Advanced One-Step Synthesis of N-phenyl-3-(benzotriazole-1-yl)propionamide for Industrial Scale-up
Introduction to Green Synthesis Technologies
The landscape of fine chemical manufacturing is undergoing a significant transformation, driven by the urgent need for safer, more efficient, and environmentally benign synthetic routes. A pivotal development in this domain is documented in patent CN113135865B, which discloses a robust method for preparing N-phenyl-3-(benzotriazole-1-yl)propionamide. This compound serves as a versatile intermediate with applications ranging from vapor phase corrosion inhibitors to polymer additives and potential pharmaceutical agents. The disclosed methodology represents a paradigm shift from conventional toxic pathways, utilizing a hypervalent iodine-mediated oxidative coupling strategy. By leveraging 3-methylthio-N-phenylpropionamide and benzotriazole as primary feedstocks, this process achieves a remarkable isolated yield of 70% under relatively mild conditions. For R&D directors and procurement specialists, this innovation offers a compelling alternative that balances high purity standards with operational safety, addressing critical pain points in the supply chain of complex heterocyclic derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-alkyl benzotriazole derivatives, including analogues like 3-benzotriazole propionamide, has relied heavily on the use of acrylamide as a key starting material. While chemically effective, acrylamide is classified as a highly toxic substance with severe health implications, including neurotoxicity, liver injury, and skin corrosion upon exposure. The reliance on such hazardous precursors imposes substantial regulatory burdens and safety costs on manufacturing facilities, necessitating specialized containment systems and rigorous waste treatment protocols. Furthermore, reactions involving acrylamide often require specific catalysts like Triton B and can suffer from selectivity issues, leading to complex impurity profiles that are difficult and costly to remove. These factors collectively hinder the scalability and economic viability of traditional routes, creating bottlenecks for reliable pharmaceutical intermediate supplier networks seeking to minimize liability and environmental impact.
The Novel Approach
In stark contrast, the innovative pathway described in the patent data circumvents these hazards entirely by employing a thioether-based strategy. The reaction utilizes 3-methylthio-N-phenylpropionamide, a significantly safer and more stable precursor, which undergoes oxidative desulfurization to form the desired C-N bond. This one-step transformation is catalyzed by iodobenzene acetate in the presence of a base, proceeding smoothly in acetonitrile at 120 °C. The elegance of this approach lies in its simplicity and atom economy; it avoids the generation of toxic byproducts associated with acrylamide polymerization or degradation. As illustrated in the reaction scheme below, the direct coupling allows for a streamlined workflow that reduces unit operations and simplifies downstream processing.
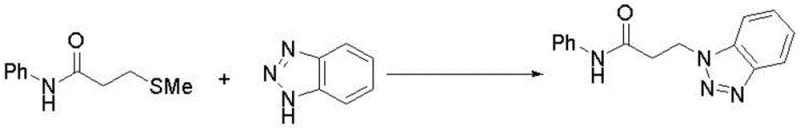
This methodological advancement not only enhances operator safety but also facilitates cost reduction in fine chemical manufacturing by eliminating the need for expensive toxicity mitigation infrastructure. The ability to synthesize high-purity N-phenyl-3-(benzotriazole-1-yl)propionamide in a single pot represents a significant leap forward in process chemistry, offering a scalable solution for the production of corrosion inhibitors and functional additives.
Mechanistic Insights into Hypervalent Iodine-Mediated Coupling
The core of this synthetic breakthrough relies on the unique reactivity of hypervalent iodine(III) species, specifically iodobenzene acetate (PhI(OAc)2). Mechanistically, the reaction is proposed to proceed through an oxidative activation of the sulfur atom in the thioether substrate. The electrophilic iodine center coordinates with the sulfur lone pair, generating a reactive sulfonium-like intermediate that significantly weakens the adjacent carbon-sulfur bond. This activation renders the beta-carbon susceptible to nucleophilic attack by the nitrogen atom of the benzotriazole ring. The subsequent elimination of the sulfur moiety, likely as a sulfoxide or sulfone byproduct, drives the formation of the new C-N bond. This mechanism is distinct from traditional nucleophilic substitution reactions, as it does not require harsh leaving groups or extreme temperatures that could degrade sensitive functional groups. The use of PhI(OAc)2 ensures a clean oxidation profile, minimizing side reactions such as over-oxidation of the amide backbone.
Furthermore, the choice of base plays a pivotal role in modulating the reaction kinetics and final yield. Experimental optimization revealed that while various bases could facilitate the reaction, potassium carbonate (K2CO3) provided the optimal balance of basicity and solubility in acetonitrile. As demonstrated in the comparative data, the absence of the oxidant leads to a negligible yield of 6%, while the absence of base results in only 10% conversion, underscoring the synergistic requirement of both components. The optimal conditions, utilizing 1.0 equivalent of PhI(OAc)2 and 3.0 equivalents of K2CO3, drive the reaction to a 70% isolated yield, as shown in the detailed reaction conditions below. This high efficiency is crucial for maintaining high-purity pharmaceutical intermediates standards, as it limits the formation of unreacted starting materials and complex side products that complicate purification.
