Scalable Synthesis of 2-Amino-3-methoxy-5-bromoxynil for Commercial API Production
The pharmaceutical industry constantly seeks robust synthetic pathways that balance high purity with operational safety, and the recent disclosure in patent CN112300028B offers a compelling solution for the production of 2-amino-3-methoxy-5-bromoxynil. This critical intermediate serves as a foundational building block for various bioactive molecules, yet its historical manufacturing processes have been plagued by significant environmental and safety hurdles. The patented methodology introduces a strategic shift away from hazardous oxidation protocols, utilizing a sequence of nucleophilic substitutions and cyclizations that are inherently more controllable and scalable. By anchoring the synthesis on readily available 4-bromo-2-fluoro-1-nitrotoluene, the process eliminates the reliance on expensive and unstable precursors, thereby establishing a new benchmark for efficiency in fine chemical manufacturing. This technical breakthrough not only addresses the immediate needs of process chemists for cleaner reaction profiles but also aligns perfectly with the long-term sustainability goals of modern supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-amino-3-methoxy-5-bromoxynil has relied heavily on the oxidation of methyl-substituted phenols using potassium permanganate, a reagent known for its volatility and potential for exothermic runaway reactions. As illustrated in the prior art pathway, this conventional approach necessitates the methylation of 3-methyl-2-nitrophenol followed by a harsh oxidation step that frequently results in suboptimal yields and the generation of substantial quantities of manganese-containing wastewater. The handling of large amounts of potassium permanganate on an industrial scale introduces severe safety liabilities, including the risk of explosion and the complexity of managing heavy metal waste streams which require costly remediation procedures. Furthermore, the starting materials for this legacy route are often subject to market volatility and supply chain bottlenecks, creating unpredictable procurement landscapes for manufacturers who depend on consistent batch-to-batch quality. The cumulative effect of these factors is a manufacturing process that is not only environmentally burdensome but also economically inefficient due to the high costs associated with waste treatment and low overall material throughput.
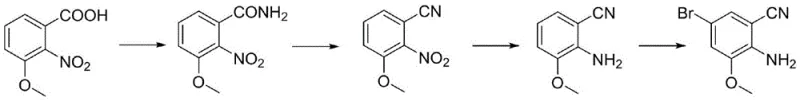
The Novel Approach
In stark contrast to the oxidative challenges of the past, the novel approach detailed in the patent leverages a constructive build-up strategy starting from 4-bromo-2-fluoro-1-nitrotoluene, which undergoes a mild nucleophilic aromatic substitution with sodium methoxide to install the crucial methoxy functionality. This initial transformation sets the stage for a series of high-yielding reactions, including an iron-mediated reduction of the nitro group that avoids the use of catalytic hydrogenation equipment, thus lowering capital expenditure requirements for the production facility. The subsequent formation of the isoxazole ring via condensation with chloral hydrate and hydroxylamine hydrochloride proceeds under controlled thermal conditions, ensuring minimal formation of regio-isomeric impurities that often plague heterocyclic syntheses. By replacing the dangerous permanganate oxidation with a phosphorus pentoxide-mediated dehydration in the final step, the process achieves a dramatic improvement in atom economy and operational safety. This redesigned synthetic tree effectively decouples the production of this key intermediate from the regulatory and logistical constraints associated with heavy metal oxidants, paving the way for a more resilient and cost-effective supply model.
Mechanistic Insights into Isoxazole Ring Construction and Dehydration
The core of this synthetic innovation lies in the precise construction of the isoxazole heterocycle, which is achieved through a sophisticated cascade involving the reaction of an amino-toluene derivative with chloral hydrate to generate an aldehyde intermediate in situ. This aldehyde immediately reacts with hydroxylamine hydrochloride to form an aldoxime species, which then undergoes an acid-catalyzed cyclization in concentrated sulfuric acid to close the ring and form the isoxazole scaffold. The mechanistic elegance of this step ensures that the bromine substituent remains intact throughout the harsh acidic conditions, preserving the halogen handle required for downstream coupling reactions in API synthesis. Following ring closure, the pathway employs a hydrogen peroxide and sodium hydroxide system to oxidize the intermediate to the corresponding carboxylic acid, a transformation that is remarkably selective and avoids over-oxidation of the sensitive amino group. The final conversion to the nitrile functionality is accomplished using phosphorus pentoxide in a high-boiling solvent like xylene, which drives the dehydration equilibrium forward by sequestering the water produced, thereby pushing the reaction to completion with exceptional conversion rates.
