Advanced Synthesis of Vitamin A Intermediates: Overcoming Chlorohydrination Limitations with TCCA
The pharmaceutical and nutraceutical industries rely heavily on the efficient production of Vitamin A derivatives, such as retinyl acetate and retinyl palmitate, which serve critical roles in medicine and food additives. At the heart of this supply chain lies the synthesis of key C5 intermediates, specifically 1-chloro-2-methyl-4-alkanoyloxy-2-butene, often referred to as the "muriate" precursor. Patent CN1844077A introduces a transformative methodology for preparing this vital intermediate, addressing long-standing inefficiencies in the chlorohydrination of isoprene. By shifting from traditional hypochlorite sources to N-chlorinated isocyanuric acid series compounds, this technology promises a paradigm shift in how high-purity vitamin precursors are manufactured, offering a robust solution for reliable pharmaceutical intermediate supplier networks seeking to optimize their upstream synthesis capabilities.
The strategic importance of this patent cannot be overstated for stakeholders focused on cost reduction in pharmaceutical intermediates manufacturing. The conventional reliance on sodium hypochlorite or bleaching powder introduces significant quantities of inorganic salts and water into the reaction matrix, complicating downstream purification and waste management. In contrast, the novel approach detailed in CN1844077A utilizes reagents with exceptionally high available chlorine content, fundamentally altering the mass balance of the reaction. This results in a cleaner reaction profile, higher atom economy, and a drastic reduction in the environmental footprint associated with saline wastewater discharge, positioning this technology as a cornerstone for sustainable commercial scale-up of complex vitamin intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
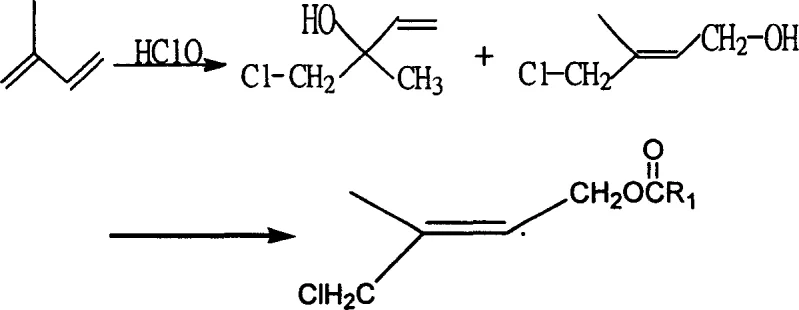
Historically, the industrial preparation of 1-chloro-2-methyl-4-alkanoyloxy-2-butene has relied heavily on the chlorohydrination of isoprene using sodium hypochlorite (bleach) or bleaching powder. While these reagents are inexpensive, they suffer from severe technical drawbacks that hinder process efficiency. Industrial grade sodium hypochlorite typically contains only 10% to 15% available chlorine, meaning that vast quantities of water and sodium ions are introduced into the reactor for every mole of active chlorine delivered. This excess water promotes hydrolysis side reactions, while the high concentration of alkali metal ions can catalyze unwanted polymerization of the sensitive isoprene diene system. Furthermore, the presence of free chlorine and heterogeneous ions often leads to significant formation of dichloro-addition byproducts, severely depressing the yield and purity of the desired chlorohydrin adducts. Comparative data from prior art indicates that these traditional methods often struggle to achieve crude product contents above 65%, necessitating energy-intensive purification steps that erode profit margins.
The Novel Approach
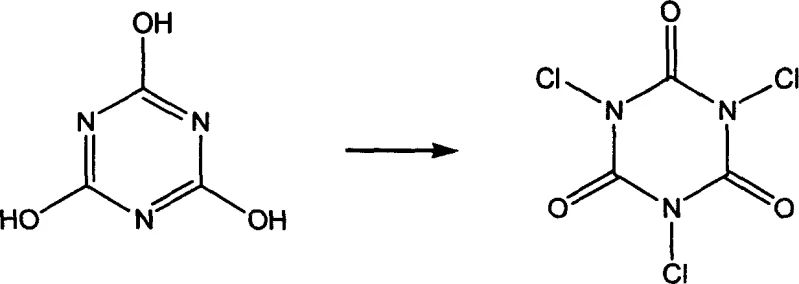
The innovation presented in Patent CN1844077A circumvents these limitations by employing N-chlorinated isocyanuric acid series compounds, such as Trichloroisocyanuric Acid (TCCA) or Sodium Dichloroisocyanurate (Surchlor GR 60), as the chlorinating agent. These solid reagents boast available chlorine contents ranging from 55% to over 90%, drastically reducing the amount of carrier water and extraneous ions required. The reaction proceeds under mild conditions, typically between -5°C and 10°C, where the reagent hydrolyzes in situ to generate hypochlorous acid in a controlled manner. This "gentle" release of the chlorinating species minimizes thermal runaways and suppresses the formation of dichloro-impurities. Experimental embodiments within the patent demonstrate that this switch in reagent technology can elevate crude product content to over 92% and yields to nearly 93%, representing a quantum leap in process performance compared to the sub-60% yields often observed with bleach. This superior selectivity directly translates to simplified workups and higher throughput for high-purity vitamin intermediates.
Mechanistic Insights into N-Chlorinated Isocyanurate Mediated Chlorohydrination
The mechanistic superiority of this process lies in the unique hydrolysis behavior of N-chlorinated isocyanurates. Unlike sodium hypochlorite, which exists as a pre-formed equilibrium of ions in solution, TCCA and its analogues react with water to release hypochlorous acid (HOCl) while precipitating cyanuric acid or its salts. This dynamic equilibrium ensures a steady, low-concentration supply of the active electrophile, which is crucial for controlling the regioselectivity of the addition to the conjugated diene system of isoprene. The absence of high concentrations of free hydroxide ions (which are prevalent in commercial bleach solutions) prevents the base-catalyzed degradation of the allylic alcohol intermediates. Furthermore, the solid nature of the byproduct (cyanuric acid) allows for easy physical separation via filtration, effectively removing the spent reagent scaffold from the organic phase without requiring extensive aqueous washing that would otherwise generate large volumes of effluent.
