Advanced Titanium-Catalyzed Synthesis of 1,4-Tetraalkoxy-2-butene for Industrial Carotenoid Production
The landscape of carotenoid synthesis is undergoing a significant transformation driven by the need for more efficient and cost-effective intermediate production methods. Patent CN114315532B, published in May 2023, introduces a groundbreaking methodology for the preparation of 1,4-tetraalkoxy-2-butene compounds, which serve as critical building blocks in the manufacture of high-value carotenoids such as beta-carotene, astaxanthin, and lycopene. This innovation addresses long-standing inefficiencies in the supply chain by replacing expensive furan-based precursors with economically viable 2,2-dialkoxyacetaldehydes. The technical breakthrough utilizes a high-valence titanium-mediated reductive coupling mechanism that operates under relatively mild conditions compared to traditional strong acid catalysis. For R&D directors and procurement specialists, this represents a pivotal shift towards more sustainable and scalable chemical manufacturing processes. The ability to achieve single-pass separation yields approaching 97% fundamentally alters the economic model for producing these essential fine chemical intermediates. 
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 1,4-tetraalkoxy-2-butene has relied heavily on the acid-catalyzed reaction of 2,5-dimethoxy-2,5-dihydrofuran with alcohol compounds. This conventional pathway is fraught with significant technical and economic drawbacks that hinder optimal production efficiency. Firstly, the starting material, 2,5-dimethoxy-2,5-dihydrofuran, commands a prohibitively high market price, often ranging between 140,000 to 160,000 RMB per ton, which drastically inflates the overall cost of goods sold. Secondly, the reliance on strong inorganic or organic acids as catalysts introduces severe corrosion risks to reactor vessels and piping, necessitating expensive specialized materials and frequent maintenance schedules. Furthermore, these acid-catalyzed reactions typically suffer from poor selectivity and moderate yields, often hovering around 72% to 78%, which results in substantial material loss and increased waste treatment burdens. The use of dehydrating agents like trimethyl orthoformate further complicates the reaction mixture, making downstream purification energy-intensive and chemically complex.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages a titanium-mediated reductive coupling of 2,2-dialkoxyacetaldehyde, offering a robust alternative that circumvents the pitfalls of the traditional furan route. By shifting the raw material base to 2,2-dialkoxyacetaldehyde, manufacturers can access a significantly more economical feedstock that is widely available in the fine chemical market. The reaction mechanism employs high-valence titanium species (Ti3+ or Ti4+) in conjunction with common reducing agents like zinc or magnesium, eliminating the need for corrosive strong acids entirely. This change not only preserves equipment integrity but also simplifies the workup procedure, as the reaction can be quenched with mild aqueous alkaline solutions. The result is a process capable of delivering exceptional yields, with experimental data demonstrating single-pass isolation efficiencies reaching up to 97%. This dramatic improvement in yield and selectivity translates directly into reduced raw material consumption and lower environmental impact, making it an ideal candidate for green chemistry initiatives in the pharmaceutical and nutraceutical sectors. 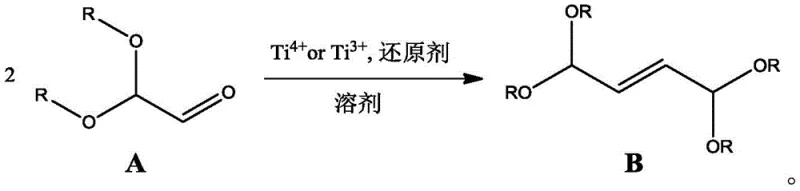
Mechanistic Insights into Titanium-Mediated Reductive Coupling
The core of this innovative synthesis lies in the sophisticated interaction between the high-valence titanium auxiliary agent and the reducing agent within the organic solvent matrix. When a reducing agent such as zinc powder is dissolved in tetrahydrofuran and cooled to temperatures between -40°C and 10°C, it creates a highly reactive environment for the subsequent addition of the titanium species. The titanium compound, whether it be TiCl3, TiBr4, or TiI4, acts as a Lewis acid mediator that facilitates the formation of low-valent titanium species in situ. These low-valent titanium centers are renowned for their ability to mediate carbon-carbon bond formation through single-electron transfer processes, effectively coupling two molecules of the aldehyde substrate. The precise control of the molar ratio between the titanium auxiliary and the aldehyde substrate, optimized between 1.5:1 and 2.5:1, is critical for maximizing the conversion rate while minimizing the formation of oligomeric byproducts. This mechanistic pathway ensures that the reaction proceeds with high stereocontrol, favoring the formation of the desired trans-alkene geometry which is crucial for downstream carotenoid assembly.
Impurity control in this system is inherently superior due to the specificity of the titanium-mediated coupling and the absence of aggressive acid catalysis. In traditional acid-catalyzed routes, side reactions such as polymerization of the furan ring or ether cleavage often lead to complex impurity profiles that are difficult to separate. However, in this reductive coupling system, the primary byproducts are typically inorganic salts derived from the titanium and reducing agent, which are easily removed during the aqueous alkaline quenching step. The use of solvents like toluene or diethyl ether further aids in phase separation, allowing for a clean isolation of the organic product. Additionally, the reaction temperature profile, which involves an initial low-temperature activation followed by a reflux period of 10 to 15 hours, ensures complete consumption of the starting material without thermal degradation of the sensitive acetal functionality. This robustness in impurity management is particularly valuable for R&D teams aiming to meet stringent purity specifications required for food-grade or pharmaceutical-grade carotenoid intermediates.