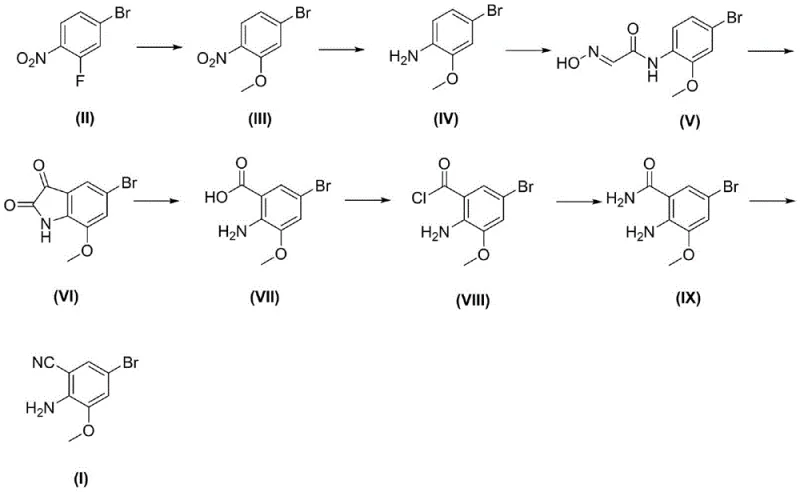
Impurity control is rigorously maintained throughout this sequence by leveraging the distinct solubility profiles of the intermediates, allowing for effective purification via simple crystallization or pulping techniques rather than complex chromatography. For instance, the isolation of the amide intermediate involves cooling the reaction mixture and pulping with water, which effectively washes away soluble inorganic salts and unreacted reagents while retaining the product in high purity. The use of iron powder for reduction, while traditional, is optimized in this process by controlling the pH with acetic acid, which prevents the formation of insoluble iron sludge that can trap product and reduce yield. Furthermore, the final dehydration step is monitored via HPLC to ensure complete consumption of the amide precursor, preventing the carryover of polar impurities into the final nitrile product. This attention to mechanistic detail and purification logic ensures that the final 2-amino-3-methoxy-5-bromoxynil meets the stringent purity specifications required for pharmaceutical applications, typically exceeding 98 percent purity without the need for extensive recrystallization.
How to Synthesize 2-Amino-3-methoxy-5-bromoxynil Efficiently
The execution of this synthesis requires careful attention to temperature control and reagent stoichiometry, particularly during the exothermic reduction and cyclization phases where thermal management is critical for safety and yield. The patent outlines a standardized protocol that begins with the nucleophilic substitution in methanol, followed by the iron reduction in a mixed solvent system of ethanol, water, and acetic acid to ensure homogeneous reaction conditions. Subsequent steps involve the formation of the aldoxime in dioxane or DMF, followed by the critical sulfuric acid cyclization which must be quenched carefully into ice water to precipitate the product.
- Perform nucleophilic aromatic substitution on 4-bromo-2-fluoro-1-nitrotoluene using sodium methoxide to install the methoxy group.
- Reduce the nitro group to an amine using iron powder in acidic media, followed by condensation with chloral hydrate and hydroxylamine.