Impurity control is another critical aspect where this mechanism excels. In traditional systems, the presence of chloride ions and metal cations can facilitate electrophilic attack at multiple sites on the diene, leading to 1,2- and 1,4-dichloro adducts that are difficult to separate from the target mono-chlorohydrin. The new system, by maintaining a lower ionic strength and a more neutral pH environment during the initial addition phase, favors the formation of the desired 1-chloro-2-hydroxy-2-methyl-3-butylene and 1-chloro-2-methyl-4-hydroxyl-2-butylene isomers. These isomers are then efficiently converted to the final acetate or palmitate esters via acid-catalyzed rearrangement. The structural integrity of the carbon skeleton is preserved with minimal skeletal isomerization, ensuring that the downstream Wittig coupling with the C15 phosphonium salt proceeds with high stereochemical fidelity.
How to Synthesize 1-Chloro-2-methyl-4-alkanoyloxy-2-butene Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this technology at scale. The process begins with the suspension of the N-chlorinated isocyanurate in water, followed by the controlled addition of isoprene at low temperatures to manage the exotherm. Following the chlorohydrination, the organic layer is separated, and the crude chlorohydrins are subjected to esterification using acid anhydrides like acetic anhydride in the presence of an acid catalyst. This one-pot or telescoped approach minimizes handling of unstable intermediates. For detailed operational parameters, stoichiometry, and safety considerations, please refer to the standardized synthesis guide below.
- Conduct chlorohydrination of isoprene using N-chlorinated isocyanuric acid (e.g., Trichloroisocyanuric acid) in water at -5°C to 10°C to form chlorohydrin adducts.
- Separate the organic layer containing the crude chlorohydrin mixture from the aqueous phase and the solid byproduct (cyanuric acid).
- Perform acid-catalyzed esterification and rearrangement of the crude chlorohydrin with acid anhydrides (e.g., acetic anhydride) to yield the final chloride intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this TCCA-based route offers compelling economic and logistical advantages that extend beyond simple yield metrics. The primary driver of value is the drastic simplification of the waste stream. Traditional bleach-based processes generate massive volumes of saline wastewater containing dissolved organics, which requires expensive treatment before discharge. By switching to a solid reagent that precipitates its own byproduct, the liquid waste load is significantly reduced, lowering both disposal costs and regulatory compliance burdens. Additionally, the higher selectivity of the reaction means that less raw material is lost to tar and heavy ends, improving the overall mass balance and reducing the cost per kilogram of the final API intermediate.
- Cost Reduction in Manufacturing: The elimination of excess water and inorganic salts from the reaction mixture reduces the energy load required for heating, cooling, and solvent recovery. Since the reagent has a high available chlorine content, the molar equivalents required are lower by weight compared to dilute bleach, optimizing logistics and storage density. The ability to recycle the cyanuric acid byproduct further offsets raw material costs, creating a circular economy element within the production facility that is absent in linear bleach consumption models.
- Enhanced Supply Chain Reliability: Isoprene is a commodity chemical, but its efficient conversion is often the bottleneck. This robust process tolerates slight variations in feed quality better than sensitive hypochlorite reactions, ensuring consistent batch-to-batch output. The use of stable, solid chlorinating agents also mitigates the supply risks associated with the transportation and storage of hazardous liquid bleach, which has strict shelf-life and stability constraints. This stability ensures that production schedules are not disrupted by reagent degradation.
- Scalability and Environmental Compliance: The mild reaction temperatures (-5°C to 10°C) are easily achievable with standard industrial glycol cooling systems, removing the need for cryogenic infrastructure. The reduction in hazardous waste generation aligns perfectly with increasingly stringent global environmental regulations, future-proofing the manufacturing site against tighter discharge limits. The process is inherently safer due to the controlled release of chlorine, reducing the risk of runaway exotherms common in large-scale chlorinations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented chlorohydrination technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering a realistic view of the process capabilities and limitations for potential adopters.
Q: Why is Trichloroisocyanuric Acid (TCCA) preferred over Sodium Hypochlorite for this synthesis?
A: TCCA offers a significantly higher available chlorine content (up to 90%) compared to industrial sodium hypochlorite (10-15%). This reduces the volume of water and inorganic salts introduced into the system, minimizing side reactions like dichloro-addition and simplifying waste treatment.
Q: What are the primary impurities controlled in this new process?
A: The process effectively suppresses the formation of dichloro-addition byproducts and polymerization tars often seen in traditional hypochlorite methods, resulting in crude product contents exceeding 90%.
Q: Can the byproduct cyanuric acid be recycled?
A: Yes, the solid byproduct generated is cyanuric acid (or its salts), which can be filtered off and potentially recycled or sold, unlike the massive amounts of saline wastewater generated by bleach-based processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Chloro-2-methyl-4-alkanoyloxy-2-butene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven manufacturing capability. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of patent CN1844077A can be realized at the volumes your business demands. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Vitamin A intermediate meets the exacting standards required for pharmaceutical and nutraceutical applications.
We invite you to collaborate with us to evaluate the feasibility of this high-efficiency route for your specific supply chain needs. Our technical team is prepared to provide a Customized Cost-Saving Analysis that quantifies the potential reductions in waste treatment and raw material consumption for your operation. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the global Vitamin A market.
