Advanced Manufacturing of Clopidogrel Active Metabolite Derivatives for Global Pharma Supply Chains
Advanced Manufacturing of Clopidogrel Active Metabolite Derivatives for Global Pharma Supply Chains
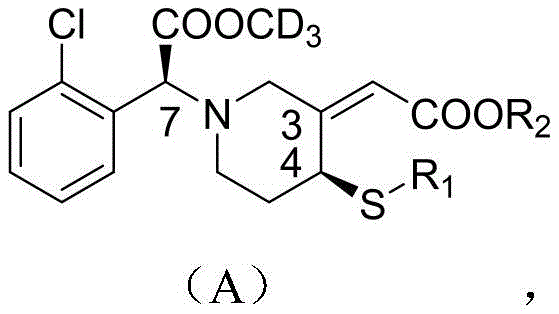
The landscape of cardiovascular therapeutics is constantly evolving, driven by the need to overcome limitations in existing anti-platelet therapies. Patent CN111848497A introduces a groundbreaking approach to synthesizing clopidogrel active metabolite derivatives and their prodrugs, addressing critical issues such as low oral bioavailability and delayed onset of action. This technology focuses on the precise construction of molecules with specific stereochemistry, particularly the Z-configuration at the C3-C16 double bond and the S-configuration at the C4 position, which are paramount for pharmacological efficacy. By incorporating deuterated methyl esters, these derivatives effectively bypass the metabolic bottleneck caused by esterase CES1 hydrolysis, a pathway that renders 85% of standard clopidogrel inactive. For R&D directors and supply chain leaders, this represents a significant opportunity to access high-purity pharmaceutical intermediates that promise superior clinical profiles and more reliable manufacturing processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of clopidogrel active metabolites has been plagued by significant technical hurdles that impact both cost and quality. The most prevalent method in the prior art relies on biological enzyme catalysis. While enzymatic routes can offer high efficiency in specific contexts, they are inherently fragile and expensive. The cost of biocatalysts is substantial, and the reaction conditions are often严苛 (harsh), requiring precise control that is difficult to maintain on a large industrial scale. Furthermore, enzymatic processes are frequently limited by substrate availability, making it challenging to secure sufficient quantities of target metabolites for commercial demand. Beyond biology, traditional chemical synthesis methods often lack the necessary stereoselectivity. They frequently fail to distinguish between the C3-C16 double bond configurations, producing a mixture of Z (active) and E (inactive) isomers. This lack of selectivity leads to low yields of the desired active ingredient and generates a complex impurity profile that requires costly and wasteful purification steps, ultimately driving up the cost of goods sold.
The Novel Approach
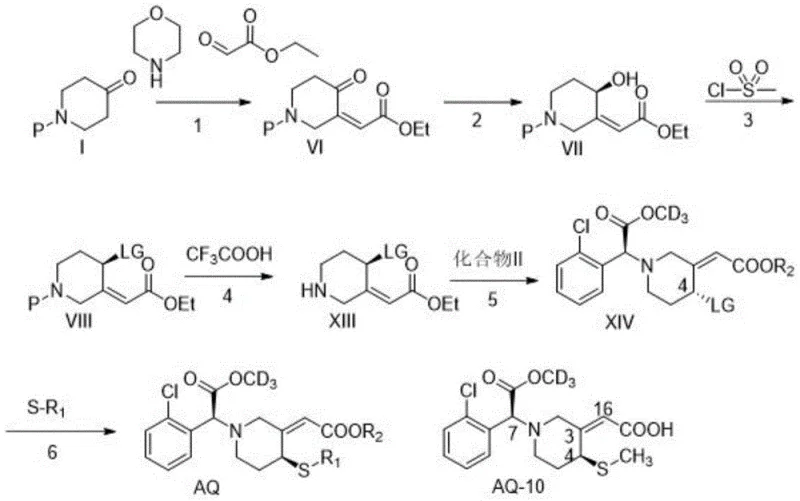
The methodology disclosed in CN111848497A offers a robust chemical alternative that overcomes these stereochemical and economic barriers. The core innovation lies in a multi-step synthetic route that prioritizes stereoselectivity through small-molecule catalysis rather than fragile enzymes. As illustrated in the comprehensive reaction scheme below, the process begins with a condensation reaction that utilizes morpholine or piperidine as a catalyst. This specific choice of catalyst facilitates the formation of an enamine intermediate, which sterically directs the reaction to favor the Z-configuration at the C3-C16 double bond with an exceptional enantiomeric excess (ee) value exceeding 95%. This chemical precision eliminates the formation of inactive E-isomers at the source. Additionally, the route incorporates a versatile SN2 substitution strategy in the later stages, allowing for the flexible introduction of various substituents at the C4 position. This modularity not only enables the synthesis of the primary metabolite AQ-10 but also allows for the rapid generation of a library of prodrugs with optimized stability and toxicity profiles.
