Advanced Synthetic Route for High-Purity Benzimidazole Intermediates in Electronic Materials
Introduction to Advanced Benzimidazole Intermediate Manufacturing
The global demand for high-performance polyimide materials, particularly those utilized in electronic applications such as flexible circuits, high-temperature films, and advanced battery separators, continues to drive innovation in precursor synthesis. Patent CN115850188A introduces a groundbreaking preparation method for 2-(2-chloro-4-aminophenyl)-5-aminobenzimidazole (CAS 70500-01-5), a critical monomer that imparts exceptional thermal stability and mechanical rigidity to polymer chains. This technical breakthrough addresses long-standing inefficiencies in traditional manufacturing, offering a pathway to ultra-high purity intermediates essential for next-generation electronic materials. By leveraging a novel sequence of chlorination, condensation, and catalytic hydrogenation, this process ensures consistent quality and scalability that modern supply chains require.
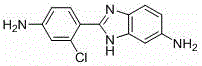
For R&D directors and procurement specialists, understanding the structural integrity of this molecule is paramount. The presence of the chlorine atom and the dual amino groups allows for versatile polymerization reactions, resulting in polyimides with superior dielectric properties and dimensional stability. The patent explicitly details a route that avoids the pitfalls of earlier methodologies, ensuring that the final product meets the stringent specifications demanded by the semiconductor and aerospace industries. This report analyzes the technical nuances of this synthesis, highlighting its potential to redefine cost structures and supply reliability for fine chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the industry standard for synthesizing this benzimidazole derivative relied heavily on the methodology disclosed in U.S. Pat. No. 4,929,947A. This legacy approach involves the direct reaction of 4-nitrophthalenediamine with 2-chloro-4-nitrobenzoyl chloride. While chemically feasible, this conventional route suffers from significant drawbacks that hinder its viability for modern high-volume manufacturing. The most critical limitation is the suboptimal reaction yield, which historically hovers around merely 69%, leading to substantial material loss and increased raw material costs per kilogram of output. Furthermore, the purification profile of the product obtained via this older method is inadequate for high-end applications, with reported purity levels often stagnating between 91% and 92%.
These purity deficiencies necessitate extensive downstream purification steps, such as repeated recrystallizations or chromatography, which further erode overall yield and extend production lead times. For supply chain managers, the inconsistency in quality poses a risk to batch-to-batch reproducibility, a critical factor when supplying multinational corporations in the electronics sector. The reliance on less efficient coupling strategies also generates higher volumes of chemical waste, complicating environmental compliance and increasing disposal costs. Consequently, there has been a pressing need for a more robust synthetic strategy that can deliver both higher throughput and superior chemical fidelity.
The Novel Approach
The innovative process outlined in patent CN115850188A fundamentally restructures the synthetic logic to overcome these historical bottlenecks. Instead of starting with phthalenediamine, the new method initiates with 2-chloro-4-nitrobenzoic acid, converting it first into a reactive acid chloride intermediate. This activated species is then condensed with 2,4-dinitroaniline to form a stable dinitro-amide precursor. This strategic shift allows for better control over the reaction kinetics and minimizes side reactions that typically plague direct amidation processes. The subsequent step involves a powerful catalytic hydrogenation that simultaneously reduces multiple nitro groups, streamlining the transformation into the diamino intermediate required for ring closure.
This novel approach delivers a dramatic improvement in process efficiency, with experimental data demonstrating overall molar yields reaching up to 87.8% in optimized examples. More importantly, the final product purity consistently exceeds 99.7% as measured by HPLC, effectively eliminating the need for complex purification protocols. The integration of a closed-loop system where byproduct hydrogen chloride is captured and reused further enhances the economic and environmental profile of the process. For manufacturers, this translates to a streamlined workflow that reduces operational complexity while maximizing output quality, positioning this method as the new gold standard for producing high-value benzimidazole intermediates.
Mechanistic Insights into Catalytic Hydrogenation and Cyclization
The core of this synthetic advancement lies in the precise orchestration of the reduction and cyclization phases. In the second stage of the process, the intermediate N-(2-chloro-4-nitrobenzoyl)-2,4-dinitrobenzene undergoes catalytic hydrogenation. This step is critical as it must selectively reduce three distinct nitro groups to amino groups without affecting the chloro substituent or the amide linkage. The patent specifies the use of noble metal catalysts such as Platinum on Carbon (Pt/C) or Palladium on Carbon (Pd/C), which provide the necessary activity to drive this multi-site reduction under moderate pressure conditions ranging from 0.2 to 2.0 MPa. The choice of solvent, typically ethanol or methanol, plays a vital role in solubilizing the intermediate while maintaining catalyst stability.
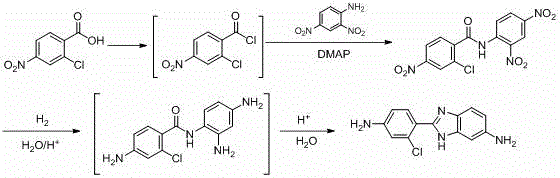
Following the reduction, the resulting tri-amino intermediate spontaneously undergoes an acid-catalyzed intramolecular cyclization to form the benzimidazole ring. The mechanism involves the nucleophilic attack of the ortho-amino group on the carbonyl carbon of the amide, followed by dehydration. The patent highlights the versatility of using either inorganic acids like hydrochloric acid or sulfuric acid to facilitate this ring closure. A key mechanistic advantage is the ability to perform this cyclization in the same pot or in a sequential manner without isolating the unstable diamine intermediate, thereby preventing oxidation and degradation. This tandem reduction-cyclization strategy not only simplifies the operational procedure but also ensures that the final molecular architecture is formed with high regioselectivity and minimal impurity formation.
