Optimizing Pharmaceutical Intermediate Production via One-Step Lewis Acid Catalysis
Optimizing Pharmaceutical Intermediate Production via One-Step Lewis Acid Catalysis
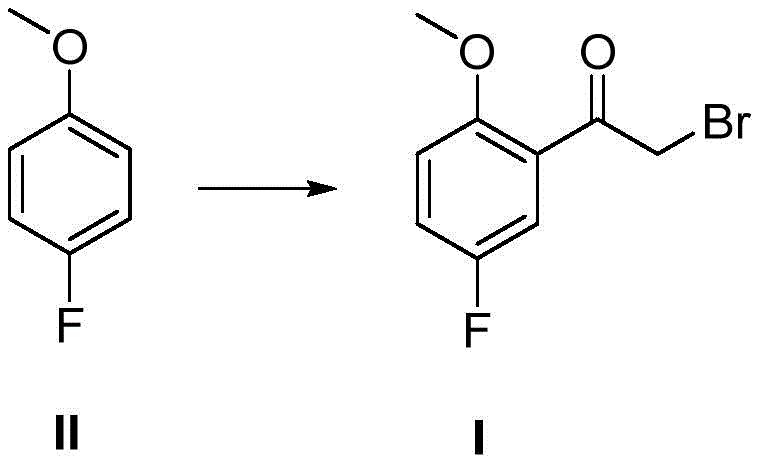
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the urgent need for more sustainable and cost-effective synthetic routes. A pivotal advancement in this domain is detailed in patent CN113354521A, which discloses a highly efficient preparation method for 2-methoxy-5-fluoro bromoacetophenone. This compound serves as a critical building block in the synthesis of various bioactive molecules, necessitating a robust supply chain capable of delivering high-purity materials consistently. The patented technology fundamentally shifts the paradigm from traditional multi-step sequences to a streamlined one-step process, utilizing p-fluoroanisole as a readily available starting material. By reacting this substrate directly with bromoacetyl bromide under controlled Lewis acid conditions, the invention achieves a remarkable simplification of the workflow. This technical breakthrough not only addresses the environmental concerns associated with hazardous reagents but also establishes a new benchmark for operational efficiency in fine chemical production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
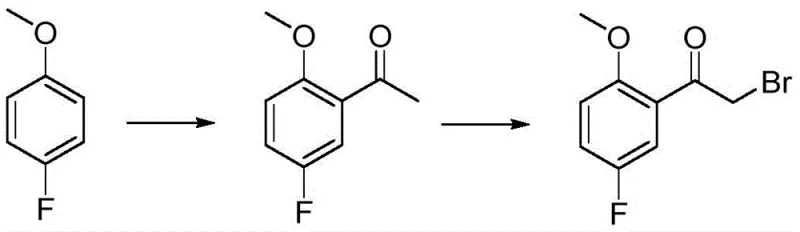
Historically, the synthesis of complex halogenated acetophenones has been plagued by inefficiencies inherent in multi-step protocols. As illustrated in prior art references such as WO2019/63748, the conventional pathway typically involves an initial acylation followed by a separate, distinct bromination step. This traditional approach often relies on the use of cuprous bromide or elemental bromine for the halogenation phase, introducing significant safety hazards and environmental burdens. The requirement to isolate and purify the intermediate ketone before proceeding to bromination inherently caps the overall yield due to mechanical losses during isolation. Furthermore, the use of stoichiometric amounts of toxic heavy metal salts or volatile halogens complicates waste stream management, leading to elevated disposal costs and regulatory scrutiny. These factors collectively render the conventional method less attractive for modern, large-scale manufacturing where green chemistry principles and cost containment are paramount.
The Novel Approach
In stark contrast to the cumbersome legacy methods, the novel approach disclosed in CN113354521A leverages the dual functionality of bromoacetyl bromide to effect both acylation and bromination in a single operational unit. This strategy capitalizes on the high reactivity of the acid bromide moiety towards electrophilic aromatic substitution, facilitated by a potent Lewis acid catalyst. By merging two chemical transformations into one pot, the process eliminates the need for intermediate isolation, thereby preserving material throughput and minimizing solvent consumption. The direct introduction of the bromoacetyl group onto the electron-rich aromatic ring of p-fluoroanisole is achieved with high regioselectivity, favoring the position ortho to the methoxy group. This consolidation of steps not only accelerates the production timeline but also drastically reduces the physical footprint required for manufacturing, offering a compelling value proposition for procurement teams seeking to optimize their supply chains for high-purity pharmaceutical intermediates.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The core of this technological advancement lies in the precise manipulation of electrophilic aromatic substitution dynamics. The reaction mechanism initiates with the coordination of the Lewis acid, such as aluminum trichloride or trifluoromethanesulfonic acid, to the carbonyl oxygen of the bromoacetyl bromide. This coordination significantly enhances the electrophilicity of the carbonyl carbon, generating a highly reactive acylium ion species. The aromatic ring of p-fluoroanisole, activated by the electron-donating methoxy group, attacks this electrophile preferentially at the ortho position relative to the methoxy substituent, despite the presence of the fluorine atom. The fluorine atom, being weakly deactivating yet ortho/para directing, does not significantly hinder the attack at the desired position due to the strong activation provided by the methoxy group. This electronic interplay ensures that the major product is the desired 2-methoxy-5-fluoro isomer, minimizing the formation of regioisomeric impurities that would otherwise complicate downstream purification efforts.
