Advanced Synthetic Route for Spongolactone: Enabling Commercial Scale Production of Potent Anticancer Intermediates
Advanced Synthetic Route for Spongolactone: Enabling Commercial Scale Production of Potent Anticancer Intermediates
The pharmaceutical industry continuously seeks robust synthetic pathways for complex natural products that exhibit profound biological activity, particularly in the realm of oncology. Patent CN1293067C introduces a significantly more practical and efficient method for the synthesis of Spongolactone, also known as Discodermolide, and its structurally related analogs. This potent polyketide natural product, originally isolated from the marine sponge Discodermia dissoluta, has demonstrated microtubule-stabilizing properties that surpass even the well-known drug Paclitaxel in certain mechanistic assays. However, the extreme scarcity of the natural source has historically hindered its development into a viable therapeutic agent. The technical breakthrough detailed in this patent provides a viable solution by establishing a concise, three-step core sequence that transforms novel ketone intermediates into the final lactone structure with high stereochemical fidelity. For research directors and procurement specialists alike, this represents a critical shift from reliance on unsustainable natural harvesting to dependable chemical manufacturing.
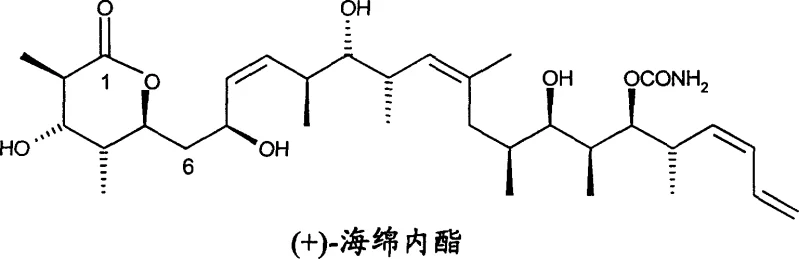
As a reliable complex polyketide intermediates supplier, understanding the nuances of such synthetic routes is paramount for ensuring supply continuity. The invention not only covers the final assembly but also claims novel intermediate compounds, such as specific ketone derivatives and beta-hydroxy ketones, which serve as essential building blocks. These intermediates allow for modular synthesis strategies, enabling manufacturers to stockpile key precursors and reduce lead times for high-purity pharmaceutical intermediates. By securing access to these patented methodologies, pharmaceutical companies can mitigate the risks associated with raw material volatility and focus their resources on clinical advancement rather than sourcing logistics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of Spongolactone-based therapeutics has been severely hampered by the lack of reliable natural sources and viable synthetic routes. Naturally occurring Spongolactones are found in minute quantities within deep-sea sponges, making large-scale harvesting ecologically damaging and logistically impractical. Furthermore, early total synthesis attempts often involved excessively long linear sequences with numerous protection and deprotection steps, leading to poor overall yields and prohibitive costs. The structural complexity of the molecule, characterized by multiple stereocenters and a delicate lactone ring, posed significant challenges for traditional organic synthesis, often resulting in mixtures of diastereomers that were difficult to separate. These inefficiencies created a bottleneck in the supply chain, preventing the compound from advancing beyond early-stage research due to the inability to produce commercially acceptable quantities.
The Novel Approach
The methodology outlined in patent CN1293067C overcomes these historical barriers by introducing a streamlined, three-step reaction sequence designed for practical scalability. Instead of constructing the entire carbon skeleton from scratch in a single campaign, this approach leverages advanced fragment coupling strategies. The core innovation lies in the use of highly stereoselective aldol reactions mediated by chiral boron reagents, which efficiently join complex fragments while establishing critical stereocenters in a single operation. This reduces the number of purification steps required and minimizes material loss. Additionally, the process utilizes robust protecting group strategies, such as acid-labile silyl ethers, which can be removed under mild conditions during the final cyclization. This novel approach effectively decouples the production of Spongolactone from natural variability, offering a consistent and reproducible manufacturing pathway that aligns with Good Manufacturing Practice (GMP) standards.
Mechanistic Insights into Stereoselective Aldol Coupling
The heart of this synthetic strategy is the stereoselective aldol coupling reaction, which joins a novel ketone compound (Formula I) with a complex aldehyde fragment (Formula II). This step is crucial because it establishes the connectivity and stereochemistry of the central portion of the molecule. The reaction is facilitated by chiral dialkylboron halides, specifically beta-chlorodiisopinocampheylborane, which act as Lewis acids to activate the ketone enolate. The chirality of the boron reagent induces a specific spatial arrangement during the transition state, ensuring that the new carbon-carbon bond forms with the desired stereochemistry. This level of control is essential for polyketides, where the biological activity is strictly dependent on the three-dimensional arrangement of hydroxyl and methyl groups. By operating at low temperatures ranging from -78°C to -20°C, the reaction kinetics are managed to favor the thermodynamic product, thereby suppressing the formation of unwanted diastereomers.
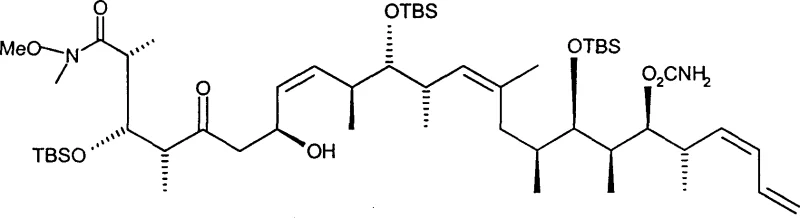
Following the coupling, the resulting beta-hydroxy ketone undergoes a chemoselective reduction to form a 1,3-diol system. This transformation is achieved using tetramethylammonium triacetoxyborohydride in the presence of acetic acid. The mechanism involves the delivery of hydride to the carbonyl carbon, guided by the neighboring hydroxyl group through a cyclic transition state, which further reinforces the stereochemical integrity of the molecule. Finally, the synthesis concludes with an acid-promoted lactonization and deprotection step. Exposure to aqueous hydrochloric acid in a methanol and tetrahydrofuran mixture simultaneously removes the acid-labile protecting groups and triggers the intramolecular esterification. This cascade reaction efficiently closes the macrocyclic lactone ring, yielding the final Spongolactone structure. This mechanistic elegance minimizes waste and simplifies the downstream processing, which is a key consideration for cost reduction in anticancer drug manufacturing.