Impurity control is inherently built into this mechanism through the high selectivity of the hydrogenation catalyst and the thermodynamic stability of the final benzimidazole ring. By avoiding harsh reducing agents like iron powder or tin chloride, which often generate heavy metal sludge and difficult-to-remove byproducts, the catalytic hydrogenation route ensures a cleaner reaction profile. The recovery of hydrogen chloride gas generated during the initial condensation step and its reuse in the cyclization step creates a synergistic effect that maintains the acidic environment necessary for ring closure while neutralizing basic amines. This careful balance of reaction parameters ensures that the final product emerges with the high purity levels required for sensitive electronic applications.
How to Synthesize 2-(2-Chloro-4-Aminophenyl)-5-Aminobenzimidazole Efficiently
Implementing this synthesis requires strict adherence to the reaction conditions detailed in the patent to ensure safety and optimal yield. The process begins with the activation of the carboxylic acid using chlorinating agents like triphosgene or thionyl chloride in a toluene solvent system, often enhanced with a catalytic amount of DMF. Once the acid chloride is formed, it is reacted with 2,4-dinitroaniline in the presence of a base catalyst such as DMAP to form the dinitro-amide. The subsequent hydrogenation step requires a high-pressure reactor capable of handling hydrogen gas safely, utilizing a Pt/C catalyst at temperatures between 60°C and 100°C. Finally, the cyclization is induced by heating the reduced intermediate in an acidic aqueous medium.
- Convert 2-chloro-4-nitrobenzoic acid to its acid chloride using thionyl chloride or triphosgene, then condense with 2,4-dinitroaniline to form the dinitro-amide intermediate.
- Perform catalytic hydrogenation on the dinitro-amide intermediate using Pt/C or Pd/C to reduce all nitro groups to amino groups simultaneously.
- Execute an acid-catalyzed cyclization reaction using hydrochloric or sulfuric acid to close the benzimidazole ring and isolate the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers tangible strategic benefits that extend beyond simple chemical transformation. The primary advantage lies in the substantial increase in overall process yield, which directly correlates to a reduction in the cost of goods sold (COGS). By improving the molar yield from the historical baseline of roughly 69% to nearly 88%, manufacturers can produce significantly more product from the same quantity of raw materials. This efficiency gain mitigates the impact of raw material price volatility and allows for more competitive pricing structures in the global market. Furthermore, the elimination of extensive purification steps reduces solvent consumption and energy usage, contributing to a leaner and more cost-effective manufacturing operation.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the high atom economy and the recycling of reagents. Specifically, the recovery and reuse of hydrogen chloride gas eliminate the need to purchase fresh acid for the cyclization step and reduce waste treatment costs associated with acidic effluents. Additionally, the use of heterogeneous catalysts like Pt/C allows for catalyst recovery and regeneration, further lowering the recurring cost of consumables. The high purity of the crude product means that expensive downstream processing, such as column chromatography or multiple recrystallizations, is largely unnecessary, resulting in significant savings in labor, time, and utility costs.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply continuity by reducing the risk of batch failures. The use of stable intermediates and well-defined reaction conditions ensures consistent output quality, which is crucial for maintaining long-term contracts with downstream polymer manufacturers. The availability of multiple options for chlorinating agents (thionyl chloride or triphosgene) and solvents (toluene, xylene, DMF) provides flexibility in sourcing, allowing supply chain managers to adapt to regional availability and regulatory constraints without compromising the process integrity. This flexibility acts as a buffer against supply disruptions, ensuring a steady flow of critical intermediates.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method is designed for industrial scalability. The avoidance of stoichiometric metal reductants reduces the generation of heavy metal waste, simplifying compliance with increasingly stringent environmental regulations. The process operates at moderate pressures and temperatures that are easily manageable in standard stainless steel reactors, facilitating seamless scale-up from pilot plant to commercial production volumes. The closed-loop nature of the HCl recovery system demonstrates a commitment to green chemistry principles, which is increasingly becoming a prerequisite for suppliers aiming to partner with top-tier multinational corporations focused on sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of this benzimidazole intermediate. These answers are derived directly from the experimental data and claims presented in patent CN115850188A, providing a reliable foundation for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this material into their existing supply chains and product formulations.
Q: What are the purity advantages of the new synthetic method compared to prior art?
A: The new method described in patent CN115850188A achieves product purity exceeding 99.7% (HPLC), significantly outperforming the conventional US Pat. 4,929,947A method which typically yields purity around 91-92%.
Q: How does this process improve environmental compliance for manufacturers?
A: The process incorporates a hydrogen chloride gas recovery system during the condensation step. The recovered HCl is dissolved in deionized water and reused as the acidic catalyst in the subsequent cyclization step, minimizing waste discharge.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the method utilizes robust reagents like triphosgene or thionyl chloride and standard catalytic hydrogenation conditions (0.2-2.0 MPa), making it highly adaptable for commercial scale-up with overall molar yields reaching up to 87.8%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(2-Chloro-4-Aminophenyl)-5-Aminobenzimidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the development of advanced electronic materials and pharmaceuticals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global clients. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. Our capability to implement the advanced synthetic routes described in recent patents allows us to offer superior quality materials that outperform traditional market standards.
We invite you to collaborate with us to optimize your supply chain for benzimidazole derivatives. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us be your partner in driving innovation and efficiency in your manufacturing processes.
