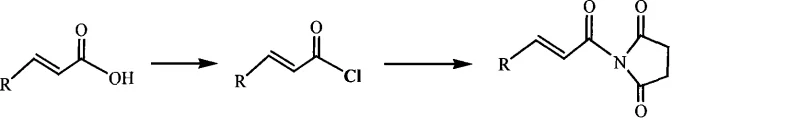
Advanced Green Synthesis of N-Substituted Acryloyl-2,5-Pyrrole-Dione Compounds for Scalable Pharmaceutical Manufacturing
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable pathways for constructing complex heterocyclic scaffolds, particularly those serving as versatile building blocks for chiral medicines and natural products. Patent CN101054360A introduces a transformative preparation method for N-substituted acryloyl-2,5-pyrrole-dione compounds, a class of molecules renowned for their utility as dienophiles in Diels-Alder reactions and receptors in Michael additions. This technology represents a significant leap forward in green chemistry by replacing hazardous traditional reagents with a sophisticated system utilizing solid phosgene and recyclable ionic liquids. For R&D directors and procurement strategists, this patent offers a compelling solution to the perennial challenges of waste management and yield optimization in fine chemical synthesis. By leveraging the unique solvation properties of ionic liquids combined with the controlled reactivity of triphosgene, this process achieves high purity and operational simplicity, positioning it as a cornerstone technology for the reliable production of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-substituted acryloyl-2,5-pyrrole-dione derivatives has relied heavily on classical acylation protocols that pose severe environmental and operational bottlenecks for large-scale manufacturing. Traditional methods typically employ aggressive chlorinating agents such as thionyl chloride (SOCl2) or phosphorus trichloride (PCl3) to activate the alpha,beta-unsaturated acid precursors. These reagents, while effective, generate substantial quantities of corrosive acidic byproducts and toxic waste streams that require complex neutralization and disposal procedures. Furthermore, the use of volatile organic solvents in these conventional routes contributes to significant atmospheric pollution and increases the overall carbon footprint of the manufacturing process. From a supply chain perspective, the handling of gaseous or highly volatile liquid chlorinating agents necessitates specialized equipment and rigorous safety protocols, driving up capital expenditure and operational costs. The resulting low atom economy and difficult purification steps often lead to inconsistent product quality and lower overall yields, creating instability in the supply of critical API intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN101054360A utilizes a greener, more efficient paradigm centered on the use of solid phosgene (triphosgene) within an ionic liquid medium. This novel approach fundamentally alters the reaction landscape by providing a controlled release of phosgene equivalents, thereby minimizing the risks associated with handling toxic gases while ensuring precise stoichiometric control. The integration of ionic liquids, such as 1-butyl-3-methylimidazolium tetrafluoroborate, serves a dual purpose: acting as a stable, non-volatile reaction medium and facilitating the easy separation of products through simple extraction techniques. This shift eliminates the need for massive volumes of traditional organic solvents and drastically reduces the generation of hazardous wastewater and solid residues. The process operates effectively across a moderate temperature range of 0 to 150°C, allowing for flexible thermal management that is conducive to energy-efficient industrial operations. By streamlining the workflow into a streamlined acylation-amidation sequence, this method not only enhances the environmental profile but also significantly improves the economic viability of producing these complex heterocyclic structures.
Mechanistic Insights into Ionic Liquid-Mediated Acylation and Amidation
The core innovation of this synthesis lies in the synergistic interaction between the organic amine catalyst, the solid phosgene, and the ionic liquid solvent system. The reaction initiates with the activation of the substituted alpha,beta-unsaturated acid by solid phosgene in the presence of an organic base such as triethylamine or pyridine. The ionic liquid environment stabilizes the transition states and intermediates, likely through hydrogen bonding and electrostatic interactions, which accelerates the formation of the reactive acyl chloride species without the degradation often seen in protic solvents. Following the acylation step, the direct addition of succinimide compounds allows for an immediate nucleophilic attack on the carbonyl carbon, forming the target N-substituted acryloyl-2,5-pyrrole-dione scaffold. The tunable nature of the ionic liquid cations and anions—ranging from imidazolium to pyridinium salts with various counterions like BF4- or PF6-—allows chemists to optimize the polarity and viscosity of the medium to suit specific substrate requirements. This level of mechanistic control ensures that side reactions, such as polymerization of the unsaturated double bond, are minimized, leading to a cleaner impurity profile.
Furthermore, the impurity control mechanism is inherently built into the phase behavior of the ionic liquid system. Upon completion of the reaction, the addition of water induces a phase separation where the organic product partitions into the extraction solvent (e.g., ethyl acetate), while the ionic liquid remains in the aqueous phase or can be recovered for reuse. This distinct phase behavior prevents the contamination of the final product with residual catalyst or solvent, which is a common issue in homogeneous catalytic systems. The ability to recycle the ionic liquid multiple times without significant loss of activity addresses one of the major cost drivers in fine chemical synthesis. For R&D teams focused on impurity profiling, this method offers a distinct advantage by reducing the formation of chlorinated byproducts and phosphorous-containing residues that are difficult to remove via standard chromatography. The result is a high-purity intermediate that meets the stringent specifications required for downstream pharmaceutical applications, reducing the burden on quality control laboratories.
