Advanced Synthetic Route for High-Purity Nicorandil Trimer Impurity Standards
The pharmaceutical industry faces continuous challenges in maintaining the rigorous quality standards required for cardiovascular medications, particularly for unstable compounds like nicorandil. Patent CN114621138A introduces a groundbreaking methodology for the synthesis of nicorandil trimer, a critical degradation impurity that must be strictly monitored to ensure drug safety and efficacy. This innovation addresses the longstanding difficulty in obtaining high-purity reference standards through traditional degradation pathways, offering a rational, stepwise synthetic route that significantly enhances process control. By shifting from stochastic thermal degradation to a directed chemical synthesis, manufacturers can now secure a consistent supply of this complex pyridinium salt derivative. This technical advancement is pivotal for R&D teams focused on impurity profiling and regulatory compliance, ensuring that every batch of nicorandil API meets the stringent specifications demanded by global health authorities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of nicorandil oligomers, including dimers and trimers, relied heavily on the forced thermal degradation of the parent nicorandil molecule, a stochastic process that inherently lacks selectivity and efficiency. As documented in prior art such as CN 109516950 A and CN 109516951 A, these conventional methods involve dissolving nicorandil in various organic solvents and subjecting the solution to prolonged heating or standing over extended periods to induce breakdown. This approach suffers from severe drawbacks, primarily the formation of complex mixtures containing both dimeric and trimeric species alongside the unreacted starting material, making isolation extremely difficult. The low conversion rates necessitate the use of costly and labor-intensive preparative chromatography to separate the target trimer from the dimer, drastically inflating production costs and limiting scalability. Furthermore, the reliance on degradation means the yield is intrinsically capped by the stability limits of the nitrate ester functionality, leading to significant material waste and inconsistent batch-to-batch quality.
The Novel Approach
In stark contrast, the novel approach delineated in patent CN114621138A introduces a rational, stepwise synthetic strategy that bypasses the thermodynamic pitfalls of degradation entirely. This method utilizes N-(2-hydroxyethyl)-nicotinamide as a robust starting material, which is first converted into a reactive N-(2-haloethyl)nicotinamide intermediate via a controlled halogenation reaction. This activated intermediate then undergoes a precise quaternization reaction with nicorandil itself, driving the formation of the trimer structure with high specificity. By decoupling the formation of the ethyl-pyridinium linkage from the instability of the nitrate group, the process achieves superior conversion rates and purity profiles without generating significant dimer byproducts. This direct synthesis pathway eliminates the need for complex chromatographic purification, allowing for simple recrystallization techniques to achieve pharmaceutical-grade purity, thereby streamlining the entire manufacturing workflow.
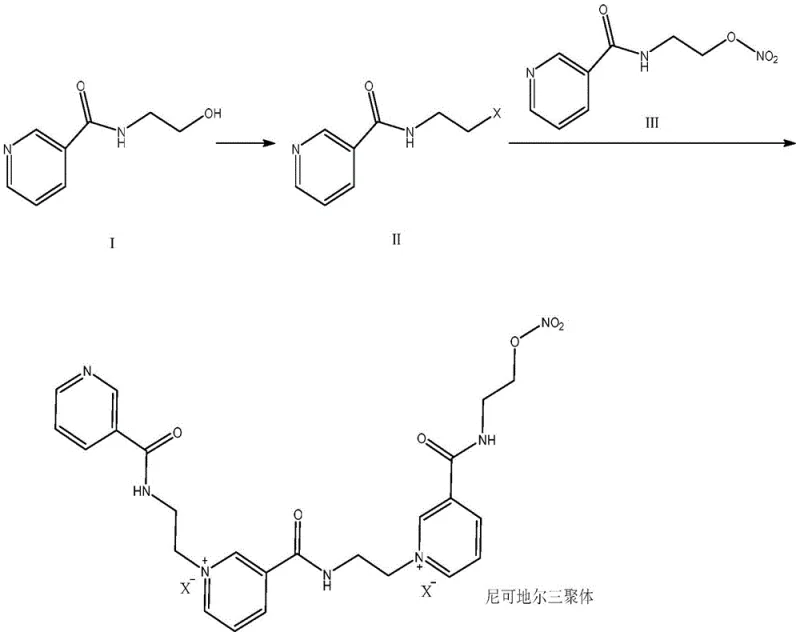
This strategic shift from degradation to construction represents a paradigm change in how critical impurity standards are manufactured, offering a reliable pathway for producing large quantities of material for quality control applications.
