Advanced Purification Technology for 3-Azabicyclo Octane Hydrochloride: Enabling Scalable Gliclazide Production
The pharmaceutical industry continuously demands higher purity standards for critical intermediates, particularly those serving as the backbone for major antidiabetic medications like Gliclazide. Patent CN103183632A introduces a significant breakthrough in the purification methodology of 3-azabicyclo[3.3.0]octane hydrochloride, a pivotal building block in the synthesis of second-generation sulfonylureas. This innovation addresses long-standing challenges regarding product stability, transportability, and impurity profiles that have historically plagued the manufacturing of this specific heterocyclic amine salt. By transitioning from unstable concentrated solutions to a highly pure solid form, this technology offers a robust foundation for downstream synthesis, ensuring that the final API meets stringent regulatory requirements for safety and efficacy. The strategic implementation of this purification protocol allows manufacturers to secure a more reliable supply chain for high-purity pharmaceutical intermediates, directly impacting the quality consistency of the final therapeutic product.
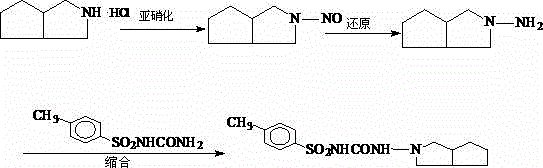
As illustrated in the reaction pathway above, the utility of 3-azabicyclo[3.3.0]octane extends critically into the production of Gliclazide, where its structural integrity determines the success of subsequent nitrosation and condensation steps. The ability to source this intermediate in a solid state with minimized impurity loads simplifies the entire synthetic sequence, reducing the burden on downstream purification units. For R&D directors and process chemists, understanding the transition from liquid to solid forms represents a key optimization in the overall process chemistry, facilitating better handling characteristics and more precise stoichiometric control in subsequent reactions. This patent effectively bridges the gap between laboratory-scale synthesis and industrial-scale reliability, providing a clear roadmap for upgrading existing production lines to meet modern quality standards without necessitating a complete overhaul of the synthetic strategy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-azabicyclo[3.3.0]octane hydrochloride has been fraught with significant logistical and quality control hurdles that impede efficient large-scale manufacturing. Conventional techniques often yielded the target compound as a concentrated solution rather than a discrete solid, resulting in products with inherently low purity and variable content levels. This liquid state presented severe disadvantages for transportation and long-term storage, as the stability of the solution could degrade over time, leading to batch-to-batch inconsistency. Furthermore, the presence of higher levels of impurities in these traditional concentrates necessitated extensive and costly downstream processing to achieve the purity required for pharmaceutical applications. The inability to isolate a stable solid form also complicated inventory management and increased the risk of contamination during transfer operations, creating bottlenecks in the supply chain that affected both cost and delivery timelines for global buyers.
The Novel Approach
The methodology disclosed in patent CN103183632A fundamentally transforms the production landscape by introducing a rigorous purification sequence that isolates the target molecule as a high-quality solid. By employing a specific recrystallization technique using a mixed solvent system of alcohol and ester, the process effectively removes trace organic impurities and residual solvents that typically persist in crude preparations. This approach not only elevates the purity of the final product to exceed 99% but also ensures that total impurities remain below 0.1%, with single impurities tightly controlled under 0.05%. The transition to a solid form dramatically enhances the material's shelf life and ease of handling, allowing for safer packaging and more efficient logistics. For procurement teams, this shift represents a tangible improvement in value, as the reduced need for additional refining steps downstream translates into lower overall processing costs and a more streamlined manufacturing workflow.
Mechanistic Insights into KBH4/ZnCl2-Catalyzed Reduction
The core chemical transformation driving this synthesis is the reduction of 1,2-cyclopentanedicarboximide to the corresponding amine, facilitated by a potent reducing system comprising potassium borohydride and anhydrous zinc chloride. This specific combination generates a highly reactive species in situ within a mixed solvent environment of dried toluene and tetrahydrofuran, capable of efficiently cleaving the imide bond under controlled thermal conditions. The reaction mechanism relies on the synergistic interaction between the Lewis acid character of zinc chloride and the hydride donor capability of potassium borohydride, which activates the carbonyl groups of the imide for nucleophilic attack. Maintaining a strict nitrogen atmosphere throughout the process is critical to prevent the decomposition of the reducing agents and to ensure the safety of the operation, given the potential for hydrogen gas evolution. The precise staging of temperature, initially holding at 70-75°C and subsequently ramping to a reflux range of 95-105°C, allows for the gradual and complete conversion of the starting material while minimizing the formation of side products that could complicate purification.
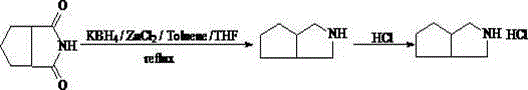
As depicted in the reaction scheme above, the reduction proceeds through a well-defined pathway that ultimately yields the free amine, which is subsequently converted to the hydrochloride salt for isolation. The control of impurities is intrinsically linked to the efficiency of this reduction step; incomplete reaction or harsh conditions can lead to over-reduction or polymerization byproducts. However, the optimized thermal profile described in the patent ensures that the reaction reaches completion without degrading the sensitive bicyclic framework. Following the reduction, the workup procedure involving pH adjustment to 13-14 followed by steam distillation serves as a critical purification checkpoint, separating the volatile amine from non-volatile inorganic salts and heavy organic residues. This multi-stage purification strategy, combining chemical selectivity during reduction with physical separation during workup and crystallization, creates a robust barrier against impurity carryover, ensuring the final solid meets the rigorous specifications demanded by the pharmaceutical sector.
