Advanced Crystallization Technology for Scalable Production of Protected Deoxypurine Nucleosides
The rapid advancement of genomic medicine and the burgeoning demand for antisense DNA therapeutics have placed unprecedented pressure on the supply chain for high-purity nucleoside intermediates. As the building blocks for oligonucleotide synthesis, 5'-protected 2'-deoxypurine nucleosides must meet stringent purity specifications to prevent the propagation of errors in the final drug substance. Historically, achieving this level of purity has been a bottleneck, relying heavily on inefficient separation techniques. However, a groundbreaking approach detailed in patent CN1380299A introduces a novel purification strategy that leverages solvent inclusion crystallization. This technology represents a paradigm shift from traditional chromatographic methods, offering a robust pathway to isolate target compounds like N2-isobutyryl-5'-O-(4,4'-dimethoxytrityl)-2'-deoxyguanosine with exceptional efficiency. By transforming the physical state of the molecule through specific solvent interactions, manufacturers can now achieve purities exceeding 99.7% without the prohibitive costs associated with large-scale column chromatography.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
For decades, the industry standard for purifying protected deoxypurine nucleosides has been column chromatography, a technique fraught with significant operational and economic drawbacks for large-scale manufacturing. While chromatography is effective at separating compounds with vastly different polarities, it struggles immensely with structural isomers that possess nearly identical physicochemical properties, such as the troublesome 3'-alternative isomer often found in these syntheses. The process requires vast quantities of silica gel and organic solvents, leading to enormous equipment footprints and substantial waste generation that complicates environmental compliance. Furthermore, the batch nature of column loading and elution creates inherent bottlenecks in throughput, making it difficult to meet the surging global demand for antisense drug precursors. The difficulty in controlling solvent ratios during traditional redeposition methods often results in oilification or the formation of thick syrups rather than distinct crystals, rendering the purification unsuccessful and forcing reliance on these cumbersome chromatographic columns.
The Novel Approach
The innovative method disclosed in the patent data circumvents these limitations by utilizing the phenomenon of solvent inclusion to drive purification through crystallization. Instead of relying on differential adsorption on silica, this process dissolves the crude reaction mixture in a liquid medium containing a specific nitrile solvent, such as acetonitrile, which acts as a lattice-forming agent. As the solution conditions are optimized, the target 5'-protected nucleoside selectively precipitates as an inclusion crystal, physically trapping solvent molecules within its crystal structure while excluding impurities. 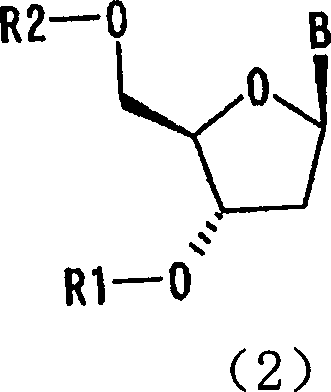 This selective crystallization is particularly powerful because the 3'-isomers and polysubstituted byproducts remain soluble in the mother liquor due to their inability to form the same stable inclusion complex. The result is a direct isolation of high-purity solid product, exemplified by the successful purification of guanosine and adenosine derivatives shown in specific embodiments, achieving yields of over 84% and purities approaching 99.7% in a single step.
This selective crystallization is particularly powerful because the 3'-isomers and polysubstituted byproducts remain soluble in the mother liquor due to their inability to form the same stable inclusion complex. The result is a direct isolation of high-purity solid product, exemplified by the successful purification of guanosine and adenosine derivatives shown in specific embodiments, achieving yields of over 84% and purities approaching 99.7% in a single step.
Mechanistic Insights into Solvent Inclusion Crystallization
The core mechanism driving this purification breakthrough is the formation of a host-guest complex between the nucleoside derivative and the nitrile solvent molecules. Unlike simple solvation where solvent molecules surround a solute loosely, inclusion crystallization involves the solvent becoming an integral part of the crystal lattice itself. 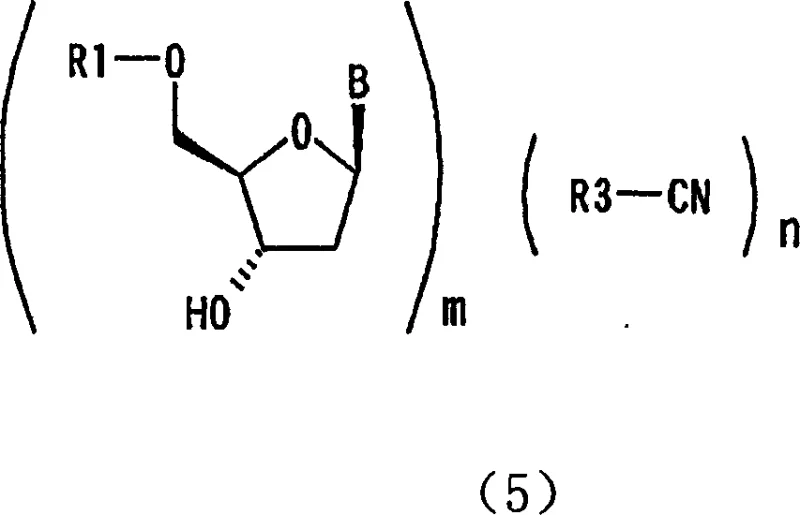 In this specific application, solvents like acetonitrile fit precisely into the voids of the nucleoside crystal structure, stabilizing the lattice energy and lowering the solubility of the target compound relative to its impurities. This thermodynamic stability ensures that as the crystals grow, they preferentially incorporate the desired 5'-protected isomer. The stoichiometry of this inclusion can vary, with experimental data indicating ratios such as 2:1 or 1:1 between the nucleoside and acetonitrile depending on drying conditions, yet the crystalline integrity remains intact. This mechanism effectively acts as a molecular sieve, where the geometric and electronic requirements for lattice formation reject the 3'-isomers which possess different spatial configurations that disrupt the inclusion geometry.