How to Synthesize Spongolactone Efficiently
Implementing this synthesis requires precise control over reaction parameters to maintain high purity and yield. The process begins with the preparation of the novel ketone intermediates, followed by the critical coupling and finishing steps described above. Operators must adhere to strict temperature protocols, particularly during the boron-mediated aldol reaction, to ensure optimal stereoselectivity. The use of anhydrous solvents and inert atmospheres is recommended to prevent reagent degradation. While the patent provides specific examples using gram-scale quantities, the chemistry is amenable to scale-up with appropriate engineering controls. For a detailed breakdown of the specific operational parameters, reagent ratios, and workup procedures, please refer to the standardized guide below.
- Perform a stereoselective aldol coupling between a novel ketone compound (Formula I) and an aldehyde (Formula II) using a chiral boron reagent like beta-chlorodiisopinocampheylborane at low temperatures (-78°C to -20°C) to form a beta-hydroxy ketone.
- Reduce the resulting beta-hydroxy ketone (Formula III) to a 1,3-diol compound (Formula IV) using tetramethylammonium triacetoxyborohydride in acetonitrile and acetic acid at controlled temperatures (-40°C to -10°C).
- Execute acid-promoted lactonization and deprotection of the 1,3-diol compound (Formula IV) using aqueous hydrochloric acid in a methanol/THF mixture at room temperature to yield the final Spongolactone (Formula V).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers substantial strategic benefits beyond mere technical feasibility. The primary advantage lies in the stabilization of the supply chain. By shifting production from an unpredictable natural source to a controlled chemical synthesis, organizations can secure long-term availability of this critical intermediate. This reliability is essential for maintaining clinical trial timelines and preparing for potential commercial launch without the risk of raw material shortages. Furthermore, the use of common industrial solvents and reagents means that the process can be implemented in existing multipurpose manufacturing facilities without requiring specialized equipment investments.
- Cost Reduction in Manufacturing: The streamlined three-step sequence significantly reduces the operational complexity compared to traditional total synthesis methods. By minimizing the number of isolation and purification stages, the process lowers labor costs and solvent consumption. The high stereoselectivity of the aldol coupling reduces the formation of impurities, which in turn decreases the burden on quality control laboratories and improves overall throughput. Eliminating the need for expensive chromatographic separations at every step contributes to a leaner manufacturing cost structure, making the final API more economically viable.
- Enhanced Supply Chain Reliability: Dependence on marine biodiversity for drug sourcing introduces uncontrollable variables such as seasonal changes and environmental regulations. This synthetic method replaces biological uncertainty with chemical predictability. The starting materials, including the novel ketone intermediates and aldehyde fragments, can be sourced from established chemical suppliers or manufactured in parallel campaigns. This modularity allows for buffer stock creation, ensuring that production schedules are not disrupted by external factors. It effectively future-proofs the supply chain against geopolitical or ecological disruptions.
- Scalability and Environmental Compliance: The reaction conditions described are compatible with standard batch reactor operations, facilitating a smooth transition from pilot plant to commercial scale. The avoidance of heavy metal catalysts in the key coupling steps simplifies waste treatment and regulatory compliance regarding residual metals in the final drug substance. Additionally, the final deprotection step uses aqueous acid, which is easier to neutralize and dispose of compared to harsh fluorinated reagents often used in silyl deprotection. This alignment with green chemistry principles supports corporate sustainability goals and reduces environmental disposal costs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Spongolactone based on the provided patent data. These insights are derived directly from the experimental examples and summary sections of the invention disclosure. They are intended to clarify the feasibility and advantages of this specific manufacturing route for stakeholders evaluating technology transfer or procurement options.
Q: Why is a synthetic route necessary for Spongolactone instead of natural extraction?
A: Natural sources of Spongolactone, specifically the sponge Discodermia dissoluta, are extremely rare and harvesting them presents significant logistical and ecological challenges. Synthetic methods provide a reliable, scalable, and sustainable supply chain essential for clinical development and commercial manufacturing.
Q: How does this patent address stereochemical control in the synthesis?
A: The process utilizes highly stereoselective reagents, particularly chiral boron species like beta-chlorodiisopinocampheylborane during the aldol coupling step. This ensures the precise formation of multiple asymmetric carbon centers required for the biological activity of the final polyketide structure.
Q: What are the scalability advantages of this specific three-step sequence?
A: The method employs robust, commercially available reagents and standard solvent systems (ethers, acetonitrile, alcohols) without requiring exotic catalysts. The conditions are optimized for practical handling, allowing for easier purification and potential telescoping of steps, which significantly enhances manufacturability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spongolactone Supplier
The successful translation of complex laboratory chemistry into commercial reality requires a partner with deep technical expertise and robust infrastructure. NINGBO INNO PHARMCHEM stands ready to support your development programs with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art reactors capable of handling the low-temperature conditions and sensitive reagents required for this synthesis. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of Spongolactone intermediate meets the highest international standards for oncology research and drug development.
We invite you to discuss how our manufacturing capabilities can optimize your supply chain for this valuable compound. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a stable supply of high-quality polyketide intermediates for your next breakthrough therapy.