How to Synthesize 3-Azabicyclo[3.3.0]octane Hydrochloride Efficiently
Implementing this synthesis requires careful attention to solvent drying and temperature gradients to maximize yield and purity. The process begins with the preparation of the reducing mixture, followed by the controlled addition of the imide substrate and a prolonged heating cycle to drive the reaction to completion. The final isolation involves a sophisticated acid-base extraction sequence culminating in a recrystallization step that defines the product's physical quality. Operators must adhere strictly to the specified pH ranges during the acidification and basification steps to ensure optimal recovery of the amine into the organic phase before salt formation.
- Prepare the reducing system by mixing potassium borohydride and anhydrous zinc chloride in a dried toluene and tetrahydrofuran solvent mixture under nitrogen protection at room temperature.
- Add 1,2-cyclopentanedicarboximide in batches, then heat the mixture to 70-75°C for 4 hours followed by reflux at 95-105°C for 10 hours to ensure complete reduction.
- Perform workup by adjusting pH to 13-14 with liquid alkali, steam distilling, extracting with organic solvent, acidifying to pH 1.5-2, and finally recrystallizing the crude solid using an alcohol-ester mixed solvent.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the adoption of this purification technology offers distinct strategic advantages that extend beyond simple chemical yield. The primary benefit lies in the transformation of the product from a hazardous, unstable liquid concentrate into a stable, free-flowing solid, which drastically simplifies warehousing and international shipping logistics. This physical change reduces the risk of leakage and degradation during transit, ensuring that the material arrives at the manufacturing site in consistent condition regardless of the journey duration. Furthermore, the high purity of the incoming raw material reduces the load on internal quality control laboratories and minimizes the consumption of solvents and reagents required for further purification, leading to substantial operational cost savings. By securing a supplier capable of delivering this upgraded specification, companies can mitigate the risk of production delays caused by out-of-spec raw materials.
- Cost Reduction in Manufacturing: The elimination of extensive downstream refining processes results in significant cost efficiencies throughout the production lifecycle. Because the intermediate arrives with purity levels exceeding 99%, manufacturers can reduce the number of recrystallization cycles or chromatographic separations typically required to meet API standards. This reduction in processing steps directly lowers energy consumption, solvent usage, and labor hours, contributing to a leaner and more cost-effective manufacturing model. Additionally, the improved yield reported in the patent examples suggests a more efficient utilization of starting materials, further driving down the cost per kilogram of the final active ingredient.
- Enhanced Supply Chain Reliability: The stability of the solid form ensures a more predictable and reliable supply chain, free from the volatility associated with liquid intermediates. Suppliers can maintain larger inventories of the stable solid without fear of rapid degradation, allowing them to respond more quickly to fluctuating market demands. This reliability is crucial for pharmaceutical companies that require just-in-time delivery to maintain continuous API production schedules. The robustness of the synthesis method also implies a lower rate of batch failures, ensuring a consistent flow of material that supports long-term production planning and contract fulfillment.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing common solvents like toluene and THF that can be readily recovered and recycled in a closed-loop system. The use of established unit operations such as steam distillation and filtration facilitates easy scale-up from pilot plant to commercial production volumes without requiring exotic equipment. From an environmental perspective, the high efficiency of the reaction and the ability to recycle solvents contribute to a reduced waste footprint, aligning with increasingly strict global regulations on chemical manufacturing emissions and waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this purified intermediate. These answers are derived directly from the experimental data and process descriptions found in the referenced patent documentation. Understanding these details is essential for technical teams evaluating the feasibility of integrating this material into their existing synthetic routes for Gliclazide and related compounds.
Q: What is the achieved purity level of the refined 3-azabicyclo octane hydrochloride?
A: According to the technical data in patent CN103183632A, the refined solid product achieves a content of greater than or equal to 99%, with total impurities controlled below 0.1% and single impurities below 0.05%.
Q: How does this purification method improve supply chain logistics compared to previous methods?
A: Traditional methods often resulted in low-purity concentrated solutions which were difficult to transport and store. This novel process yields a stable solid product, significantly enhancing storage stability and reducing transportation risks associated with liquid intermediates.
Q: What are the key reaction conditions for the reduction step?
A: The reduction utilizes a KBH4/ZnCl2 system in a toluene/THF mixed solvent. The process requires a staged heating profile: maintaining 70-75°C for 4 hours followed by a higher temperature reflux at 95-105°C for 10 hours under nitrogen protection.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Azabicyclo[3.3.0]octane Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful manufacture of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the rigorous standards outlined in patents like CN103183632A are met with precision. We operate stringent purity specifications and utilize rigorous QC labs to verify that every batch of 3-azabicyclo[3.3.0]octane hydrochloride meets the >99% purity benchmark required for Gliclazide synthesis. Our commitment to quality assurance means that our clients receive a product that is not only chemically superior but also fully documented and compliant with international regulatory expectations.
We invite global partners to engage with our technical procurement team to discuss how our advanced purification capabilities can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how switching to our high-purity solid intermediate can reduce your overall manufacturing expenses. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your production needs, ensuring a seamless integration of our materials into your pharmaceutical manufacturing processes.