How to Synthesize N-Substituted Acryloyl-2,5-Pyrrole-Dione Efficiently
The practical implementation of this synthesis route is designed for seamless translation from laboratory benchtop to pilot plant and full-scale commercial production. The protocol involves a straightforward charging sequence where the unsaturated acid, solid phosgene, and amine catalyst are introduced into the ionic liquid under controlled thermal conditions. Detailed standard operating procedures regarding specific molar ratios, temperature ramping rates, and workup parameters are critical for maximizing yield and ensuring batch-to-batch consistency. The following guide outlines the generalized steps derived from the patent embodiments to assist process chemists in replicating this high-efficiency transformation.
- Charge substituted alpha,beta-unsaturated acid, solid phosgene, and organic amine catalyst into an ionic liquid solvent at a molar ratio of approximately 1: 0.4:1.
- Heat the mixture to 90-110°C for 3-5 hours to complete the acyl chloride formation, then cool to room temperature.
- Add succinimide compounds directly to the reaction mixture, heat to 90-110°C for 1-3 hours for amidation, then perform aqueous workup and extraction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ionic liquid-based synthesis offers profound strategic advantages that extend beyond mere technical feasibility. The shift from volatile, hazardous chlorinating agents to solid phosgene significantly mitigates supply chain risks associated with the transportation and storage of dangerous goods. Moreover, the recyclability of the ionic liquid solvent represents a substantial long-term cost saving, as the expense of purchasing fresh solvent for every batch is eliminated. This process intensification leads to a drastic simplification of the downstream processing workflow, reducing the time and resources required for purification and waste treatment. By minimizing the generation of hazardous waste, manufacturers can also anticipate lower compliance costs related to environmental regulations and waste disposal fees. Ultimately, this technology enables a more resilient and cost-effective supply chain for high-purity pharmaceutical intermediates, ensuring continuity of supply even in fluctuating market conditions.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous traditional solvents, coupled with the ability to recycle the ionic liquid medium, drives down the variable cost per kilogram of the final product. The use of solid phosgene improves atom economy and reduces the need for extensive scrubbing systems required for gaseous reagents, leading to lower capital and operational expenditures. Additionally, the high yields reported in the patent embodiments suggest that raw material utilization is optimized, further enhancing the overall economic efficiency of the manufacturing process.
- Enhanced Supply Chain Reliability: Solid phosgene is a stable crystalline solid, which simplifies logistics and inventory management compared to managing pressurized cylinders of toxic gases. The robustness of the ionic liquid system allows for flexible sourcing of raw materials, as the reaction tolerates a wide range of substituted acids and succinimides. This flexibility ensures that production schedules are less vulnerable to disruptions in the supply of specific reagents, providing a more stable and predictable delivery timeline for downstream customers.
- Scalability and Environmental Compliance: The process operates under mild to moderate thermal conditions and does not require extreme pressures, making it highly amenable to scale-up in standard stainless steel reactors. The significant reduction in VOC emissions and aqueous waste aligns with increasingly stringent global environmental standards, future-proofing the manufacturing site against regulatory changes. This green chemistry approach not only enhances the corporate sustainability profile but also facilitates smoother permitting processes for capacity expansion.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ionic liquid-mediated synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the operational nuances and strategic benefits of this method.
Q: What are the advantages of using ionic liquids over traditional solvents in this synthesis?
A: Ionic liquids serve as both solvent and catalyst support, allowing for recyclability and significantly reducing volatile organic compound (VOC) emissions compared to traditional chlorinated solvents.
Q: Is solid phosgene safer to handle than thionyl chloride for industrial scale-up?
A: Yes, solid phosgene (triphosgene) is a crystalline solid at room temperature, making it much easier and safer to weigh and transport than gaseous or liquid chlorinating agents like thionyl chloride.
Q: Can this method accommodate diverse R-groups on the unsaturated acid?
A: The patent demonstrates successful synthesis with various R-groups including alkyl chains (C1-C20) and aromatic radicals (C6-C20), indicating broad substrate tolerance for complex intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Substituted Acryloyl-2,5-Pyrrole-Dione Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical market. Our team of expert process chemists has extensively evaluated the technology described in CN101054360A and possesses the technical capability to implement this green synthesis route at commercial scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of high-quality intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of N-substituted acryloyl-2,5-pyrrole-dione meets the highest industry standards for chiral medicine and agrochemical synthesis.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project requirements. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this ionic liquid-based process for your specific volume needs. We encourage you to request specific COA data and route feasibility assessments to validate the performance of our materials in your downstream applications. Let us be your trusted partner in transforming complex chemical challenges into commercially viable realities.