- Execute acid-catalyzed cyclization to form the isoxazole ring, followed by oxidation, amidation, and final dehydration with phosphorus pentoxide.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this patented route offers transformative advantages by substituting volatile and regulated reagents with stable, commodity-grade chemicals that are readily available in the global market. The elimination of potassium permanganate removes a significant regulatory burden related to the transport and storage of strong oxidizers, while simultaneously reducing the costs associated with hazardous waste disposal and environmental compliance reporting. By utilizing starting materials like 4-bromo-2-fluoro-1-nitrotoluene, which is produced on a massive scale for other agrochemical and pharmaceutical applications, manufacturers can leverage existing supply chains to secure favorable pricing and ensure continuity of supply even during market disruptions. The robustness of the process, characterized by high yields in every step, means that less raw material is required to produce the same amount of finished goods, directly translating to a lower cost of goods sold and improved margin protection for downstream API producers.
- Cost Reduction in Manufacturing: The replacement of expensive and low-yielding oxidation steps with high-efficiency cyclization and dehydration reactions drastically reduces the consumption of raw materials per kilogram of product. By avoiding the use of precious metal catalysts or specialized oxidizing agents, the process minimizes the capital intensity of the production line and lowers the operational expenditure related to catalyst recovery and regeneration. The simplified workup procedures, which rely on filtration and pulping rather than complex extractions or distillations, further contribute to cost savings by reducing solvent usage and energy consumption during the isolation phases. These cumulative efficiencies create a lean manufacturing profile that is highly competitive in the global marketplace for fine chemical intermediates.
- Enhanced Supply Chain Reliability: The reliance on common industrial solvents such as methanol, toluene, and acetic acid ensures that production is not held hostage by the availability of niche or specialty reagents that often suffer from supply shortages. The modular nature of the synthesis allows for flexible production scheduling, where intermediates can be stockpiled at various stages to buffer against demand fluctuations or upstream supply delays. Additionally, the high stability of the intermediates reduces the risk of degradation during storage and transport, enabling manufacturers to maintain strategic inventory levels without compromising product quality. This resilience is crucial for maintaining uninterrupted supply to pharmaceutical clients who operate on tight just-in-time delivery schedules.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily transferable from pilot plant to multi-ton commercial reactors without significant re-engineering. The absence of heavy metal waste streams simplifies the effluent treatment process, allowing facilities to meet increasingly stringent environmental regulations with lower investment in wastewater treatment infrastructure. The use of iron powder, which generates benign iron oxide sludge, and the recycling of solvents like xylene and toluene further enhance the green chemistry profile of the manufacture. This alignment with sustainable manufacturing principles not only mitigates regulatory risk but also enhances the brand reputation of suppliers who can certify their products as being produced via environmentally responsible methods.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, providing clarity on its operational feasibility and strategic value for industry stakeholders. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: Why is the new synthesis route safer than the conventional method?
A: The conventional method relies on potassium permanganate oxidation, which poses explosion risks and generates heavy metal waste. The new route utilizes iron powder reduction and phosphorus pentoxide dehydration, significantly improving process safety and environmental compliance.
Q: What is the overall yield advantage of this patented process?
A: The patented process achieves a total yield of approximately 66.6 percent, with individual step yields consistently exceeding 90 percent in optimized conditions, offering a substantial improvement over the low-yield oxidation steps of prior art.
Q: Is this intermediate suitable for large-scale API manufacturing?
A: Yes, the process uses common industrial solvents like methanol, toluene, and acetic acid, and avoids exotic catalysts, making it highly scalable for commercial production of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-3-methoxy-5-bromoxynil Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to a superior synthetic route is only valuable if it can be executed with precision and consistency at a commercial scale. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in every batch we deliver. We operate stringent purity specifications and utilize rigorous QC labs equipped with advanced analytical instrumentation to verify that every shipment of 2-amino-3-methoxy-5-bromoxynil meets the exacting standards required for GMP pharmaceutical manufacturing. Our commitment to quality assurance extends beyond simple testing; we actively monitor critical process parameters to prevent the formation of genotoxic impurities and ensure batch-to-batch reproducibility that our partners can rely on.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis can be integrated into your supply chain to drive efficiency and reduce costs. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic impact of switching to this greener, high-yield route for your specific application. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your project timelines, ensuring a seamless transition to a more robust and sustainable sourcing strategy for your critical intermediates.