In this specific application, solvents like acetonitrile fit precisely into the voids of the nucleoside crystal structure, stabilizing the lattice energy and lowering the solubility of the target compound relative to its impurities. This thermodynamic stability ensures that as the crystals grow, they preferentially incorporate the desired 5'-protected isomer. The stoichiometry of this inclusion can vary, with experimental data indicating ratios such as 2:1 or 1:1 between the nucleoside and acetonitrile depending on drying conditions, yet the crystalline integrity remains intact. This mechanism effectively acts as a molecular sieve, where the geometric and electronic requirements for lattice formation reject the 3'-isomers which possess different spatial configurations that disrupt the inclusion geometry.
From an impurity control perspective, this mechanism offers a decisive advantage over thermal or polarity-based separations. The 3'-isomer, which is the most persistent contaminant in tritylation reactions, typically has a polarity very similar to the 5'-product, making chromatographic separation inefficient and solvent-intensive. However, in the inclusion crystallization system, the 3'-isomer fails to co-crystallize with the nitrile solvent. Consequently, it stays in the supernatant liquid phase even as the bulk of the product precipitates out. Thermal analysis data from the patent, specifically DSC and TG-DTA curves, confirms the presence of the solvent within the crystal lattice through distinct endothermic peaks corresponding to solvent release. This provides a verifiable quality control marker; the presence of the specific endotherm confirms the formation of the correct polymorph and high purity, whereas amorphous oils or incorrect crystal forms lack this thermal signature. This intrinsic link between crystal structure and purity simplifies the analytical burden on quality assurance teams.
How to Synthesize 5'-Protected 2'-Deoxypurine Nucleoside Efficiently
Implementing this purification technology requires precise control over solvent composition and temperature to maximize the yield of the inclusion crystal. The process begins with the preparation of a crude mixture containing the target nucleoside along with typical synthesis byproducts. This crude material is dissolved in a minimal amount of a primary solvent, often followed by the addition of the critical nitrile component or a mixture where the nitrile is the dominant species. 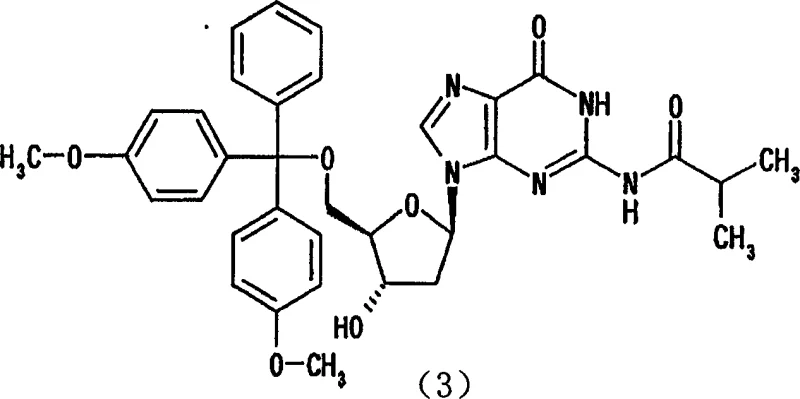 The solution is then subjected to controlled cooling or standing at ambient temperature to induce nucleation. It is crucial to maintain the solvent ratio within the optimal range described in the patent, typically ensuring the nitrile content is sufficient to drive inclusion but not so high as to cause premature precipitation of impurities. Once the crystals have fully formed, they are separated via filtration and washed with a non-inclusion solvent to remove surface impurities. The final drying step must be carefully managed; while vacuum drying removes excess surface solvent, the core inclusion solvent may remain bound until higher temperatures are applied, which is acceptable as long as the chemical purity is maintained. Detailed standardized synthetic steps see the guide below.
The solution is then subjected to controlled cooling or standing at ambient temperature to induce nucleation. It is crucial to maintain the solvent ratio within the optimal range described in the patent, typically ensuring the nitrile content is sufficient to drive inclusion but not so high as to cause premature precipitation of impurities. Once the crystals have fully formed, they are separated via filtration and washed with a non-inclusion solvent to remove surface impurities. The final drying step must be carefully managed; while vacuum drying removes excess surface solvent, the core inclusion solvent may remain bound until higher temperatures are applied, which is acceptable as long as the chemical purity is maintained. Detailed standardized synthetic steps see the guide below.
