Advanced Manufacturing of N-Heterocyclic Carbene Silver Complexes for Industrial Catalysis
The landscape of organometallic catalysis is continually evolving, driven by the demand for more robust and cost-effective synthetic methodologies for high-value intermediates. A pivotal advancement in this domain is detailed in patent CN102276634A, which discloses a highly efficient method for preparing N-heterocyclic carbene (NHC) silver complexes. These complexes serve as crucial precursors for various transition metal catalysts used in pharmaceutical and fine chemical synthesis, particularly for allylic alkylation and amination reactions. The disclosed technology addresses critical bottlenecks associated with traditional synthesis routes, offering a pathway that is not only chemically superior but also commercially viable for large-scale manufacturing. By shifting away from sensitive reagents to stable silver salts, this innovation promises to enhance the reliability of the supply chain for these essential catalytic species.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of N-heterocyclic carbene silver complexes has relied heavily on the reaction between imidazolium salts and silver oxide (Ag2O). While chemically effective, this conventional approach suffers from significant practical drawbacks that hinder its industrial application. The primary limitation is the extreme photosensitivity of silver oxide, which necessitates rigorous light-proof operating conditions throughout the synthesis and purification stages. This requirement imposes substantial capital expenditure on specialized reactor shielding and increases operational complexity, as any exposure to ambient light can degrade the reagent and compromise the reaction yield. Furthermore, silver oxide is generally more expensive and less atom-economical compared to simple silver halides, driving up the raw material costs for the final catalyst precursor.
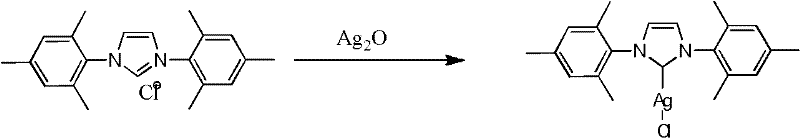
The Novel Approach
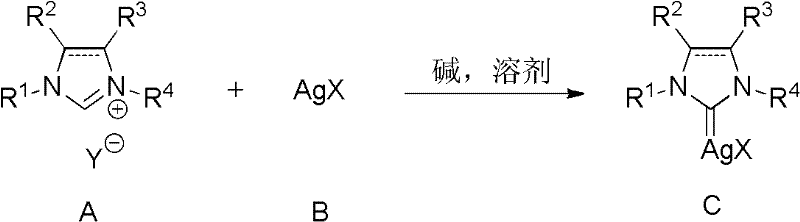
In stark contrast to the legacy methods, the novel approach described in the patent utilizes silver halides, such as silver chloride, as the silver source in the presence of a base and solvent. This strategic substitution fundamentally alters the operational profile of the synthesis, rendering the process insensitive to light and significantly more robust against environmental variables. The reaction proceeds efficiently in a one-step protocol where the imidazolium salt and silver salt are heated together, eliminating the need for multi-step activation or specialized handling equipment. This simplicity translates directly into process intensification, allowing for higher throughput and reduced downtime between batches. The ability to operate without stringent light controls represents a major leap forward in process safety and ease of execution for chemical manufacturers.
Mechanistic Insights into Silver Salt Mediated Transmetallation
The core of this innovative synthesis lies in the in situ generation of the silver-carbene bond through a base-mediated deprotonation and coordination mechanism. In this system, the inorganic base, such as potassium carbonate or cesium carbonate, acts to deprotonate the acidic C2 proton of the imidazolium ring. This deprotonation generates the free carbene species transiently, which immediately coordinates with the silver cation released from the silver halide lattice. The choice of solvent plays a critical role in facilitating this ionization and coordination; polar aprotic solvents or coordinating solvents like 3-chloropyridine stabilize the transition state and ensure high conversion rates. The reaction conditions, typically ranging from 50°C to 150°C, provide sufficient thermal energy to overcome the lattice energy of the silver salt while maintaining the stability of the resulting organometallic complex.
From an impurity control perspective, this mechanism offers distinct advantages over oxidative methods. Since the reaction does not involve redox-active silver oxide, there is a reduced risk of forming oxidized byproducts or metallic silver precipitates that can contaminate the final product. The purification strategy outlined involves a straightforward dissolution in dichloromethane followed by rapid filtration through a short silica plug. This step effectively removes inorganic salts and unreacted starting materials, which are insoluble in the organic phase. Subsequent precipitation in non-polar alkanes like pentane ensures the isolation of the complex as a high-purity solid. This streamlined workup minimizes solvent usage and waste generation, aligning with green chemistry principles while ensuring the stringent purity specifications required for catalytic applications.