Impurity control is further enhanced by the mild reaction conditions employed, typically ranging from 20°C to 40°C. Maintaining the temperature within this narrow window prevents the occurrence of poly-acylation or unwanted side reactions that could degrade the product quality. The choice of solvent plays a critical role as well; chlorinated solvents like dichloromethane or carbon disulfide provide an ideal medium for stabilizing the ionic intermediates without participating in side reactions. Following the reaction completion, monitored rigorously via HPLC to ensure full conversion of the starting material, the workup procedure is designed to remove residual Lewis acids and unreacted reagents efficiently. The quenching into ice water followed by pH adjustment ensures the decomposition of the Lewis acid complex, while subsequent extraction and recrystallization from n-hexane yield a product with purity exceeding 99%, meeting the stringent specifications required for reliable API intermediate suppliers.
How to Synthesize 2-Methoxy-5-Fluoro Bromoacetophenone Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and thermal management to maximize yield and safety. The process begins with the preparation of the reaction mixture under anhydrous conditions to prevent premature hydrolysis of the sensitive bromoacetyl bromide. Operators must ensure that the Lewis acid is added in a slight molar excess relative to the substrate to drive the equilibrium towards product formation effectively. Throughout the addition of the acylating agent, vigorous stirring is essential to maintain homogeneity and dissipate the heat of reaction, preventing localized hot spots that could lead to degradation. Once the reaction is deemed complete by analytical tracking, the quenching step must be performed cautiously to manage the exotherm associated with acid neutralization. The final purification via recrystallization is a critical control point that determines the final crystal habit and purity profile of the bulk material.
- Combine p-fluoroanisole, a chlorinated solvent, and a Lewis acid catalyst such as aluminum trichloride in a reaction vessel.
- Add bromoacetyl bromide gradually and maintain the reaction temperature between 20°C and 40°C until HPLC indicates complete conversion.
- Quench the reaction with ice water, extract the organic layer with dichloromethane, and purify the crude product via n-hexane recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this one-step synthesis methodology translates into tangible strategic benefits that extend beyond mere chemical efficiency. The elimination of the second synthetic step removes an entire layer of logistical complexity, reducing the number of raw materials that need to be sourced, qualified, and stocked. This simplification inherently lowers the risk of supply disruptions caused by the shortage of specialized reagents like cuprous bromide, which can be subject to market volatility. Furthermore, the reduction in processing time means that manufacturing cycles are shorter, allowing for more responsive production scheduling and the ability to react swiftly to fluctuating market demands. The overall simplification of the process flow also implies a reduction in labor hours and utility consumption per kilogram of product, contributing to a leaner and more cost-competitive manufacturing model.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the removal of expensive and toxic brominating agents required in the second step of conventional routes. By utilizing p-fluoroanisole, a commodity chemical with stable pricing, as the sole starting material, the raw material cost base is significantly lowered. Additionally, the avoidance of heavy metal catalysts eliminates the need for costly scavenging steps and specialized waste treatment protocols, resulting in substantial operational expenditure savings. The higher overall yield achieved through the one-pot strategy means that less starting material is wasted, further enhancing the cost-efficiency of the production campaign and providing a competitive edge in pricing negotiations.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the robustness of this method offers superior reliability. The reliance on widely available solvents and catalysts reduces dependency on niche suppliers, mitigating the risk of bottlenecks. The simplified workflow reduces the potential for human error during intermediate transfers and isolations, leading to more consistent batch-to-batch quality. This consistency is crucial for maintaining long-term contracts with pharmaceutical clients who require unwavering quality standards. Moreover, the reduced processing time allows for increased throughput within existing facility constraints, effectively expanding capacity without the need for capital-intensive infrastructure upgrades.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of hazardous gas evolution or highly exothermic events that are difficult to control at scale. The use of standard extraction and crystallization techniques ensures that the process can be easily adapted to existing multipurpose reactors. Environmentally, the process aligns with green chemistry initiatives by minimizing waste generation and avoiding the use of persistent organic pollutants or heavy metals. This compliance facilitates smoother regulatory approvals and reduces the environmental liability associated with chemical manufacturing, making it a sustainable choice for long-term production partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2-methoxy-5-fluoro bromoacetophenone using this advanced methodology. These insights are derived directly from the experimental data and technical disclosures found in the patent literature, providing a transparent view of the process capabilities. Understanding these details helps stakeholders make informed decisions regarding sourcing strategies and technical feasibility for their specific applications.
Q: How does the new one-step method improve yield compared to traditional routes?
A: The novel one-step route avoids the yield losses associated with isolating intermediate compounds and eliminates side reactions common in separate bromination steps, achieving yields over 68%.
Q: What are the primary cost drivers eliminated in this synthesis?
A: By bypassing the need for toxic brominating agents like cuprous bromide or elemental bromine in a second step, the process significantly reduces reagent costs and waste disposal expenses.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the use of common solvents like dichloromethane and standard Lewis acids, combined with a simple workup procedure involving extraction and recrystallization, facilitates easy industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methoxy-5-Fluoro Bromoacetophenone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent theory to commercial reality requires deep technical expertise and rigorous quality control. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this innovative one-step synthesis are fully realized at an industrial level. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to manage complex chemistries allows us to offer a secure supply of critical building blocks, minimizing the risk of production delays for our global partners.
We invite you to collaborate with us to leverage this cost-effective synthesis route for your pharmaceutical projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this process can optimize your bill of materials. We encourage you to contact us to request specific COA data and route feasibility assessments, ensuring that our solutions align perfectly with your development timelines and quality expectations. Let us be your partner in driving efficiency and innovation in your supply chain.
