reagents required for this racemization process?
A: The process utilizes commercially available reagents including a nitrogenous base such as 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU) and a halosilane like trimethylchlorosilane, performed in common inert solvents like toluene or THF.
Q: Is this process scalable for industrial manufacturing?
A: Yes, the reaction conditions are mild (0-150°C) and utilize standard organic synthesis unit operations. The absence of exotic catalysts or extreme pressures makes it highly suitable for multi-kilogram to ton-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable PPAR Alpha Modulator Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the development of next-generation metabolic therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are successfully translated into robust industrial processes. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to handle complex chiral chemistry, including the racemization strategies described in CN1835918A, positions us as a strategic partner for your long-term supply needs.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can support your project goals. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can reduce your overall production costs. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to quality and efficiency in the supply of specialized pharmaceutical intermediates.