Mechanistic Insights into Stereoselective Enamine Catalysis and SN2 Substitution
The success of this synthesis hinges on two critical mechanistic features: the initial stereocontrol during alkene formation and the final dual-function nucleophilic substitution. In the first step, the reaction between the protected piperidone and ethyl glyoxylate is mediated by secondary amines like morpholine. Mechanistically, the amine forms a transient enamine with the ketone, which alters the electronic and steric environment of the alpha-carbon. This enamine intermediate attacks the aldehyde in a specific orientation that minimizes steric clash, thermodynamically and kinetically favoring the formation of the Z-alkene over the E-alkene. This is a crucial distinction because, as metabolic studies confirm, the E-configuration renders the metabolite completely inactive. By locking in the Z-geometry early in the synthesis, the process ensures that downstream steps are working with the correct scaffold, maximizing overall yield and minimizing waste. The subsequent chiral reduction using lithium tri-tert-butoxy aluminum hydride further refines the molecular architecture by establishing the S-configuration at the C4 hydroxyl group, setting the stage for the final functionalization.
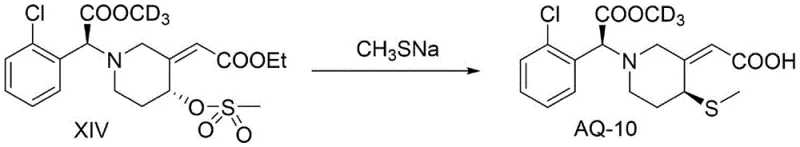
The final transformation involves a cleverly designed SN2 reaction that serves a dual purpose. The intermediate compound XIV, containing a good leaving group (LG) such as a nitrobenzenesulfonyl group at the C4 position, reacts with a sulfur nucleophile like sodium methanethiolate (NaSCH3). This step accomplishes two goals simultaneously: it installs the critical methylthio group required for P2Y12 receptor binding, and due to the strong basicity of the thiolate salt, it concurrently hydrolyzes the ethyl ester at the side chain to the free carboxylic acid. This tandem reaction simplifies the workflow by combining substitution and deprotection into a single operational unit. The result is the direct formation of the target acid AQ-10 without the need for separate hydrolysis steps, streamlining the process and reducing solvent usage. This mechanistic efficiency is a key driver for the process's scalability and cost-effectiveness.
How to Synthesize Clopidogrel Active Metabolite Derivative Efficiently
Implementing this synthesis requires strict adherence to the stereoselective protocols outlined in the patent to ensure the high purity required for pharmaceutical applications. The process is designed to be linear and convergent, allowing for the preparation of key intermediates like the deuterated mandelic acid derivative and the functionalized piperidine core in parallel before their final coupling. Operators must pay close attention to the catalyst loading in the first step and the temperature control during the chiral reduction to maintain the high ee values reported. The following guide outlines the standardized operational framework derived from the patent examples, providing a clear roadmap for laboratory and pilot-scale execution.