Mechanistic Insights into Halogenation and Quaternization
The core of this synthetic breakthrough lies in the efficient activation of the hydroxyethyl side chain through a modified Appel-type halogenation mechanism. In the first stage, N-(2-hydroxyethyl)-nicotinamide reacts with a carbon tetrahalide, preferably carbon tetrachloride, in the presence of triphenylphosphine within an organic solvent system such as tetrahydrofuran or acetonitrile. This reaction generates a highly reactive N-(2-haloethyl)nicotinamide intermediate, where the hydroxyl group is replaced by a halogen atom, creating a potent electrophile capable of alkylating the pyridine nitrogen of nicorandil. The reaction conditions are carefully optimized, typically ranging from -20°C to 80°C, to maximize the formation of the halo-intermediate while minimizing side reactions that could compromise the integrity of the nicotinamide moiety. Following the reaction, the mixture is quenched in ice water, and the intermediate is isolated through extraction and drying, ensuring that the reactive species is stable enough for the subsequent coupling step.
The second mechanistic phase involves the nucleophilic attack of the pyridine nitrogen of nicorandil on the activated ethyl chain of the intermediate. When nicorandil is dissolved in a polar aprotic solvent like DMF and heated, the addition of the halo-intermediate facilitates a quaternization reaction that links the two molecular units via an ethylene bridge. This forms the characteristic bis-pyridinium structure of the trimer, stabilized by chloride counterions derived from the halogenation step. The structural integrity of the final product is rigorously confirmed through advanced spectroscopic techniques, including high-resolution mass spectrometry and comprehensive NMR analysis. As illustrated in the structural assignment below, the distinct chemical shifts of the pyridinium protons and the methylene bridges provide unequivocal evidence of the trimeric architecture, distinguishing it clearly from the dimeric analogues.
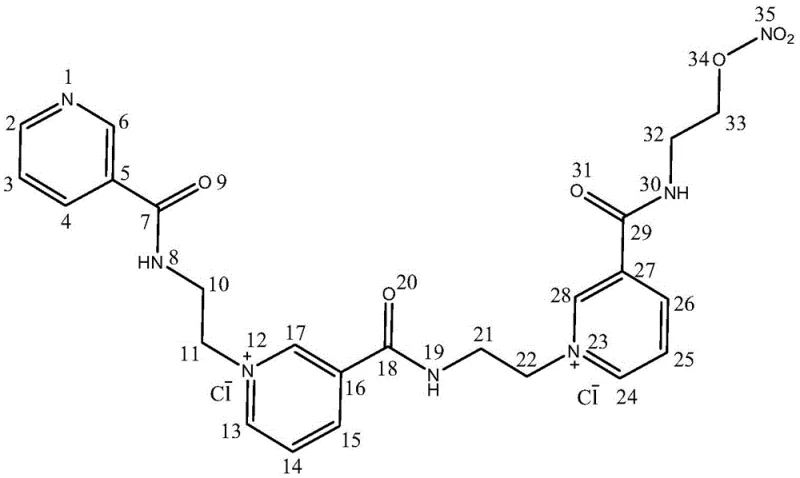
The precise control over these reaction parameters ensures that the final product exhibits the exact physicochemical properties required for use as a certified reference material in HPLC assays, with a retention time and spectral profile that matches the theoretical degradation product perfectly.
How to Synthesize Nicorandil Trimer Efficiently
The implementation of this synthesis protocol requires careful attention to stoichiometry and temperature control to ensure optimal yields and purity. The process begins with the preparation of the halo-intermediate under inert atmosphere to prevent moisture interference, followed by a controlled coupling reaction where the molar ratio of intermediate to nicorandil is maintained between 2:1 and 4:1 to drive the equilibrium towards the trimer. Detailed operational parameters, including specific solvent volumes, reaction times, and crystallization temperatures, are critical for reproducibility. For a comprehensive guide on executing this synthesis with maximum efficiency and safety, please refer to the standardized procedure outlined below.