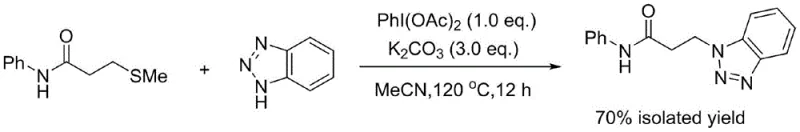
How to Synthesize N-phenyl-3-(benzotriazole-1-yl)propionamide Efficiently
Implementing this synthesis on a pilot or commercial scale requires strict adherence to the optimized parameters identified in the patent literature to ensure reproducibility and safety. The process is designed to be operationally simple, involving the sequential addition of reagents into a sealed vessel followed by thermal activation. The use of acetonitrile as the solvent provides an excellent medium for dissolving both the organic substrates and the inorganic base, ensuring homogeneous reaction conditions. It is imperative to maintain the reaction temperature at 120 °C, as lower temperatures may result in incomplete conversion, while higher temperatures could risk solvent pressure issues in sealed systems. The detailed standardized synthesis steps, including precise molar ratios and workup procedures, are outlined in the guide below to assist technical teams in replicating this high-yielding protocol.
- Combine 3-methylthio-N-phenylpropionamide, benzotriazole, iodobenzene acetate, and potassium carbonate in acetonitrile solvent within a sealed reaction vessel.
- Heat the reaction mixture to 120 °C and maintain vigorous stirring for a duration of 12 hours to ensure complete conversion.
- Upon completion, concentrate the reaction liquid and perform column chromatography separation to isolate the pure target product with high yield.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible strategic benefits beyond mere chemical curiosity. The primary advantage lies in the substantial reduction of regulatory and safety overheads. By eliminating acrylamide from the supply chain, companies can avoid the rigorous reporting, storage, and disposal costs associated with hazardous air pollutants and neurotoxins. This shift directly contributes to cost reduction in agrochemical intermediate manufacturing and related sectors where benzotriazole derivatives are used as stabilizers. Moreover, the one-pot nature of the reaction significantly shortens the production cycle time. Traditional multi-step syntheses often require isolation of intermediates, solvent swaps, and multiple purification stages, each adding days to the lead time. In contrast, this streamlined approach consolidates the transformation into a single thermal step, thereby enhancing throughput and reducing the overall manufacturing footprint.
- Cost Reduction in Manufacturing: The economic implications of this process are profound, primarily driven by the simplification of the operational workflow. By removing the need for toxic acrylamide, facilities save on the capital expenditure required for specialized ventilation and containment systems. Additionally, the high isolated yield of 70% minimizes raw material waste, improving the overall atom economy of the process. The reagents used, such as potassium carbonate and iodobenzene acetate, are commercially available commodity chemicals, ensuring stable pricing and avoiding the volatility associated with specialty catalysts. This stability allows for more accurate long-term budgeting and reduces the risk of cost overruns due to raw material scarcity.
- Enhanced Supply Chain Reliability: Supply continuity is a critical metric for global manufacturers, and this route enhances reliability by utilizing robust, shelf-stable starting materials. Unlike acrylamide, which requires careful temperature control during transport and storage to prevent polymerization, 3-methylthio-N-phenylpropionamide is a stable solid that is easier to handle and ship. This stability reduces the risk of supply disruptions caused by transportation incidents or storage degradation. Furthermore, the reaction conditions are tolerant and do not require exotic catalysts that might have long lead times or single-source dependencies. This resilience ensures that production schedules can be maintained consistently, reducing lead time for high-purity fine chemicals and enabling faster response to market demand fluctuations.
- Scalability and Environmental Compliance: From an environmental perspective, this method aligns perfectly with modern green chemistry mandates. The avoidance of heavy metal catalysts and toxic monomers simplifies wastewater treatment and reduces the burden on effluent processing units. The byproducts generated are generally less hazardous and easier to treat than those from acrylamide-based routes. This compliance ease facilitates smoother permitting processes for plant expansions or new facility constructions. The scalability of the reaction has been validated through the patent examples, demonstrating that the chemistry holds up under the defined conditions, paving the way for the commercial scale-up of complex heterocyclic intermediates without the typical teething problems associated with process intensification.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the experimental data and background analysis provided in the patent documentation, offering clarity on reaction optimization and safety profiles. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their specific production needs.
Q: Why is this new synthesis method preferred over traditional acrylamide routes?
A: Traditional methods often rely on acrylamide, a known neurotoxin that poses significant safety risks and requires stringent handling protocols. This novel approach utilizes 3-methylthio-N-phenylpropionamide, eliminating the need for highly toxic precursors and aligning with modern green chemistry principles for safer industrial manufacturing.
Q: What represents the optimal base for this oxidative coupling reaction?
A: Experimental data indicates that potassium carbonate (K2CO3) provides superior results compared to other bases like sodium carbonate or cesium carbonate. While cesium carbonate showed moderate activity (48% yield), potassium carbonate achieved the highest isolated yield of 70%, making it the most economically viable choice for large-scale production.
Q: Is the hypervalent iodine oxidant essential for reaction success?
A: Yes, the presence of iodobenzene acetate (PhI(OAc)2) is critical. Control experiments demonstrate that omitting the oxidant results in a drastic drop in yield to merely 10%, confirming that the oxidative activation of the sulfur moiety is the key driving force for the C-N bond construction.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-phenyl-3-(benzotriazole-1-yl)propionamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to stay competitive in the global fine chemicals market. Our technical team has thoroughly analyzed the potential of the hypervalent iodine-mediated coupling route described in CN113135865B and is prepared to support its industrial realization. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of N-phenyl-3-(benzotriazole-1-yl)propionamide meets the highest international standards for pharmaceutical and industrial applications.
We invite you to collaborate with us to leverage this greener, more efficient synthesis technology. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage potential partners to contact us directly to request specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both your supply chain resilience and your bottom line. Let us help you secure a sustainable and cost-effective supply of this vital intermediate.