- Condensation of Boc-protected piperidone with ethyl glyoxylate using morpholine/piperidine catalyst to form the Z-configuration alkene intermediate.
- Chiral reduction of the ketone group using lithium tri-tert-butoxy aluminum hydride to establish the C4 stereocenter.
- Activation of the hydroxyl group via sulfonylation followed by deprotection to generate the reactive amine intermediate.
- SN2 coupling with deuterated mandelic acid derivative to install the side chain with high stereochemical fidelity.
- Final nucleophilic substitution with sodium methanethiolate to introduce the methylthio group and simultaneously hydrolyze the ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from enzymatic or non-selective chemical methods to this patented stereoselective route offers tangible strategic benefits. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By eliminating the dependence on specialized, high-cost enzymes, manufacturers can source standard chemical reagents that are readily available in the global market, reducing the risk of supply disruptions. Furthermore, the robustness of the chemical catalysts means that reaction conditions are less sensitive to minor fluctuations, leading to more consistent batch-to-batch quality and reducing the rate of failed batches that can derail production schedules.
- Cost Reduction in Manufacturing: The elimination of expensive biocatalysts directly lowers the raw material cost base. Additionally, the high stereoselectivity (>95% ee) means that less material is wasted on inactive isomers that must be discarded or recycled. The tandem nature of the final step, which combines substitution and hydrolysis, reduces the number of unit operations, saving on labor, energy, and solvent consumption. These efficiencies compound to deliver a significantly lower cost of goods without compromising on the quality of the high-purity pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: Chemical synthesis routes are generally more scalable and predictable than biological ones. The ability to synthesize the core scaffold using common organic building blocks ensures that production is not bottlenecked by the lead times associated with fermenting or isolating enzymes. This reliability is crucial for maintaining continuous supply to downstream API manufacturers, especially in the event of sudden demand surges for cardiovascular medications. The process design supports a steady flow of materials, mitigating the risks of stockouts.
- Scalability and Environmental Compliance: The process utilizes standard organic solvents and reagents that are well-understood in terms of waste management and environmental impact. By avoiding complex biological waste streams and reducing the total number of reaction steps through tandem transformations, the overall environmental footprint of the manufacturing process is reduced. This aligns with modern green chemistry principles and simplifies regulatory compliance regarding waste disposal, making it easier to scale production from pilot plants to multi-ton commercial facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these clopidogrel metabolite derivatives. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy for our technical partners.
Q: Why is the Z-configuration at the C3-C16 double bond critical for clopidogrel metabolites?
A: Metabolic studies indicate that the pharmacological activity of clopidogrel depends heavily on stereochemistry. Specifically, the Z-configuration at the C3-C16 double bond is essential for anti-platelet aggregation activity, whereas the E-configuration results in inactive metabolites. The patented process ensures >95% ee for the Z-isomer.
Q: How does the deuterated methyl ester improve drug performance compared to standard clopidogrel?
A: The deuterated methyl ester at the C7 position effectively avoids hydrolysis by esterase CES1 in the human body. This significantly reduces the formation of inactive carboxylic acid derivatives (which account for 85% of metabolism in standard clopidogrel), thereby enhancing bioavailability and onset speed.
Q: What advantages does this chemical synthesis route offer over enzymatic methods?
A: Unlike enzymatic methods which suffer from high costs, harsh reaction conditions, and substrate limitations, this chemical route utilizes robust small-molecule catalysts like morpholine. It offers higher universality, easier scale-up potential, and eliminates the dependency on expensive biological reagents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Clopidogrel Metabolite Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation cardiovascular therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated stereoselective chemistry described in CN111848497A can be translated from the lab bench to industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of clopidogrel metabolite derivative meets the exacting standards required for global regulatory submissions. Our commitment to technical excellence ensures that your supply chain remains uninterrupted and compliant.
We invite you to collaborate with us to optimize your sourcing strategy for these advanced intermediates. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your R&D and commercial goals efficiently and reliably.