- Dissolve the crude 5'-protected 2'-deoxypurine nucleoside in a suitable liquid medium, preferably containing a nitrile solvent such as acetonitrile.
- Allow the solution to stand or cool to facilitate the formation of inclusion crystals where the solvent molecules are trapped within the crystal lattice.
- Filter the formed crystals and wash them to remove impurities like 3'-isomers remaining in the mother liquor, then dry under vacuum to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from column chromatography to inclusion crystallization represents a substantial opportunity for cost optimization and risk mitigation. The traditional reliance on chromatography introduces significant variability in lead times due to column packing, equilibration, and the slow flow rates required for high-resolution separation. By replacing this with a batch crystallization process, manufacturers can drastically reduce the cycle time per batch, thereby increasing the overall throughput of the facility without the need for capital-intensive expansion. Furthermore, the elimination of tons of silica gel and the reduction in total solvent volume per kilogram of product directly translates to lower raw material costs and reduced waste disposal fees. This process intensification allows for a more predictable supply of critical antisense DNA intermediates, shielding downstream drug manufacturers from the volatility often associated with complex purification steps.
- Cost Reduction in Manufacturing: The economic impact of eliminating column chromatography cannot be overstated, as it removes the need for expensive stationary phases and the massive infrastructure required to handle large solvent volumes. In a chromatographic process, the ratio of solvent to product can be exceedingly high, driving up both procurement costs for solvents and the energy costs for their recovery and distillation. By shifting to a crystallization-based workflow, the solvent usage is optimized to the saturation limits of the inclusion complex, significantly lowering the operational expenditure per unit. Additionally, the simplicity of the equipment—standard reactors and filters versus specialized chromatography columns—reduces maintenance costs and minimizes the risk of equipment failure that could halt production lines.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the scalability inherent in this crystallization method. Chromatography is notoriously difficult to scale linearly; increasing column diameter often leads to channeling and loss of resolution, requiring parallel trains of columns that complicate operations. In contrast, crystallization scales predictably with vessel size, allowing manufacturers to move seamlessly from pilot batches to multi-ton commercial production. This scalability ensures that as demand for oligonucleotide therapies grows, the supply of key intermediates can be ramped up rapidly without re-engineering the entire process. The use of common industrial solvents like acetonitrile further secures the supply chain, as these materials are readily available globally, unlike specialized chromatographic resins which may have limited suppliers.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this technology offers a greener alternative that aligns with modern sustainability goals. The reduction in solvent waste directly lowers the carbon footprint of the manufacturing process and simplifies the burden on wastewater treatment facilities. Moreover, the avoidance of silica dust and the reduced handling of hazardous solvent mixtures improve workplace safety conditions. The ability to recycle the mother liquor, which contains the dissolved impurities and excess solvent, further enhances the atom economy of the process. This environmental efficiency not only reduces regulatory compliance risks but also appeals to pharmaceutical partners who are increasingly scrutinizing the sustainability practices of their active pharmaceutical ingredient (API) and intermediate suppliers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this inclusion crystallization technology for nucleoside purification. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for evaluating the process viability. Understanding these details is crucial for technical teams assessing the feasibility of adopting this method for their specific product portfolios. The answers highlight the robustness of the method against common impurities and its adaptability to various protecting group strategies.
Q: Why is acetonitrile preferred for this purification process?
A: Acetonitrile acts as a specific inclusion solvent that forms a stable crystal lattice with the target 5'-protected nucleoside. This unique interaction allows the target compound to crystallize while leaving structurally similar impurities, such as 3'-isomers and polysubstituted byproducts, dissolved in the mother liquor.
Q: What specific impurities does this method remove effectively?
A: This crystallization method is highly effective at removing difficult-to-separate impurities like 3'-alternative isomers and 3',5'-bis-substituted compounds, which are notoriously challenging to eliminate using standard recrystallization or redeposition techniques.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, unlike column chromatography which requires massive equipment volumes and is difficult to scale, this inclusion crystallization method utilizes standard batch reactors and filtration equipment, making it ideal for commercial-scale production of antisense DNA intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5'-Protected 2'-Deoxypurine Nucleoside Supplier
At NINGBO INNO PHARMCHEM, we recognize that the purity of nucleoside intermediates is the foundation of successful antisense drug development. Our technical team has extensively analyzed advanced purification technologies like the solvent inclusion crystallization method to ensure our manufacturing processes meet the highest global standards. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you are in preclinical development or full-scale commercialization, our supply capabilities match your needs. Our rigorous QC labs employ stringent purity specifications, utilizing HPLC and thermal analysis to verify that every batch of 5'-protected deoxypurine nucleoside is free from critical 3'-isomers and other structural impurities.
We invite you to collaborate with us to optimize your supply chain for oligonucleotide synthesis. Our experts are ready to provide a Customized Cost-Saving Analysis demonstrating how our advanced crystallization capabilities can reduce your overall cost of goods. Please contact our technical procurement team today to request specific COA data for our current inventory or to discuss route feasibility assessments for your custom nucleoside projects. Let us be your partner in delivering high-quality, cost-effective solutions for the next generation of genomic medicines.