How to Synthesize 1,4-Tetraalkoxy-2-butene Efficiently
The practical implementation of this synthesis route requires careful attention to the order of addition and temperature control to ensure safety and reproducibility. The process begins with the preparation of the reducing environment under an inert nitrogen atmosphere to prevent oxidation of the sensitive titanium species. Operators must dissolve the chosen reducing agent in the organic solvent and stabilize the temperature before introducing the titanium catalyst, as premature mixing can lead to exothermic events. Once the catalytic system is activated, the 2,2-dialkoxyacetaldehyde is introduced, and the mixture is allowed to warm to reflux to drive the coupling reaction to completion. Following the reaction period, the addition of an aqueous alkaline solution serves a dual purpose: it quenches any remaining reactive titanium species and neutralizes acidic byproducts, facilitating a straightforward phase separation.
- Under nitrogen protection, dissolve a reducing agent (such as Zn, Mg, or LiAlH4) in an organic solvent like tetrahydrofuran and cool the mixture to between -40°C and 10°C.
- Add a high-valence titanium auxiliary agent (Ti3+ or Ti4+ compound such as TiCl3 or TiBr4) to the reaction vessel under stirring conditions.
- Introduce the 2,2-dialkoxyacetaldehyde substrate, raise the temperature to reflux for 10-15 hours, and finally quench with an aqueous alkaline solution to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this titanium-catalyzed process offers compelling strategic advantages that extend beyond simple yield improvements. The most significant impact is observed in the raw material cost structure, as the substitution of expensive dihydrofuran derivatives with commodity aldehydes drastically reduces the direct material costs associated with production. This shift allows companies to hedge against volatility in the specialty furan market and leverage the stable supply chains associated with bulk aldehyde manufacturing. Furthermore, the elimination of strong mineral acids from the process workflow removes a major bottleneck related to equipment maintenance and regulatory compliance. Facilities no longer need to invest heavily in Hastelloy reactors or specialized acid-resistant lining, nor do they face the same level of scrutiny regarding hazardous acid waste disposal. This simplification of the chemical bill of materials and waste stream directly contributes to a leaner, more agile manufacturing operation.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substantial difference in raw material pricing between the traditional furan precursors and the new aldehyde substrates. By utilizing 2,2-dialkoxyacetaldehyde, which is produced on a much larger industrial scale, manufacturers can achieve significant savings on input costs without compromising on product quality. Additionally, the high reaction yield minimizes the loss of valuable carbon atoms, ensuring that a greater proportion of the purchased raw material ends up in the final saleable product. The reduction in downstream processing requirements, such as extensive neutralization and purification steps, further lowers utility consumption and labor costs, creating a compounding effect on overall profitability.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like zinc, magnesium, and simple aldehydes enhances supply chain resilience compared to sourcing niche heterocyclic compounds. These standard reagents are produced by multiple global suppliers, reducing the risk of single-source dependency and supply disruptions. The robustness of the reaction conditions also means that production schedules are less likely to be impacted by equipment failures related to corrosion or fouling. This reliability is crucial for maintaining consistent delivery timelines to downstream customers in the nutraceutical and pharmaceutical industries, where batch consistency and on-time delivery are paramount contractual obligations.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, as the reagents and solvents used are compatible with standard large-scale batch reactors. The absence of corrosive acids simplifies the engineering requirements for scaling up from pilot plant to commercial production volumes. From an environmental perspective, the generation of inorganic salt waste is easier to manage and treat compared to complex organic acid waste streams. This alignment with green chemistry principles not only reduces environmental fees but also enhances the corporate sustainability profile, which is increasingly important for securing contracts with environmentally conscious multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These answers are derived directly from the experimental data and technical disclosures found in the patent literature to provide accurate guidance for potential adopters. Understanding these nuances is essential for evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: What are the primary advantages of this titanium-catalyzed method over traditional acid-catalyzed routes?
A: The primary advantage lies in the substitution of expensive 2,5-dimethoxy-2,5-dihydrofuran with economically superior 2,2-dialkoxyacetaldehyde. Furthermore, this method eliminates the need for strong inorganic or organic acids, thereby reducing equipment corrosion and simplifying post-reaction neutralization processes while achieving yields exceeding 90%.
Q: Which reducing agents are compatible with this high-valence titanium system?
A: The patent specifies a broad range of compatible reducing agents including metallic powders like Zinc (Zn) and Magnesium (Mg), as well as hydride sources such as Lithium Aluminum Hydride (LiAlH4) and Sodium Borohydride (NaBH4), allowing flexibility based on cost and safety requirements.
Q: How does this process impact the scalability of carotenoid intermediate production?
A: By utilizing readily available aldehyde precursors and avoiding corrosive strong acids, the process significantly enhances operational safety and equipment longevity. The high single-pass separation yields (up to 97%) minimize waste generation and downstream purification burdens, facilitating smoother commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4-Tetraalkoxy-2-butene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this titanium-catalyzed synthesis route for the global carotenoid market. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of high-purity intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the exacting standards required for food and pharmaceutical applications. We are committed to leveraging advanced process chemistry to deliver value-driven solutions that optimize both performance and cost for our international partners.
We invite procurement leaders and technical directors to engage with our team to explore how this innovative manufacturing method can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic impact of switching to this aldehyde-based route. We encourage you to contact our technical procurement team today to obtain specific COA data and comprehensive route feasibility assessments tailored to your production volume needs.