- Dissolve N-(2-hydroxyethyl)-nicotinamide in an organic solvent and react with carbon tetrahalide and triphenylphosphine to form the halo-intermediate.
- Isolate the N-(2-haloethyl)nicotinamide intermediate via extraction and purification after quenching in ice water.
- React nicorandil with the isolated intermediate in a heated organic solvent, followed by recrystallization in water-alcohol mixtures to obtain the pure trimer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers substantial strategic benefits that extend far beyond simple cost metrics. By eliminating the reliance on unpredictable thermal degradation, manufacturers can stabilize their production schedules and reduce the risk of supply disruptions caused by low-yield batches. The ability to produce the trimer directly from stable, commercially available starting materials means that raw material sourcing becomes more predictable and less susceptible to market volatility associated with specialized degradation precursors. Furthermore, the removal of complex chromatographic purification steps significantly reduces the consumption of expensive solvents and stationary phases, leading to a leaner and more environmentally sustainable manufacturing process. This efficiency gain translates into a more competitive pricing structure for high-purity reference standards, allowing pharmaceutical companies to allocate resources more effectively across their quality control budgets.
- Cost Reduction in Manufacturing: The elimination of preparative HPLC purification represents a massive reduction in operational expenditure, as chromatography is notoriously resource-intensive and difficult to scale. By relying on crystallization for purification, the process utilizes standard chemical engineering unit operations that are far cheaper to run and maintain. The higher conversion rates also mean that less starting material is wasted, improving the overall atom economy of the synthesis. Additionally, the use of common organic solvents like DMF and alcohols simplifies solvent recovery and recycling protocols, further driving down the variable costs associated with large-scale production runs.
- Enhanced Supply Chain Reliability: Sourcing high-purity impurity standards has historically been a bottleneck for pharmaceutical quality assurance, often leading to delays in batch release testing. This robust synthetic method ensures a consistent and reliable supply of nicorandil trimer, mitigating the risk of stockouts that could halt production lines. The scalability of the process means that suppliers can rapidly ramp up production to meet surges in demand without compromising on quality or lead times. This reliability is crucial for maintaining compliance with regulatory timelines and ensuring that finished drug products reach patients without unnecessary delays caused by pending impurity analysis.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions and workup procedures that are easily transferable from laboratory to pilot and commercial scales. The avoidance of hazardous degradation byproducts simplifies waste management and reduces the environmental footprint of the manufacturing facility. Recrystallization from water-alcohol mixtures is a green chemistry approach that minimizes the use of chlorinated solvents in the final purification steps. This alignment with environmental, social, and governance (ESG) goals makes the supply chain more resilient to future regulatory changes regarding chemical manufacturing and waste disposal.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is essential for stakeholders involved in the procurement and quality assessment of pharmaceutical intermediates. The following questions address common inquiries regarding the application, stability, and regulatory utility of the nicorandil trimer produced via this method. These insights are derived directly from the experimental data and structural characterization provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: Why is the new synthesis method superior to thermal degradation?
A: The new method avoids the non-selective thermal degradation of nicorandil, which produces complex mixtures of dimers and trimers that are difficult to separate. The stepwise approach ensures high conversion directly to the trimer.
Q: What is the primary advantage for quality control laboratories?
A: This method provides a reliable source of high-purity nicorandil trimer reference standards, which are essential for accurate HPLC quantification of impurities in nicorandil API batches.
Q: Is this process scalable for commercial production?
A: Yes, the process utilizes standard unit operations such as solvent extraction and recrystallization, avoiding complex chromatographic separations, making it highly suitable for kilogram-to-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nicorandil Trimer Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality impurity standards play in the development and manufacturing of safe cardiovascular therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistent quality. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every gram of nicorandil trimer we supply meets the highest analytical standards required for HPLC calibration and method validation. Our commitment to technical excellence allows us to support your R&D and QA teams with materials that facilitate accurate and compliant drug testing.
We invite you to contact our technical procurement team to discuss how our advanced synthesis capabilities can optimize your supply chain for nicorandil impurity standards. Request a Customized Cost-Saving Analysis today to understand the economic benefits of switching to our streamlined production method. We are ready to provide specific COA data and route feasibility assessments tailored to your project needs, ensuring a seamless integration of our materials into your quality control workflows.