How to Synthesize N-Heterocyclic Carbene Silver Complex Efficiently
The synthesis protocol defined in the patent provides a clear roadmap for producing high-quality NHC silver complexes with minimal operational friction. The process begins with the precise weighing of the imidazolium salt and the silver halide, typically in a molar ratio ranging from 1:1 to 1:5, depending on the specific steric and electronic properties of the ligand. These solids are suspended in a selected solvent, such as 3-chloropyridine or acetonitrile, along with a stoichiometric excess of an inorganic base. The detailed standardized synthesis steps below outline the exact parameters for temperature, time, and workup procedures to ensure reproducible results across different scales of production.
- Mix imidazolium salt and silver halide (AgX) in a suitable solvent such as 3-chloropyridine or acetonitrile.
- Add an inorganic base like potassium carbonate and heat the mixture to 50-150°C for 2 to 48 hours.
- Purify the resulting complex by silica gel filtration and precipitation in alkanes to obtain the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift from silver oxide to silver halide-based synthesis represents a significant opportunity for cost optimization and risk mitigation. The elimination of light-sensitive reagents removes the need for expensive infrastructure upgrades and reduces the likelihood of batch failures due to accidental light exposure. This robustness ensures a more predictable production schedule, which is critical for maintaining continuity in the supply of downstream catalytic processes. Furthermore, the use of commodity chemicals like silver chloride and potassium carbonate stabilizes the raw material cost base, shielding the manufacturing process from the volatility often associated with specialized fine chemical reagents.
- Cost Reduction in Manufacturing: The substitution of expensive silver oxide with widely available silver halides drastically lowers the direct material costs associated with catalyst production. Additionally, the simplified purification process, which avoids complex chromatographic separations in favor of simple filtration and precipitation, reduces solvent consumption and labor hours. The removal of light-proofing requirements further decreases utility and maintenance costs related to specialized reactor setups. These cumulative efficiencies result in a substantially lower cost of goods sold, enhancing the overall margin profile for manufacturers of pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including various imidazolium salts and inorganic bases, are commercially available from multiple global suppliers, reducing dependency on single-source vendors. The insensitivity of the reaction to air and moisture means that storage and transportation conditions for intermediates are less stringent, lowering logistics costs and risks. This flexibility allows for larger inventory buffers and more resilient supply chains capable of withstanding market fluctuations or logistical disruptions without impacting production timelines.
- Scalability and Environmental Compliance: The one-step nature of the reaction and the use of recyclable solvents facilitate easy scale-up from laboratory to commercial production volumes. The process generates minimal hazardous waste compared to oxidative methods, simplifying effluent treatment and ensuring compliance with increasingly strict environmental regulations. The ability to run the reaction at moderate temperatures without high-pressure equipment further enhances the safety profile, making it an attractive option for contract manufacturing organizations looking to expand their catalytic capabilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable basis for process evaluation. Understanding these nuances is essential for R&D teams planning to integrate this technology into their existing workflows.
Q: Why is the silver salt method superior to the silver oxide method?
A: The silver salt method eliminates the need for light-proof operations required by photosensitive silver oxide, significantly simplifying reactor requirements and reducing operational costs.
Q: What are the typical reaction conditions for this synthesis?
A: The reaction typically proceeds at temperatures between 50°C and 150°C for 2 to 48 hours, using common inorganic bases and solvents like 3-chloropyridine.
Q: Is this process scalable for industrial production?
A: Yes, the process is insensitive to air and moisture and uses cheap, readily available raw materials, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Heterocyclic Carbene Silver Complex Supplier
The technological advancements detailed in patent CN102276634A underscore the potential for more efficient and economical production of critical organometallic catalysts. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring these innovations to market. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, ensuring that every batch of NHC silver complex meets the exacting standards required for high-performance catalysis in the pharmaceutical industry.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific catalytic needs. By partnering with our technical procurement team, you can access specific COA data and route feasibility assessments that demonstrate the tangible benefits of switching to this robust synthesis method. Let us help you optimize your supply chain and reduce manufacturing costs without compromising on quality or performance.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
